Fig. 22.1
Appearance of honeycomb collagen sponge (HC) and in vitro evaluation of dorsal root ganglions (DRGs) on HC. HC consists of serial tunnel structures resembling a honeycomb (A). HC with DRGs explants, indicated by red circles, in agarose gel (B). Neurofilament (NF)-positive fibers from DRG explants are visible in HC pores of different sizes (C, a–d). The length of neurites sprouting into HC400 was significantly longer than that of neurites sprouting into HC130 (*P < 0.05) and RPC (**P < 0.01). Scale bar: 300 μm (C) (Image adapted from Fukushima et al. [13])
22.2.2 Nerve Regeneration After Implantation of a Honeycomb Collagen Sponge into the Transected Spinal Cord
The exposed T8-9 spinal cord of 9-week-old female Sprague Dawley rats was completely transected for 2 mm with an ophthalmic blade. A 2-mm-long scaffold of HC400 or RPC was then implanted between the rostral and caudal spinal cord stumps. Four weeks after implantation, animals were perfused transcardially with 4 % paraformaldehyde in 0.1 M PB, and a 14-mm-long spinal cord section containing the implant was dissected under a microscope. Sagittal sections (30 μm) through the scaffolds were cut on a cryostat and immunostained with NF 200 antibody. In RPCs, NF-positive fibers had penetrated the implanted site, but few fibers had crossed the rostral border (Fig. 22.2a). In contrast, NF-positive fibers were observed beyond the borders between the HCs and the host spinal cords 4 weeks after implantation (Fig. 22.2b). At higher magnification, many NF-positive fibers were observed parallel to the longitudinal midline of the implants (Fig. 22.2d), while several fibers were seen crossing the border between the implanted area and the stumps (Fig. 22.2c, e). These results suggest that the serial tunnel structure of the HCs have a guidance ability which promotes the regeneration of axons across the implanted area.
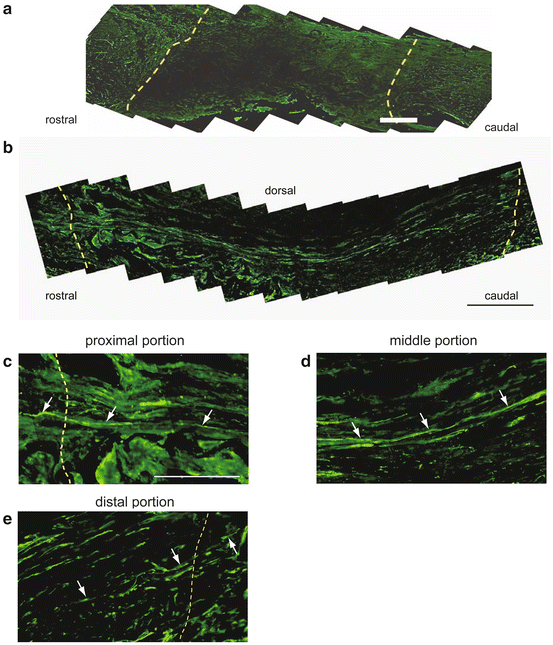

Fig. 22.2
Neurofilament staining of spinal cord-implanted HC. Yellow dotted lines indicate the border between the implant and spinal cord (a–c, e). Few NF-positive fibers are visible within the RPC-implanted site (a). Many NF-positive fibers are linear and bridge the length of the spinal cord-implanted HC 400 (b). In a higher magnification view of the rostral border between the HC and the spinal cord, NF-positive fibers (arrows) enter the implanted area across the rostral border between the spinal cord and the HC (c). In the middle portion, the NF-positive fibers (arrows) run in the sagittal direction (d). Some NF-positive fibers (arrows) cross the caudal border between the HC and spinal cord (e). Scale bar: 250 μm (a, b); 100 μm (c–e) (Image adapted from Fukushima et al. [13])
Several rats were examined by transcranial electrostimulation 10 weeks after HC implantation. Muscle-evoked potentials were observed in the right quadrant muscle with HC implantation. In addition, the spinal cord was re-transected at the distal stump. Both MEPs and contraction of the quadriceps disappeared after the transection. These results indicate that some descending pathways across the HC-implanted area had reconnected.
