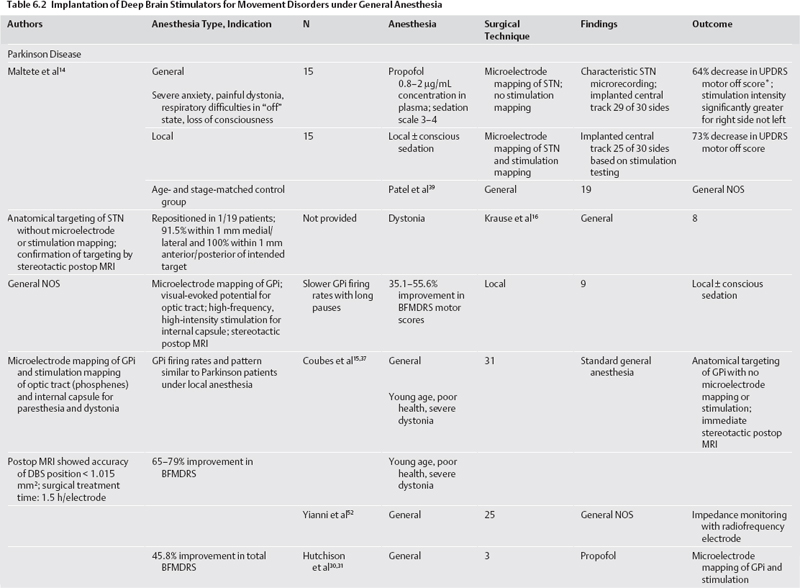
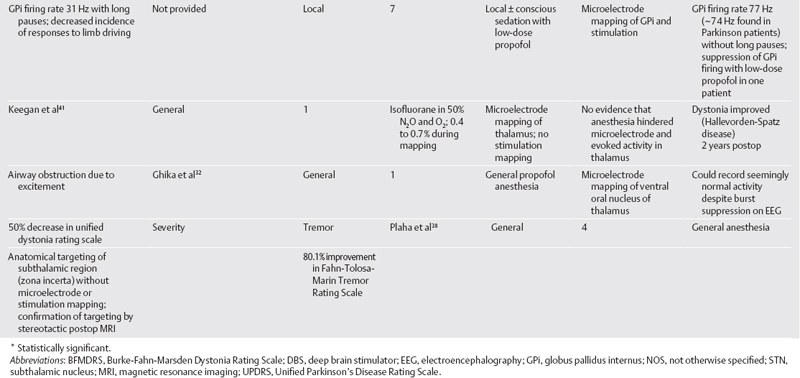
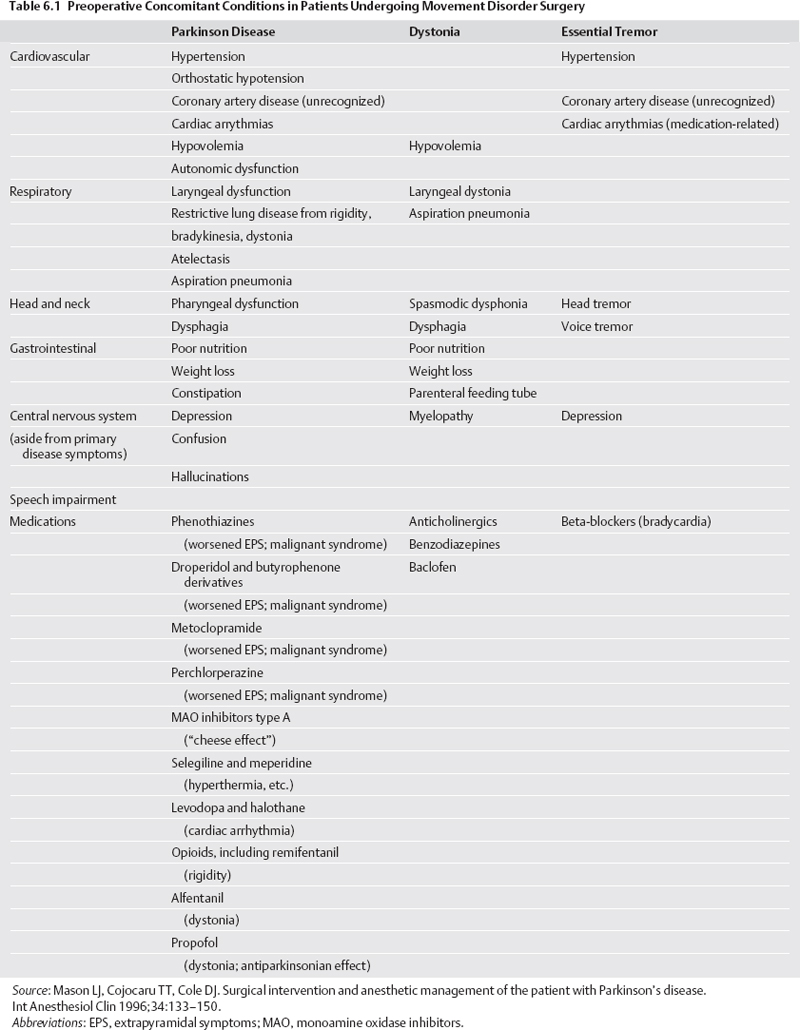
6 Anesthesia for Movement Disorder Surgery Robert E. Gross, Klaus Mewes, and Ghaleb Ghani Surgical treatment of Parkinson disease (PD) and other movement disorders has been performed with local anesthesia since shortly after the advent of stereotactic functional neurosurgery.1 Electrophysiological mapping, testing the effects of a radio frequency (RF) lesion, and electrical stimulation testing, both prior to an RF lesion and as part of implantation of a deep brain stimulator (DBS) lead, are all facilitated by having an awake and responsive patient. However, increasing experience and technology and expanding indications are driving a small but increasing number of centers to perform surgery under general anesthesia. The important role of the anesthetist in intraoperative monitoring of local anesthetic procedures is thus expanding to more difficult monitoring of the anesthetized patient, often in more difficult settings such as the intraoperative magnetic resonance imaging (MRI) or the radiology suite. This chapter reviews anesthetic procedures for surgery on a difficult, often elderly but occasionally pediatric, patient population, before, during, and after the operating room. We also address the use of electrophysiological mapping techniques under general anesthesia. Specific considerations relating to the most common indications for functional stereotactic movement disorder surgery—PD, essential tremor (ET), and dystonia—are highlighted. There are unique challenges in treating patients undergoing movement disorder surgery. PD and ET patients are often elderly, which can predispose them to cardiac, respiratory, and other age-related complications. In addition, these patients are usually on several medications related to their movement disorder and other health issues, and such polypharmacy may lead to important drug interactions affecting surgery.2 Older patients may also have differences in pharmacodynamics and pharmacokinetics affecting many drugs, including those used for anesthesia and sedation (e.g., benzodiazepines).2,3 It is therefore extremely important that each patient undergo a careful preoperative screening to identify potential risk factors for surgery and drug interactions (Table 6.1). Cardiovascular risks are common in this patient population. Hypertension must be well treated prior to surgery because of the risk of hemorrhage.4,5 There should be a low threshold to obtain a thallium stress test prior to surgery because ischemic disease may go unnoticed in PD patients who are limited in their mobility. PD patients also commonly suffer from orthostatic hypotension, contributed to by use of L-dopa and dopamine agonists, as well as other autonomic disturbances.2,3,6 In addition, they can be hypovolemic, malnourished, and debilitated. Respiratory dysfunction is well known in PD.2 This includes an obstructive ventilatory pattern, dysfunction of upper airway musculature, rigidity, bradykinesia, and dystonia of respiratory muscles.7 Disturbances of posture and muscle tone can cause atelectasis, restrictive lung disease, and loss in lung volume. These problems are only exacerbated by withdrawal from antiparkinsonian medications. PD patients with significant respiratory issues are predisposed to aspiration in the postoperative period. Cognitive issues affect many PD patients; although frank dementia excludes patients from surgery, nondemented patients may have decreased mental stamina or excessive anxiety that may be indications to perform surgery under general anesthesia.8 Patients with ET are generally in better physical health than those with PD. However, lifestyle issues related to the disability may lead to poor health. General issues related to older age pertain to this group (we have operated on several patients > 75 years of age with ET), including decline in cognitive function. Possible medication interactions should be noted: most patients will be on a badrenergic antagonist such as propanolol, and possibly mysoline, predisposing them to hypotension and bradycardia during surgery. In