Fig. 23.1
Drug fluency: a 1-min marker of processing bias in addiction. Subjects were asked to “call to mind and name as many drug-related words as possible for 1 min.” These could be “names of drugs, people, places, or states of mind related to getting, using, or recovering from drugs” (Goldstein et al. 2007d). While healthy controls (left) produced a mean of 14 correct words, cocaine-addicted individuals (current users: cocaine urine positive) (right) produced 17 words. In addition, qualitative differences were noted such that the addicted individuals reported more words related to paraphernalia and drug administration via smoking, while the control subjects reported more words related to nonaddictive prescription drugs (Image created from TagCrowd.com)
23.3 Use of Money in Neuroimaging Studies in Drug-Addicted Individuals
Bar some exceptions, functional magnetic resonance imaging (fMRI) studies report that compared to controls, drug-addicted individuals show enhanced fMRI blood oxygenation level-dependent responses in the PFC (and subcortical regions) to drug-related cues relative to control cues (Goldstein and Volkow 2011). Such cue-induced PFC responses correlate with craving (Brody et al. 2002) and severity of drug use (Yalachkov et al. 2009) and predict both subsequent performance on a primed emotion recognition task (Artiges et al. 2009) and drug use 3 months later (Grusser et al. 2004), indicating that these measures have clinical significance. Comparisons between these drug-related stimuli with the processing of nondrug reinforcers have not been frequently inspected. Given the conditioning between monetary availability and drug procurement, using money as a reinforcer in drug addiction research is particularly important. If processing of this secondary generalizable reinforcer is compromised in addicted individuals, it is possible that for this population, only more immediate drug-related cues [e.g., pictures or a video; see (Garavan et al. 2000)] or the drug itself could activate this circuit at a comparable level with that induced by a nondrug-related reward in the nondrug-addicted individual. Importantly, because money is an effective reinforcer that acquires its value by social interaction, its compromised processing would suggest a socially disadvantageous emotional learning mechanism in addiction. Such a deficit, all the more distinct given the strong motivational and arousal value that is normally associated with this reward, would corroborate the notion that in addiction, brain reward circuits are “hijacked” by drugs, although the possibility for a preexisting deficit in reward processing also cannot be ruled out.
We used fMRI to investigate how cocaine-addicted individuals and controls responded to receiving monetary reward for correct performance on a sustained attention/forced-choice task (Goldstein et al. 2007a). In controls, sustained monetary reward (gain that did not vary within task blocks and that was fully predictable) was associated with a robust and complex neuronal activation pattern: there was a trend for the left orbitofrontal (OFC) to respond in a graded fashion (high money > low money > no money), the lateral and medial PFC (including anterior cingulate cortex) responded instead to the two conditions of monetary value equally (high = low > no money), while the mesencephalon displayed a third pattern of sensitivity to the highest available reward only (high > low = no money). In general, these results were consistent with role of the (a) OFC in relative reward processing in the primate (Tremblay and Schultz 1999) and in healthy human subjects (Breiter et al. 2001; Elliott et al. 2003; Knutson et al. 2000; Kringelbach et al. 2003; O’Doherty et al. 2001), (b) PFC in the control of attention (Hornak et al. 2004) possibly irrespective of reward magnitude (Watanabe 1989), and (c) mesencephalon in an all-or-nothing reward processing in the primate (Tobler et al. 2005) and in healthy human subjects (Elliott et al. 2003). The cocaine-addicted subjects did not display this complex pattern of activation to monetary reward, demonstrating instead reduced fMRI signals in the left OFC for high gain compared to controls and showing less sensitivity to differences between the monetary rewards in the left OFC and in the dorsolateral PFC. Remarkably, more than half of the cocaine-addicted subjects rated the value of all monetary amounts equally (i.e., $10 = $1,000) (Goldstein et al. 2007b). Eighty-five percent of the variance in these ratings could be attributed to the lateral OFC and medial frontal gyrus (and amygdala) responses to monetary reward in the addicted subjects. Although these findings need to be replicated in a larger sample size and with more sensitive tasks, they nonetheless suggest that some cocaine-addicted individuals may have reduced sensitivity to relative differences in the value of rewards. Such “flattening” of the perceived reinforcer gradient (Fig. 23.2) may underlie overvaluation or bias toward immediate rewards (such as an available drug) (Roesch et al. 2006) and the discounting of greater but delayed rewards (Kirby and Petry 2004; Monterosso et al. 2007), therefore reducing sustained motivational drive.