22.2.3 Neurite Extension Through Honeycomb Collagen Sponges with Bone Marrow Stromal Cells In Vitro and Application to the Hemisected Spinal Cord
BMSCs were prepared from adult Fischer 344 rats as previously described [15]. After one passage, BMSCs were transduced with lentiviral vectors expressing green fluorescent protein. HCs coated with 1 % fibronectin were prepared and mixed with dissociated BMSCs at a concentration of 2 × 105 cells per scaffold. HCs with or without BMSCs were stabilized in the agar filled with DRG medium. Tuj-1-positive neurites and GFP-positive BMSCs were observed in the tubular HC sections after 10 days in vitro (Fig. 22.3a). Quantitatively, neurite length was increased over twofold in the HC combined with BMSCs compared with the control HC, indicating that BMSCs directly support the elongation of neurites into HC in vitro (unpublished data).
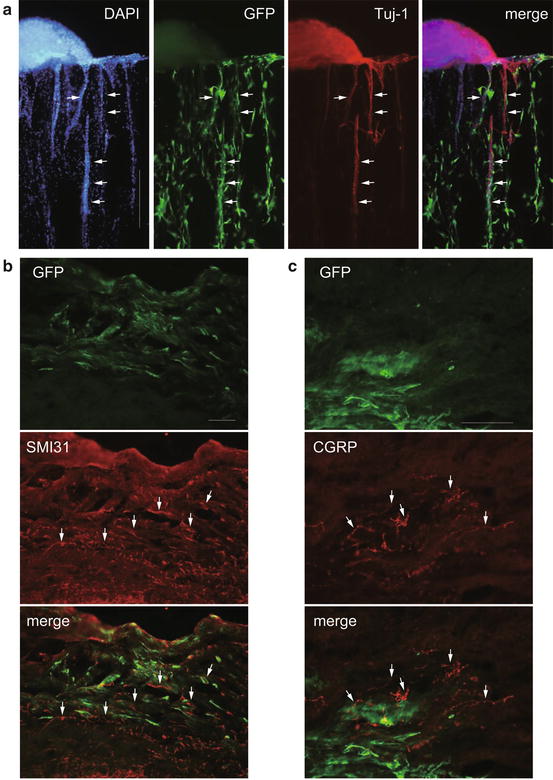

Fig. 22.3
Neurites extending through HC with bone marrow stromal cells in vitro and application to the hemisected spinal cord. The DRG explant is attached to the HC and extends Tuj-1-positive neurites toward and into the porous structure (a, arrows). Green fluorescent protein (GFP)-labeled BMSCs were distributed in the HC pores (a, green). GFP-positive cells (b, c, green) are visible at the implantation site. The regenerated nerve fibers are shown as SMI31-positive fibers (b, arrows). CGRP-positive fibers (c, arrows) close to the GFP-positive cells. Scale bar: 500 μm (a); 200 μm (b); 100 μm (c)
For in vivo study, the exposed spinal cord of adult female Fischer 344 rats was hemisected at T9 and a 3-mm hemicord section on the right side was resected. HC or HC combined with BMSCs was implanted between the rostral and caudal spinal cord stumps. Four weeks after implantation, animals were perfused, and a 9-mm-long spinal cord section containing the implant was dissected under a microscope. Horizontal sections of the scaffolds were cut on a cryostat and immunostained with mouse monoclonal anti-SMI31 (a marker for neurofilament M+H) and either rabbit polyclonal anti-GFP antibody or calcitonin gene-related peptide (CGRP) antibody. These antibodies were visualized with appropriate fluorescent secondary antibodies.
Four weeks after implantation, GFP-positive cells had survived in the implanted area of the HC combined with BMSCs, and SMI31-positive fibers were observed at the site (Fig. 22.3b). Quantitative analysis showed that more SMI31-positive fibers were observed in the HC combined with BMSCs than in the HC only (unpublished data). To analyze the growth of sensory fibers, we observed CGRP-positive fibers in the implanted spinal cord. These fibers were observed close to the GFP-positive area (Fig. 22.3c) as regenerated fibers possibly guided by the transplanted BMSCs.