general, dystonic patients are younger than most PD and ET patients, and many patients with idiopathic torsion dystonia will be pediatric.9,10 Severe dystonia can lead to extreme weight loss, malnutrition, and debilitation, with attendant anesthetic risks. Cervicoaxial involvement may cause respiratory difficulties and, with longstanding disease, spondylosis, kyphoscoliosis, and myelopathy. Careful attention must be directed at determining whether the severity of the dystonia, especially in the cervical and thoracic regions, makes the patient a poor risk for undergoing surgery under local anesthesia. It is not uncommon to operate on these patients under conscious sedation or general anesthesia (see later discussion). It is well known that drugs with dopamine antagonist actions can exacerbate PD and even precipitate “malignant syndrome.”11 These drugs include phenothiazines, butyrophenone derivatives (e.g., droperidol), and other gastrointestinal drugs, including metoclopramide (Reglan, Baxter Healthcare Corp., Deerfield, IL) and prochlorperazine (Compazine, GlaxoSmithKline, Middlesex, England). Monoamine oxidase inhibitors (MAOIs) have multiple potential drug interactions. The commonly used type B MAOIs (e.g., selegiline), do not precipitate tyramine crisis (the “cheese effect”) like type A MAOIs, but rigidity, hyperpyrexia, sweating, and agitation have been reported following meperidine in patients on selegiline; this combination should therefore be avoided.12 Meperidine (Demerol [Sanofi–Aventis, Bridge-water, NJ]) and other opiates may worsen rigidity in both healthy and PD patients. Alfentanil has been reported to cause an acute dystonic reaction in an undertreated PD patient.2 This problem has not been encountered with the use of remifentanil,13 although at high doses it can cause rigidity in any patient.14 PD patients are particularly sensitive to central nervous system (CNS) depressants; thus hypnotics and narcotics should be used judiciously.15 Anesthetics can interact with parkinsonian medications. Halothane can lead to dysrhythmias in patients on L-dopa by sensitizing the heart to catecholamines.3 Propofol is generally well tolerated during surgery8,16 but can be unpredictable in PD.17 It can be antiparkinsonian or prodyskinetic or both, which may interfere with surgical treatment if clinical response to stimulation or an RF lesion is to be evaluated in the responsive patient.18,19 The main drug interaction to be concerned with in patients with ET is related to their use of propanolol, which is a central Β-blocker, and care should be taken with the use of other antihypertensives. Propofol (Diprivan, B. Braun Melsungen AG, Melsungen, Germany) has been reported to precipitate either or both dystonic and myoclonic reactions in healthy patients emerging from anesthesia.19,20 Although isolated patients with dystonia have experienced exacerbation of symptoms with propofol,21 we and others have used propofol in dystonic patients with minimal adverse effects (see Table 6.2). Medication interactions should be scrutinized because many patients are on anticholinergics and benzodiazepines, and possibly dopamine antagonists or baclofen. Anticholinergic drugs can predispose to urinary retention, mouth dryness, constipation, and tachycardia. Centrally acting anticholinergics used in anesthetic practice can have additive effects.2 Most centers use extensive intraoperative electrophysiological mapping and/or clinical testing of benefits and adverse reactions to electrical stimulation when implanting a DBS or creating an RF lesion. These approaches require an awake, responsive, and reliable patient, most critically at the time of stimulation testing or RF lesion production. For this reason the majority of procedures are done with local anesthetic only, or with some degree of conscious sedation with a rapidly reversible agent such as remifentanil (often with midazolam)13,16 or low infusion of intravenous propofol because of its rapid onset and emergence.8,22 However, patients may not tolerate a lengthy awake surgical procedure in the setting of (1) advanced age, (2) mild dementia, (3) pain, (4) adverse health conditions or respiratory