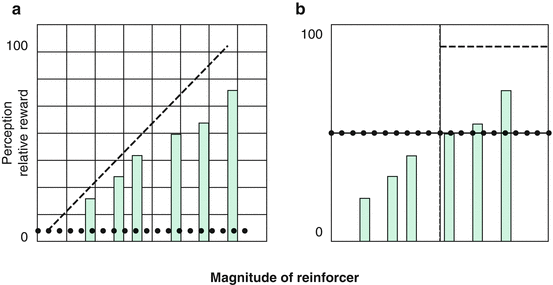

Fig. 23.2
Diagrammatic representation of the changes in relative and absolute reward in addiction. Dotted lines reflect the threshold for a stimulus to be perceived as reinforcing: the threshold is lower in the nonaddicted (a) and higher in the drug-addicted (b) individual. Dashed lines reflect the function that describes the perception of a stimulus as subjectively valuable. The high sensitivity to the reinforcers (small squares, a) allows the detection of small reinforcers and differences in magnitude between reinforcers in control subjects (monotonically positive function). The low sensitivity in cocaine abusers (large squares, b) does not permit the distinction between stimuli of different gradations but rather identify only those that reach the threshold required for the stimulus to be perceived as reinforcing (step function) (With permission from Goldstein et al. (2007b) Figure #4)
Using the same sustained attention task and monetary reward quantities for the high, low, and no gains (45 1, and 0¢) while recording ERPs, we replicated the impact of monetary reward on neural responses (measured here with the P300 ERP component) in healthy young adults (Goldstein et al. 2006). This result was consistent with a large body of literature implicating the P300 in processing reward magnitude and valence [e.g., (Yeung and Sanfey 2004)]. Importantly, we subsequently replicated these results in 18 healthy individuals matched on age and other demographic factors to 18 cocaine-addicted individuals (abstinence 0–4 days) (Goldstein et al. 2008): while in the control subjects the amplitude of the P300 component was higher in the 45¢ condition than the 0¢ condition, a similar P300 response to money was not significant in the cocaine-addicted subjects. In parallel, only the control subjects reacted faster to the highest monetary condition (45¢) as compared to the neutral cue (0¢). Importantly, only in the control subjects these P300 amplitude differentials were directly intercorrelated with the respective behavioral adjustments to the monetary incentive (45¢ > 0¢ with accuracy and 1¢ > 0¢ with reaction time); in the cocaine-addicted subjects, the better the accuracy adjustment for the high monetary condition, the less frequent the cocaine use during the year preceding the study. Our most recent results show that such compromised P300 responses to money characterize both cocaine urine-positive (current users, shorter abstinence) and cocaine urine-negative (longer abstinence) addicted individuals, who show the most compromised P300 amplitudes (Parvaz et al. 2012) as potentially driven by structural integrity of the PFC (Parvaz et al. 2011).
These results may be therapeutically relevant as monetary reinforcement in well-supervised environments has been shown to enhance drug abstinence (Kampman 2010) and may also be relevant in predicting clinical outcomes. Indeed, in a similar population of subjects, the degree of midcingulate hypoactivation in a task where correct performance was monetarily remunerated correlated with frequency of cocaine use, whereas the degree of rostroventral cingulate cortical hypoactivation correlated with task-induced craving (Goldstein et al. 2009a). There was also an inverse association between these PFC regions of interest with cue reactivity in the mesencephalon in cocaine-addicted but not in control subjects, which implicates these cingulate subdivisions in the regulation of automatic drug responses (Goldstein et al. 2009c). It remains to be determined whether treatment could normalize these neural responses to money as potentially associated with better clinical outcome. Promising lines of evidence indicate an association between the lowest self-reported alcohol craving with the highest VStr monetary gain activations in abstinent alcoholics (Wrase et al. 2007). Because this region is important for reward anticipation [e.g., (Knutson et al. 2005)] and for processing both gains and losses (gain > loss) (Wrase et al. 2007) in healthy control subjects, its normalization may be consequential for the processing of numerous reinforcers.
