Fig. 26.1
The structure of high molecular weight neurofilament. NF-H is the longest form of neurofilament, having 150 kd as its molecular weight. Near its C-terminus NF-H contains more than 50 repeats of the serine–lysine–proline (KSP) motif, which is phosphorylated by kinases such as CDK5, physiologically
26.2 Measurement of pNF-H in Rodent Models
To evaluate the sensitivity of blood pNF-H level in discriminating the severity of SCI, we used a mouse SCI model, in which we took daily blood samples from the tail vein. The blood pNF-H values were determined by an enzyme-linked immunosorbent assay (ELISA). The severity of SCI was determined by setting damage force in a spinal cord contusion device. The trend of average pNF-H values showed a single peak pattern regardless of the severity. The values marked their peak value at the third day after injury, returning to the pre-injured level (below minimal detection value) after 2–3 weeks. Although the trends are similar, their peak values differ depending on the severity. The mice who received the most severe injury (90 kdyn) had the highest pNF-H value at 3 days after injury, while mice with less severe SCI show lower pNF-H values. Therefore, blood pNF-H seems to correlate with the degree of trauma in the spinal cord (Fig. 26.2).
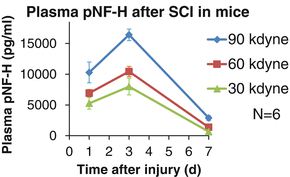

Fig. 26.2
The time course of blood pNF-H levels in rodent SCI model. Mice received various degrees of SCI (impact force: 30, 60, or 90 kdyn). Serial peripheral blood samples were collected to determine pNF-H levels. Regardless of the severity of SCI, the peak pNF-H values were observed at 3 days after injury and higher peak pNF-H values correspond to more severe damage to spinal cord
26.3 Measurement of pNF-H in Humans
To confirm the usability of pNF-H in clinical practice, we collected blood samples from acute SCI patients. In this study, we set inclusion criteria as patients having cervical SCI and not having severe traumatic brain injury. In some cases, blood samples were repeatedly collected until 3 weeks after injury. We recorded clinical symptoms on admission and also at the final examination (average duration of 6 months).
First of all, we confirmed that pNF-H is elevated in SCI patients’ blood samples. In most patients who showed complete SCI, pNF-H was initially detected as early as 18 h after injury. Serial samples indicated that the pNF-H level increases gradually for several days after injury (Fig. 26.3). In contrast to the rodent model, in which pNF-H marks its peak value at 3 days after injury, human SCI samples showed sustained elevation of pNF-H even after 3 days. When we plotted the pNF-H values according to the severity of paralysis at end point, we observed higher pNF-H values in complete SCI (AIS A) group compared to incomplete SCI (AIS C and D) group with a statistically significant difference. Thus, the difference was observed at both 24 and 96 h after injury (Fig. 26.4).
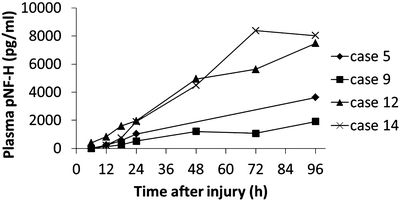
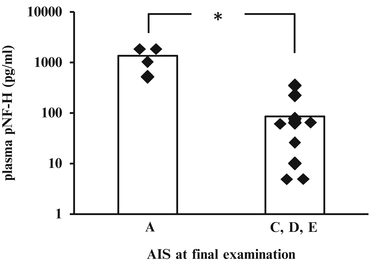

Fig. 26.3
Serial pNF-H measurements in complete SCI patients. The plasma pNF-H levels of four patients who were graded as AIS A at the final follow-up are shown. In those patients, pNF-H was detectable in plasma from 18 h after injury (221–825 pg/ml), and it increased until 96 h after injury (1,919–8,023 pg/ml)

Fig. 26.4
Blood pNF-H level was elevated in accordance with the severity of the injury. There was a marked difference between AIS A patients (complete SCI) and AIS C–E patients (incomplete SCI), with statistical significance at 24 h after injury
In some cases, we continued to measure pNF-H for up to 3 weeks. All four cases (two cases with AIS A and two cases with AIS C) showed sustained elevation of pNF-H even at 3 weeks after injury. At the third week, the difference in pNF-H value between AIS A and C become unclear (data not shown).
26.4 Discussion
We evaluated the usefulness of pNF-H as a biomarker for SCI in both animal models and clinical cases and found that blood pNF-H level in the early phase of SCI is correlated with its overall severity [12, 13].
26.4.1 Comparison with Other Biomarkers
The characteristics of pNF-H compared to preexisting biomarkers for CNS disorders are (1) it is measureable in blood samples and (2) it remains elevated for several days or even more. The first point enables easy access to samples compared to cerebrospinal fluid, in which most of the biomarkers, including pNF-H, exist at higher concentration than in peripheral blood [14]. Repeated measurement would be useful to achieve more information about the state of injured spinal cord. As for the second point, pNF-H in complete SCI patients remains elevated for more than 1 month. Other biomarkers, such as S100 and NSE, are elevated within blood but their levels return to baseline rapidly (within 24–48 h) [8]. The short duration of the detection time-window makes it difficult to obtain proper samples to evaluate the severity of patients. The gradual increase of pNF-H (detectable at 18 h after injury) may have some disadvantages. Since most of the therapeutic interventions are intended to start as early as possible, the ideal biomarker should be detectable immediately after injury. It would be beneficial to use pNF-H in combination with other biomarkers which become detectable at an early time point, such as S100B or UCHL1 [15]. Table 26.1 shows a list of possible biomarkers in the CNS.
Table 26.1
The list of biomarkers for central nervous system disorders
Name | Cell types | Physiological function | Reference |
|---|---|---|---|
pNF-H | Neurons (axon) | Cytoskeletal molecule | [13] |
Tau | Neurons | Microtubule associated protein | [20] |
UCHL1 | Neurons (cell body) | Deubiquitinating enzyme | [15] |
NSE | Neurons | Glycolytic enzyme | [21] |
S100B | Astrocytes | Calcium-binding protein | [8] |
GFAP | Astrocytes | Intermediate filament | [22] |
MBP | Oligodendrocytes | A structural protein of myelin | [23] |
26.4.2 Possible Applications of pNF-H
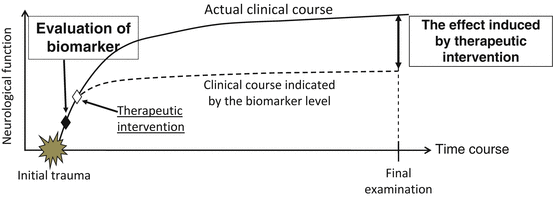
One of the possible applications of pNF-H measurement is the prognostic evaluation of acute SCI. A precise prognostic evaluation is helpful to organize rehabilitation programs for each patient. Furthermore, such evaluation may contribute to designing clinical trials of novel therapies for SCI. Several approaches to improve the outcomes of SCI by intervention at an early time point, such as within 1 week after injury, are in the preclinical stage. At the moment, we cannot distinguish patients with “definite complete paralysis,” who will remain completely paralyzed after several months, and those with “temporary complete paralysis,” who manifest complete paralysis on admission and gradually recover to incomplete paralysis over 1–3 months. If we could set up a definite cutoff line of pNF-H values at 24 h after injury to distinguish these two subgroups at recruitment, we can exclude “temporary paralysis” from clinical trials to improve the specificity of the outcome (schematically shown in Fig. 26.5). To set up such a cutoff value, further clinical studies are required to associate early pNF-H levels with neurological findings in the chronic stage [13].