difficulties related to the “off medication” state,8 (5) severe torsion dystonia or choreoathetosis with resultant risk of pulling out of the frame,9,10,22 or (6) pediatric age.9,10,22,23 Such patients are operated on under deep sedation, possibly with reversal of the sedation for intraoperative testing at intervals,23,24 or under general anesthesia throughout (Table 6.2). The latter has been facilitated by the advent of DBS, where the position of the lead can be checked prior to removal of the stereotactic frame, and in the worst case, the lead can be repositioned. In contrast, surgeons are less comfortable creating a permanent lesion without having a responsive patient to test during the process. Irrespective of the type of anesthesia the patient is to undergo, a careful evaluation of the airway according to the American Society of Anesthesiology (ASA) Guidelines25 is important to predict the ease of mask ventilation and endotracheal intubation. If general anesthesia is planned, or if the patient has a high-risk airway that may create a higher risk for respiratory compromise intraoperatively (e.g., history of sleep apnea) then a frame with an intubation ring should be used to allow greater airway access, and the frame is positioned with the anesthesia mask in place to ensure the ability to ventilate the patient with the mask before securing the airway. In most cases, local anesthesia with mild sedation suffices for placement of the stereotactic frame. We routinely use a cocktail of 1% lidocaine, 0.5% bupivacaine, and 1:10 bicarbonate (to decrease the discomfort of the injection in part related to its acidity), infiltrated directly into the pin sites; however, a ring block approach can also be employed. Midazolam and fentanyl are administered prior to local injections. This approach is well tolerated by the vast majority of patients. It is important to anticipate the need for sedation during the imaging or the procedure itself, with possible airway issues, and apply the frame with this in mind. If general anesthesia is to be induced, it can be done either prior to frame placement or in the operating room after imaging. The former helps to achieve a motionless magnetic resonance imaging (MRI) scan. There is increasing use of frameless DBS placement, which actually involves use of a miniframe and fiducials that, rather than being attached to the frame, are implanted into the patient’s skull up to 1 week prior to the actual surgery.26 The fiducials are implanted with local anesthetic with or without light sedation. If the scan is to be obtained with deeper sedation, the patient may be intubated or have a laryngeal mask (LMA) inserted, which is certainly easier in the absence of the stereotactic frame. A high-quality MRI scan can be obtained in the vast majority of cases with minimal sedation. PD patients, when scanned on the morning of surgery, are scanned in the off medication state, which eliminates dyskinesia. Because tremor from any etiology disappears with sleep, a small dose of midazolam is usually all that is required, and only very rarely have we needed to use deeper sedation to counter tremor. However, patients with more extreme hyperkinetic movement disorders (e.g., severe generalized dystonia, cervical dystonia, choreoathetosis) or with excessive anxiety will often require deeper sedation with propofol or remifentanil, and intubation or insertion of a laryngeal mask. Although a high-quality MRI scan is always important, it is critical when the procedure is performed with only anatomical control and no electrophysiological mapping or intraoperative stimulation testing.27–29 Centers that perform surgery in this way generally obtain the MRI scan with the patient under general anesthesia to eliminate any movement artifact and obtain the highest-quality scan. When imaging the patient under general anesthesia, we prefer to use an LMA rather than endotracheal intubation. The patient does not require muscle relaxants and can be allowed to ventilate spontaneously in the scanner with monitoring. This avoids the need to bring the ventilator into the MRI chamber. Standard electrocardiography (ECG), pulse oximetry, and