23.4 Other Nonmonetary Reward
Studies of neural responses to drug-related vs. nondrug-related cues (including potential reinforcers) in addicted vs. nonaddicted individuals have generally utilized electrophysiological recordings while subjects view drug-related, pleasant, unpleasant, and neutral pictures. Including nondrug emotional stimuli, in addition to the usual drug-related vs. neutral pictures, is important for emotional neuroscience studies in drug addiction (Aguilar de Arcos et al. 2005; Verdejo-Garcia et al. 2006). For example, we used an ERP component, the late positive potential (LPP), as a psychophysiological measure of automatic (Hajcak et al. 2007) or bottom-up (Ferrari et al. 2008) motivated attention bias elicited by drug-related stimuli in 55 cocaine-addicted individuals (27 abstinent and 28 current users) as compared to 29 matched controls (Dunning et al. 2011). Results showed that the LPPs elicited by cocaine pictures were similar to LPPs elicited by the other emotional pictures only in the addicted individuals; in the controls, LPPs elicited by the cocaine pictures were instead comparable to LPPs elicited by the neutral pictures, and both were significantly smaller than LPPs elicited by the other emotional pictures. These findings suggest that for the cocaine-addicted subjects, but not controls, cocaine stimuli automatically increase attention. This study also supports a relatively early attention bias to cocaine stimuli in the cocaine-addicted individuals, further suggesting that recent cocaine use may be associated with deficient sustained processing of any emotional stimuli. As mentioned above, a follow-up study showed these LPPs predict prospective choice (drug > pleasant for both) in cocaine-addicted individuals whose insight is compromised, of relevance to developing new intervention and prevention efforts (Moeller et al. 2012b). A study in active heroin-addicted individuals (24 h abstinence) reported similar drug-related P300 enhancements (Lubman et al. 2009) as correlated with baseline craving (Lubman et al. 2008). The more recent study (Lubman et al. 2009) also showed lack of the typical P300 reward enhancement to pleasant vs. neutral or drug pictures, consistent with inhibited responding to nondrug reinforcers in addicted individuals.
Compromised responsiveness to pleasant pictures in heroin-addicted individuals was similarly reported in an fMRI study, where the bilateral dorsolateral PFC was activated to pleasant pictures in 18 healthy controls but not in 16 abstinent (1–24 weeks) inpatient male heroin-addicted individuals (Zijlstra et al. 2009). Interestingly, in initially detoxified alcoholic subjects, VStr (and thalamic) response to pleasant vs. neutral stimuli predicted drinking days and alcohol intake within a 6-month follow-up period (Heinz et al. 2007). Taken together, the preserved responding to nondrug reinforcers may characterize individuals with less pronounced illness severity or reflect a protective factor in drug-addicted individuals. Indeed, offspring of alcoholics with higher striatal DA D2 receptor availability may be protected against developing alcoholism through more adaptive recruitment of corticolimbic circuits (including the OFC) needed for emotion regulation (Volkow et al. 2006).
23.5 The Underlying Mechanism for a Possible Reward Value Shift in Drug Addiction
Dopamine is an essential neurotransmitter in processing reward and reward prediction errors (McClure et al. 2004; Schultz 2006; Volkow et al. 1993) and in salience enhancement (Volkow et al. 2002b, 2004b). Positron emission tomography studies have documented that with chronic drug use striatal DA D2 receptor availability is reduced (Nader and Czoty 2005; Nader et al. 2006; Volkow et al. 1990, 1997b) as associated with blunted responses to drug rewards (e.g., intravenous methylphenidate) (Volkow et al. 1997a) and altered function in dopaminergically innervated corticolimbic areas (encompassing in the PFC the OFC and anterior cingulate cortex) that mediate processing of reward salience and motivation (McClure et al. 2004; Schultz 2006; Volkow et al. 1993).
The abnormalities in reward processing in drug addiction are therefore not surprising and are consistent with similar compromises in other dopaminergically mediated psychopathologies. For example, monetary incentives do not modulate grip force in patients with bilateral striatal-pallidal damage (Schmidt et al. 2008), and Parkinson’s disease patients are less proficient in learning the predictive value of monetary reward cues, displaying diminished functional connectivity of the mesencephalon and VStr (Schott et al. 2007). In healthy individuals, a preliminary pharmacological fMRI study suggested that a dopaminergic agent (0.25 mg/kg oral dextroamphetamine vs. placebo) modulated VStr responses during anticipation of gains such that responses were blunted in peak amplitude but extended in duration (Knutson et al. 2004). The association between DA and reward processing is thus not linear, and potentially modulated by baseline DA levels. For example, higher baseline striatal DA synthesis is associated with better reward learning but worse learning with further dopaminergic intervention (with bromocriptine) in healthy controls; in contrast, lower baseline striatal DA synthesis is associated with better punishment learning and DA enhancement improves reward learning (Cools et al. 2009). It is therefore possible that DA agonists could be used to improve reward processing in selected subgroups of drug-addicted individuals. Suggestive of the success of such an intervention, preliminary results in our laboratory showed a correlation between baseline DA receptor availability (measured with positron emission tomography with C11 raclopride) and thalamic and medial PFC response to money (measured with fMRI) in seven cocaine-addicted individuals (Asensio et al. 2009).
