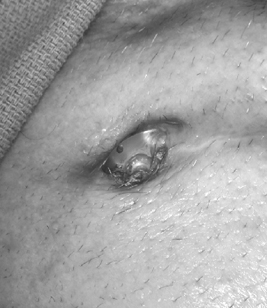
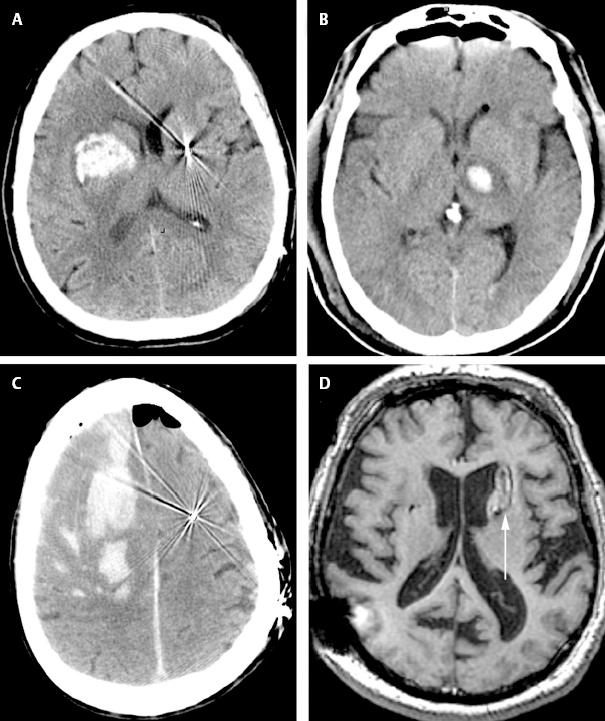
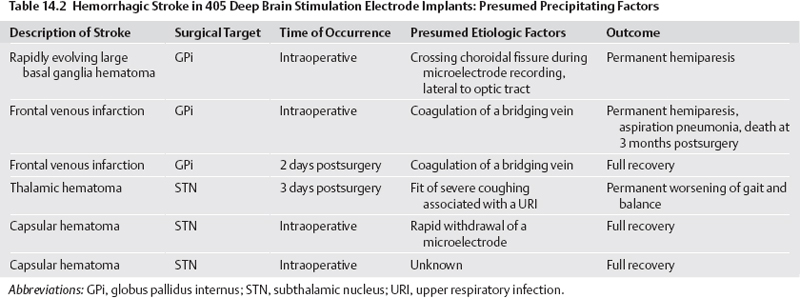
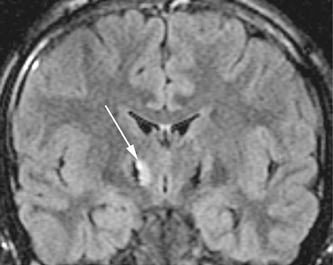
14 Avoiding Complications and Correcting Errors Philip A. Starr Several recent publications have reviewed complications of deep brain stimulation (DBS)1–8 and lesioning surgery4,9–12 for movement disorders. A summary of our group’s perioperative and device-related complications in 405 DBS implants for movement disorders is provided in Table 14.1. Procedures were performed with frame-based stereotaxy using magnetic resonance imaging (MRI) and microelectrode recording (MER). Medtronic Activa (Medtronic, Inc., Minneapolis, MN) DBS hardware was used in all cases. The total incidence of unexpected returns to the operating room for management of a complication was 38 cases, or 9.6% of implanted leads. This chapter describes our current methods for complication avoidance and management. These methods continue to evolve. Although this discussion is oriented primarily toward DBS, many of the principles apply to other stereotactic procedures. Complications uniquely associated with stereotactic lesioning procedures are discussed briefly at the end of the chapter. Stroke is the most serious potential complication of movement disorders surgery. Stroke is defined as a new neurological deficit of vascular origin, lasting longer than 24 hours. Using this definition, our DBS series includes seven strokes, for an incidence of 1.7% per lead and 3.0% per patient. Six of these were hemorrhagic strokes. Fig. 14.1 shows postoperative brain imaging for representative hematomas. Table 14.2 provides details of presumed etiologic factors. Ischemic infarction is very infrequent following DBS surgery, occurring only once in our series (Fig. 14.2). In contrast, delayed ischemic capsular infarction has been well described following pallidotomy10,13 and is probably a more frequent complication of stereotactic lesioning surgery than of DBS. Asymptomatic hemorrhage is more common than symptomatic hemorrhage but is only detected if postoperative imaging is performed systematically. Our rate of asymptomatic hemorrhage, detected on routine postoperative magnetic resonance imaging (MRI) was 2.2% per lead. A typical asymptomatic hematoma is shown in Fig. 14.1D. Most of the asymptomatic hematomas occurred subcortically, 25 to 35 mm superior to the target, corresponding to the location where the guide tube for the microelectrode terminated. Based on our experience, we recommend the following measures to reduce the incidence of stroke: Should a new neurological deficit occur during the procedure, surgery is immediately stopped. The anesthesiologists are asked to redouble efforts at blood pressure control and ensure appropriate oxygenation. All instruments are removed from the brain and the cortex is inspected. Superfcial bleeding sources are coagulated and surgery continues. If no surface bleeding is seen and no bleeding is seen from the cortical entry, the scalp is rapidly closed, the headframe is removed, and the patient is taken for a computed tomographic (CT) scan. Short of a neurological deficit, there may be other more subtle signs of intraparenchymal hemorrhage that is initially asymptomatic and may remain so depending on its final size and location. When we have observed blood coming from the subcortical guide tube after withdrawal of a stylet or microelectrode, a small hematoma is almost always observed on postoperative imaging at a depth corresponding to the termination of the guide tube. During MER,if a significant region of electrical silence is observed at a depth where neuronal tissue is predicted based on prior adjacent MER tracks, the