blood pressure monitoring are instituted. The pulse oximeter, blood pressure cuff, and intravenous (IV) line, if possible, should be on the side contralateral to the side that is to be tested during the surgery. Rest tremor can interfere with pulse oximetry and give the appearance of fibrillations on ECG. Systolic blood pressure is kept below 150 mm Hg with hydralazine or labetolol, although the latter is contraindicated in the setting of bradycardia, especially in ET patients taking propanolol. We do not typically use arterial monitoring of blood pressure. Oxygen is given via nasal cannula in all patients under local, with or without conscious sedation. We routinely monitor endtidal CO2 concentration in all patients because there is a risk of air embolism during the opening (see later discussion). The patient is offered the option of having a catheter inserted in the bladder; some men find it difficult to use a urinal in the supine position. Fluids are monitored and it is important to keep the patient well hydrated throughout the procedure, especially given hypovolemia and hypotension issues with these patients. The head and neck should be positioned with some degree of flexion at the lower cervical spine and extension at the atlanto-occipital junction. This allows the patient’s airway to remain patent and makes it possible for the anesthesiologist to instrument the airway should it become necessary (e.g., in case of airway obstruction, seizures with airway compromise, or neurological deterioration). PD patients can have moderate to severe kyphosis and, in the extreme, camptocormia,30 making positioning a challenge. Depending on the stereotactic frame, bars can protrude into the patient’s shoulders and need to be carefully padded; this issue needs to be considered during the initial frame application. Severe kyphoscoliosis in PD and dystonia patients can lead to spondylotic decrease in spinal canal diameter and increasing the risk for spinal cord compression, so positioning of the neck in the operating room (OR) must be performed with extreme care. Extra padding is utilized on our OR table because of the lengthiness of the procedure in an awake patient. We use a mechanical OR bed to avoid interference with the electrophysiological mapping, although given current shielding of the equipment this may not be necessary. In contrast to some, we use only sequential compression stockings in patients under general anesthesia. The skin opening, burr holes, and dural opening can all be done with local anesthesia alone. However, the anxiety and subjective experiences during the burr hole procedure prompt many centers to perform this stage with conscious sedation (low-dose propofol or remifentanil13). Short-acting agents are recommended to minimize carryover into the mapping phase. We tend to avoid propofol for conscious sedation in patients with PD because of its antiparkinsonian effect.18 Although propofol has been found to temporarily induce dystonia in healthy patients as discussed earlier, we do not avoid its use in patients with dystonia who require conscious sedation. Electrophysiological mapping and electrical stimulation are best performed with an alert and responsive patient; most centers therefore perform this stage of the procedure under local anesthesia with minimal sedation, letting short-acting agents, if used previously, wear off after the burr holes are made. However, surgery can be performed with conscious sedation with little impact on electrophysiological mapping results and electrical stimulation testing.8,22,31,32 An important question, however, is how deep can sedation be without interfering with the mapping and stimulation phase of the surgery. Until recently, there was insufficient clinical experience with these situations to even examine these questions. The goal of microelectrode mapping is to precisely identify the target structure and its borders. For the globus pallidus internus (GPi) and subthalamic nucleus (STN), the sensorimotor region is characterized. For the ventral intermediate (Vim) nucleus in tremor cases, the passive movement-activated cells in Vim and cells with cutaneous sensory receptive