More direct evidence for the role of DA in the modified reward valuation in addicted individuals derives from another positron emission tomography study where blunted amphetamine-induced DA release in the VStr (and dorsal striatum) was predictive of actual choice for cocaine over money (and not of positive subjective drug effects) in 24 cocaine-addicted individuals (14 days abstinence) as compared to 24 controls (Martinez et al. 2007). These results were all the more compelling as subjects could choose to receive $5 or self-administer smoked cocaine (6 or 12 mg) with street value <$5. Although results of this study need to be validated using a more immediate monetary gain (the $5 gain was delayed, given as a merchandise voucher redeemable at local stores and paid upon discharge from the study), choice on this self-administration paradigm may model relapse (drug choice followed a priming dose/drug cue). In fact, the authors interpreted these results to indicate that the cocaine-addicted individuals who are most vulnerable to relapse are those with the lowest presynaptic DA function because their DA levels may be insufficient to provide the signal that could shift habitual behavior (drug choice, lesser reward) to a more advantageous behavior (monetary choice, greater reward). A follow-up fMRI study used a similar paradigm in cigarette smokers, where subjects could win cigarettes or money. Occasional smokers were more motivated to obtain money than cigarettes, whereas dependent smokers made similar efforts to win money or cigarettes (Buhler et al. 2010). A similar group by reward interaction was observed in the right OFC, bilateral dorsolateral PFC, and left anterior cingulate cortex, such that in the occasional smokers these regions showed higher activity to stimuli predicting an increasing monetary reward than to stimuli predicting a cigarette reward, whereas the dependent smokers showed no significant differences in such anticipatory brain activity. These regions also showed higher activation to money in the occasional than dependent smokers (Buhler et al. 2010). Dopaminergic agonists may reverse these abnormalities as remains to be studied.
23.6 Conflicting Results
Throughout most of this chapter, results have generally pointed to decreased valuation of nondrug reward in addiction. Indeed animal research suggests that after chronic drug administration, the value of saccharin is decreased (Grigson and Twining 2002). Similarly, human cocaine-addicted subjects but not controls showed reduced activation of corticolimbic brain areas when viewing an erotic video than when exposed to a cocaine video (Garavan et al. 2000). Yet evidence from animal studies also suggests that drug sensitization can increase the incentive value of other rewards, such as sucrose or other foods, a sexually receptive female (for male rats), and conditioned stimuli for such rewards (Fiorino and Phillips 1999a, b; Nocjar and Panksepp 2002; Taylor and Horger 1999; Wyvell and Berridge 2001). Similarly, in human addicted individuals, evidence suggests that some cocaine-addicted individuals are hypersexual (Washton and Stone-Washton 1993) and some substance-dependent individuals may be hyperresponsive to money rewards (Bechara et al. 2002). These latter lines of evidence are consistent with the notion of a generally drug-sensitized brain reward circuit where heightened drug motivation may “spillover” to nondrug rewards (Robinson and Berridge 2003). A resolution of this apparent conflict may reside in a better demarcation of the role different brain regions carry within the reward circuit [e.g., different brain regions may show the opposite patterns of response to reward (Konova et al. 2012)], types of drugs used (e.g., cocaine vs. heroin), or in other individual differences (e.g., severity of addiction, impulsivity) as remains to be studied.
23.7 Summary and Conclusions
Evidence for a modified valuation of monetary and other nondrug-related reward in drug-addicted individuals was reviewed. The selective diversion of motivational resources away from conventional rewards and toward drug rewards in addicted individuals offers strong support for the hypothesis that the value of drug-related cues overpowers that of nondrug-related cues in addicted individuals. These abnormalities may contribute to the deficits in controlling drug-taking behavior in drug-addicted individuals. For example, restricted range of subjective valuation of reward may play a mediating role in the ability to use internal cues and feedback from the environment to inhibit inappropriate (drug-escalated) behavior. Moreover, a “flattened” sensitivity to gradients in reward may predispose individuals to disadvantageous decisions (e.g., trading a car for a couple of cocaine hits). Without a relative context, drug use and its intense effects (craving and high) could become all the more overpowering.