22.3 Poly (d,l-Lactic Acid) Macroporous Scaffold
22.3.1 Implantation of a Poly (d,l-Lactic Acid) Scaffold Combined with Schwann Cells Secreting D15A, a Bifunctional Neurotrophin into the Transected Spinal Cord
The generation and characterization of SC cultures from adult Fischer 344 rat sciatic nerves were described previously [16]. SCs secreting D15A, which has been found to bind and activate TrkB, TrkC, and p75NTR [17], were generated. Freeze-dried poly (d,l-lactic acid) (PLA) macroporous guidance scaffolds have been developed and applied to the transected spinal cord [18]. The scaffolds have longitudinally oriented macropores connected by micropores (Fig. 22.4A), and the diameter of the macropores varies between 75 and 200 μm. Hurtado et al. filled this scaffold with genetically modified rat SCs and analyzed cell survival, scaffold integration, and the axonal regeneration response following implantation into the completely transected thoracic spinal cord of adult rats. The scaffolds were filled with 1 % fibrinogen in DMEM containing 2 % CaCl2, 0.2 % gentamicin, and 2 % aprotinin. A fibrin mixture at a concentration of 1 × 106 cells per scaffold was placed on top of the scaffold, which was drawn gently inside by placing it onto Whatman filter paper. Cells were generally uniformly distributed within the pores of the scaffold, although some small clusters of cells could also be discerned (Fig. 22.4A).
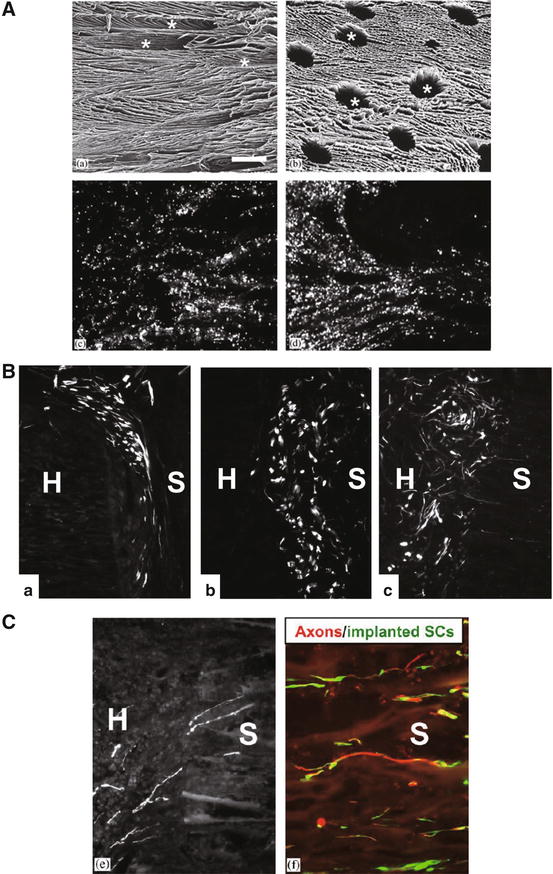

Fig. 22.4
Schwann cell-seeded d,l-polylactic acid scaffolds in the transected spinal cord. Scanning electron micrographs showing a longitudinal section (A, upper left) and cross section Fig. 22.4 (continued) (A, upper right) of the scaffold. Horizontal cryostat sections counterstained with DAPI reveal GFP-positive Schwann cells (SCs) in the scaffold at 1 h (A, lower left) and 6 h (A, lower right) after seeding. GFP-positive SCs in the zone between the scaffold (S) and the host spinal cord (H) at 1 week (B, a), 2 weeks (B, b), and 6 weeks (B, c) after implantation. The SCs are mostly elongated and express fluorescent GFP throughout the soma. Axons rapidly extended to the scaffold seeded with D15A-GFP-SCs (C). Some axons had grown into the scaffold with either type of SC at 2 weeks (C, left). RT97-positive axons were associated with GFP-expressing SCs within the scaffold at 6 weeks (C, right) (Image adapted from Hurtado et al. [11])
For in vivo study, the exposed T9-10 spinal cord of adult female Fischer 344 rats was transected and 3 mm of tissue which included visible spinal roots was resected. A 4-mm-long scaffold with GFP-SCs or D15A-GFP-SCs, or the 1 % fibrin solution alone as a negative control, was implanted between the rostral and caudal spinal cord stumps. At 1, 2, and 6 weeks after implantation, animals were perfused and a 10-mm-long spinal cord section containing the implant was dissected under a microscope. Horizontal sections of the scaffolds were cut on a cryostat and immunostained with mouse monoclonal antibody against p75, a low-affinity nerve growth factor receptor (192IgG, a marker for SCs), or anti-neurofilament antibody RT97 (a marker for neurofilament H). These antibodies were visualized with appropriate fluorescent secondary antibodies.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