fields in the ventral caudal (Vc) thalamic nucleus must be identified. Thus, to accomplish adequate microelectrode mapping, identifiable units must be found, distinctions between adjacent nuclei must be made (e.g., internal and external segments of GP), and movement-related changes in firing rates should be observed. Recent reports demonstrate that these goals can be met, at least in part, with various degrees of anesthesia (Table 6.2). Anesthesia can be induced prior to the placement of the frame. However, if general anesthesia is to be induced with the stereotactic frame in place, then conventional rigid laryngoscopy will not be easy. Our technique of choice is asleep fiberoptic endotracheal intubation, using the apneic technique through an intubating airway or through an LMA utilizing the Aintree Intubation Catheter (Cook Critical Care, Bloomington, IN). Another option is using an LMA for the entire procedure; the Flexible LMA is our first choice. If one is concerned about gastric distention, especially with controlled ventilation, the ProSeal LMA (LMA North America, San Diego, CA) can be used. When the LMA is used, we prefer to have the patient breathe spontaneously or with assisted ventilation, although some do not hesitate to give the patient some muscle relaxants and control the patient’s ventilation. The effects of anesthetic agents are not homogeneous across different regions of the CNS. For example, brainstem and thalamic evoked potentials can be performed in a patient despite lack of conscious awareness during intravenous or inhalational anesthesia due to the selective effects of anesthetics on the frontal cortex and auditory cortex with preservation of brainstem and thalamic afferent pathways.33–37 There is a generalized suppression of background activity leaving reactive capabilities facilitated at deep levels, and amplitude of evoked potentials is actually augmented in a dose-related manner.33 In our experience, this dissociation between neocortical and lower cortical/brain stem effects of intravenous and inhalational agents renders the use of the Bispectral Index (BIS) (Aspect Medical Systems, Natick, MA) for monitoring anesthetic depth37 somewhat irrelevant for determining whether electrophysiological mapping can be performed. It is uncertain to what extent general anesthesia affects single-unit recording. In fact, single-unit recordings are routinely obtained in animals under general anesthesia.38,39 In patients, successful recordings have been made from Vim under propofol narcosis.31,32 However, there may be differences in characteristics of neuronal activity as a function of anesthetic depth. Compared with dystonia patients mapped under local anesthesia, the firing rates in GPi were substantially decreased, and long pauses were present in dystonia patients mapped with deep sedation or general IV anesthesia (propofol) (Fig. 6.1).10,22 Moreover, although limb driving was obtained under propofol, fewer arm-responsive units were observed.22 These findings are consistent with enhancement of gamma-aminobutyric acid (GABA)ergic striatal and globus pallidus externus (GPe) afferents to GPi by propofol.40 We examined GPi firing rates and patterns in the monkey under inhalational general anesthesia and similarly found decreased firing rates and increased pauses in GPi, which substantially blurred the distinction between the external and internal pallidal segment (GPe and GPi) in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated model of PD.39
Preoperative Evaluation
Parkinson Disease
Essential Tremor
Dystonia
Medication Interactions
Parkinson Disease
Essential Tremor
Dystonia
Peri- and Intraoperative Anesthetic Approaches
Choosing the Anesthetic Approach
Stereotactic Frame Placement
Magnetic Resonance Imaging and Computed Tomographic Scanning
Surgery under Local Anesthesia
Monitoring
Positioning and Draping
The Opening
Electrophysiological Mapping and Stimulation Mapping
Surgery under Deep Sedation or General Anesthesia
Intubation with the Stereotactic Frame in Place and Airway Control
Microelectrode Mapping under Deep Sedation or General Anesthesia

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree