Fig. 1
Scanning electron microscopy and Raman spectroscopy of an example of one formulation of nimodipine microparticles. Scanning electron microscopy (a, b) and Raman spectroscopy (c, d, red is form 1 of nimodipine, green is amorphous nimodipine, and blue is form 2 of nimodipine) at baseline. After attempted forced degradation at 30–35 °C for 30 days, the microparticles showed very slight polymer melt (scanning electron microscopy (e, f), and Raman spectroscopy (g, h), where red is form 1 of nimodipine, green is amorphous nimodipine, and blue is form 2 of nimodipine), which was avoided after optimization of the formulation
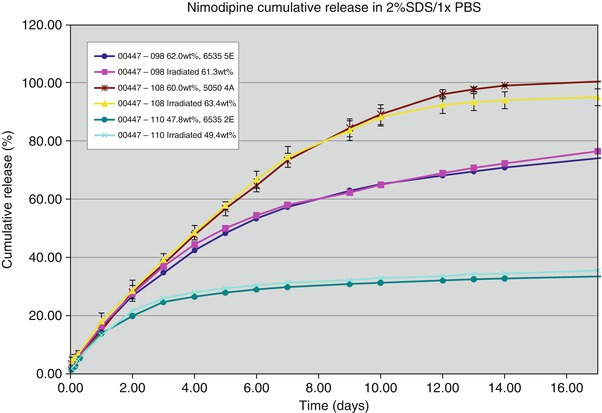
Fig. 2
Nimodipine cumulative release in vitro from several different formulations of microparticles showing the ability to synthesize formulations with different release characteristics and that there is no change in the release characteristics after gamma irradiation with 27 kilogray radiation
Preclinical Studies on Release Characteristics and Toxicity
Release characteristics of different formulations or neat nimodipine were tested different in vitro and in different models [32–34]. More recently, we tested this in Wistar rats at subcutaneous doses of 20 or 200 mg/kg and buffer or plasma nimodipine concentrations, which were measured daily. We found sustained nimodipine release for all doses and formulations, including the neat nimodipine, which was likely because of its high lipid solubility within subcutaneous fat (Fig. 3).
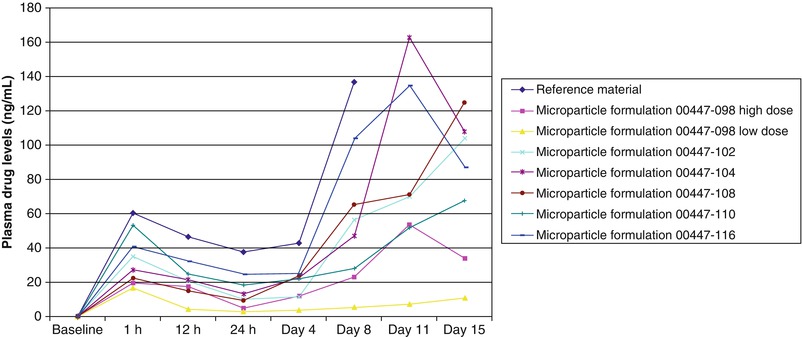

Fig. 3
Release characteristics in vivo in the subcutaneous space of rats of neat nimodipine (reference material) or six different formulations of microparticles containing nimodipine. The release characteristics can be varied to alter the initial burst release at 1 h as well as the time course of release over 15 days
Because nimodipine microparticles in hyaluronic acid have never been injected into the subarachnoid or intraventricular CSF, potential toxicity was investigated in rats and beagles in escalating doses up to threefold times the estimated human dosage (1,200 mg). All animals survived until their sacrifice and there were no EG-1962-related toxicity findings in clinical observations, neurobehavioral evaluations, body weight, food consumption, ophthalmoscopy, hematology, coagulation, clinical chemistry, urinalysis, organ weight, or macroscopic pathology.
Future Perspective
We are initiating two clinical trials. The PROMISE (Prolonged Release nimOdipine MIcro particles after Subarachnoid hemorrhage) trial will be designed as an unblinded, non-randomized, single-center, single-dose, dose-escalation safety and tolerability phase 1 study in patients neurosurgically treated for aSAH at the Department of Neurosurgery, Medical Faculty of the Heinrich-Heine University, Düsseldorf, Germany, and will solely investigate the effect of intracisternal EG-1962 administration. The NEWTON (Nimodipine microparticles to Enhance recovery While reducing TOxicity after subarachNoid hemorrhage) trial will be a phase 1/2a multicenter, controlled, randomized, open-label, dose escalation, safety, tolerability, and pharmacokinetic study comparing EG-1962 and nimodipine in patients with aneurysmal SAH treated by neurosurgical clipping or endovascular coiling. In this study, EG-1962 will be administered by intraventricular catheter that would be in place as part of standard of care in eligible patients.
Conclusion
Due to the promising preclinical data, there is a strong rationale to investigate the effects of EG-1962 in patients suffering from high-grade SAH. Therefore, we are initiated two clinical trials on intracisternal and intraventricular EG-1962 application.
Conflict of Interest Statement
N. Etminan and D. Hänggi are scientific advisors for Edge Therapeutics, Inc. R.L. Macdonald is chief scientific officer of Edge Therapeutics, Inc. Cara Davis and Kevin Burton are employess of Evonik.
References
1.
Lovelock CE, Rinkel GJ, Rothwell PM (2010) Time trends in outcome of subarachnoid hemorrhage: population-based study and systematic review. Neurology 74:1494–1501PubMedCrossRefPubMedCentral
2.
Dreier JP, Major S, Manning A, Woitzik J, Drenckhahn C, Steinbrink J et al (2009) Cortical spreading ischaemia is a novel process involved in ischaemic damage in patients with aneurysmal subarachnoid haemorrhage. Brain 132:1866–1881PubMedCrossRefPubMedCentral
3.
4.
5.
6.
Vergouwen MD, Vermeulen M, Coert BA, Stroes ES, Roos YB (2008) Microthrombosis after aneurysmal subarachnoid hemorrhage: an additional explanation for delayed cerebral ischemia. J Cereb Blood Flow Metab 28:1761–1770PubMedCrossRef
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