Taken together, these results contribute to our understanding of how relative reward preferences may change in addiction, such that preference for the drug competes with (and sometimes exceeds) preference for other reinforcers, with a concomitant decrease in the ability to assign relative values to nondrug-related rewards. Insofar as emotions are defined as “states elicited by reinforcers” (Rolls 2000), such reward valuation abnormalities may contribute to pervasive disruptions in emotional processing in drug-addicted individuals, encompassing empathy (Kim et al. 2010), self-awareness and awareness of disease severity, and need of treatment (Goldstein et al. 2009b). Because reinforcement is crucial in most types of learning, these disruptions may also contribute to cognitive dysfunction spanning attention, working memory, decision-making, and inhibitory control (Goldstein et al. 2011), together threatening sustained abstinence (Garavan and Hester 2007), increasing attrition from treatment (Aharonovich et al. 2003, 2006), or even predisposing individuals to develop drug addiction (Moffitt et al. 2011; Tarter et al. 2003).
23.8 Future Directions
A lingering question is whether altered sensitivity to nondrug reinforcers in addicted individuals also applies to negative reinforcers such as money loss. Studies in animals show that “addicted” subjects manifest persistent drug seeking even if the drug is associated with receiving an electric shock (Deroche-Gamonet et al. 2004). In humans, hypoactivation in the right ventrolateral PFC in smokers during monetary loss, and in gamblers during monetary gain, has been reported (de Ruiter et al. 2009). Although more studies are clearly needed, the implication of reduced sensitivity to negative reinforcers in addiction has practical implications as, in addition to positive reinforcers (such as vouchers and privileges), negative reinforcers (such as incarceration) are increasingly being used in the management of drug abusers.
Future studies could also help to ascertain whether addicted individuals may resort to taking drugs because they are easily bored, frustrated, angry or fearful, perhaps as a result of altered reward valuation. Low threshold for experiencing any of these emotions, or the inability to sustain goal-directed behavior (e.g., complete a boring task) especially when experiencing these emotions, may be associated with impaired inhibitory control (i.e., enhanced impulsivity). In cocaine-addicted individuals, PFC activity habituates prematurely to repeated presentation of an incentive-sustained attention task (Goldstein et al. 2007c), in the absence of a compensatory response in the dopaminergic midbrain during such mental fatigue (Moeller et al. 2012c), which could be indicative of compromised sustainability of effort and may result in inadequate engagement in treatment activities and relapse.
Future studies need to identify the drug-addicted individuals who evidence a compromised sensitivity to nondrug reward for tailored interventions to improve associated cognitive-emotional skills (e.g., attention, response shifting, learning and memory, general value estimations) with the goal of modulating choice behavior such that nondrug rewards would be chosen over drug use. In this quest, one could explore promising new approaches that have been shown to modify behavior and decision-making [e.g., virtual environments and computer games that can acquire persistent motivational properties (McCabe et al. 2009)]. Direct brain stimulation or pharmacological enhancement of the dopaminergically innervated corticolimbic brain regions may also be beneficial, especially if combined with targeted cognitive-behavioral exercises (Goldstein et al. 2010a; Moeller et al. 2012a, c) or in a more sustained fashion during resting state (Konova et al. 2013), for neural rehabilitation purposes (Vinogradov et al. 2011).
Time dedicated to working on this chapter was supported by grants from the National Institute on Drug Abuse (1R01DA023579).
References
Aguilar de Arcos F, Verdejo-Garcia A, Peralta-Ramirez MI, Sanchez-Barrera M, Perez-Garcia M (2005) Experience of emotions in substance abusers exposed to images containing neutral, positive, and negative affective stimuli. Drug Alcohol Depend 78:159–167PubMed
Aharonovich E, Nunes E, Hasin D (2003) Cognitive impairment, retention and abstinence among cocaine abusers in cognitive-behavioral treatment. Drug Alcohol Depend 71(2):207–211PubMed
Aharonovich E, Hasin DS, Brooks AC, Liu X, Bisaga A, Nunes EV (2006) Cognitive deficits predict low treatment retention in cocaine dependent patients. Drug Alcohol Depend 81(3):313–322PubMed
Ahmed SH, Koob GF (1998) Transition from moderate to excessive drug intake: change in hedonic set point. Science 282(5387):298–300PubMed
Ahmed SH, Kenny PJ, Koob GF, Markou A (2002) Neurobiological evidence for hedonic allostasis associated with escalating cocaine use. Nat Neurosci 5(7):625–626PubMed
Aigner TG, Balster RL (1978) Choice behavior in rhesus monkeys: cocaine versus food. Science 201(4355):534–535PubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