cause may be a small hematoma. If either of these signs of potential hematoma formation is observed, we halt the procedure and closely observe the patient for subtle new neurological deficit while recon-firming strict blood pressure control. If no deficit occurs in 5 to 10 minutes we proceed with surgery. Fig. 14.1 Postoperative axial computed tomographic (CT) scans (A–C) or magnetic resonance imaging (MRI) (D) of hemorrhages complicating stereotactic deep brain stimulation (DBS) surgery in our series. (A) Hematoma that occurred during penetration of a mi-croelectrode into the choroidal fissure, lateral to the optic tract, during microelectrode mapping of globus pallidus internus (GPi). (B) Hematoma that occurred during rapid withdrawal (> 1 mm/s) from the subthalamic nucleus (STN). (C) Hemorrhagic venous infarct that occurred following inadvertent interruption and coagulation of a bridging dural vein. (D) An asymptomatic hematoma in the left caudate nucleus (white arrow), immediately anterior to the DBS lead. (The hyperintensity in the right parietal cortex is artifact from the overlying connector of the lead extender). Fig. 14.2 Coronal magnetic resonance imaging (MRI) (FLAIR sequence) showing a delayed ischemic capsular infarction that occurred 1 week after bilateral subthalamic nucleus deep brain stimulation (DBS). The infarcted area (white arrow) is adjacent to the right DBS lead. In our series, the incidence of serious infection, defined as infection requiring a return to the operating room for removal of all or part of the DBS hardware, was eight cases, or 2.0% per lead and 3.5% per patient (Table 14.1). All of these infections have occurred subcutaneously, starting at the lead extender or the implantable pulse generator (IPG). The major offending organisms were Staphylococcus (staph) aureus and Staph epidermidis. We have had no infections in the brain, and in fact cerebral abscess or cerebritis complicating DBS has not been reported in most recent series. Most of our infections presented with some combination of swelling, redness, pain, or drainage over the connector of the lead extender, or over the IPG. Most presented within 1 to 8 weeks of surgery, although one presented as a stitch abscess over an anchoring suture on the connector 2 years after implantation. Our approach to avoidance of infection, in addition to the obvious meticulous attention to sterile technique, is as follows: The management of hardware infections has not been standardized. In the report of Oh et al,6 infections of any part of the device were ultimately treated with removal of all hardware, despite initial attempts at more localized treatment. Our approach is as follows: Four of our patients have presented within 1 month post-surgery with tense swellings around the IPG, which were fluctuant but painless and without redness or warmth. Surgical exploration of one of these revealed a sterile clear fluid collection consistent with cerebrospinal fluid (CSF). This appears to occur more frequently with burr hole– based anchoring methods that are not watertight (such as the Stimloc system from Medtronic, Inc., www.medtronic.com/physician/activa) because CSF can track down the hardware to accumulate in the pectoral cavity. Sealing the burr hole with Gelfoam and fibrin glue prior to closure has reduced the incidence of these sterile collections. If a swelling around an IPG is not red, tender, or warm, and the incision is healed, our practice is to observe it. Sterile fluid collections typically resolve spontaneously.
Operative Complications
Stroke
Avoidance of Stroke
Management of Stroke
Infection
Infection Avoidance
Infection Management
Sterile Fluid Collections
Neupsy Key
Fastest Neupsy Insight Engine