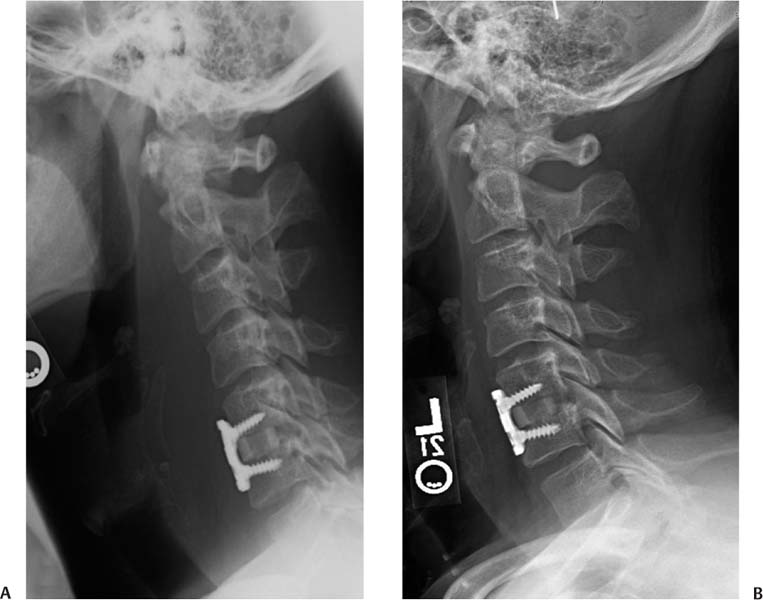
23 In July 2002, the U.S. Food and Drug Administration (FDA) approved the use of recombinant human bone morphogenetic protein-2 (rhBMP-2) on an absorbable collagen sponge (ACS; INFUSE®Bone Graft, Medtronic Sofamor Danek, Memphis, TN) contained in a tapered titanium cage (LT-CAGE Lumbar Tapered Fusion Device) for single-level anterior interbody fusion in skeletally mature patients with degenerative disk disease.1 The FDA also granted rhBMP-7 (OP-1 Putty, Stryker Biotech, Hopkinton, MA) humanitarian device exemption approval in 2004 as an autogenous bone graft alternative for revision posterolateral lumbar fusion in compromised patients, in whom autogenous bone or bone marrow harvest are not feasible or not likely to promote fusion.2 The use of BMPs in the lumbar spine resulted in a reduction of operative time and blood loss, eliminated the risk of significant donor-site morbidity that is associated with iliac crest bone graft (ICBG) use, and achieved fusion rates equivalent or superior to autograft. At that time, without any reports of significant complications associated with this practice, many surgeons began exploring the “off-label” use of these osteoinductive proteins in other areas of the spine. With the “off-label” use of these proteins in the cervical spine, there have been several reports of significant complication rates.3–9 These primarily involved the use of high doses of rhBMP-2 in the anterior cervical spine resulting in substantial postoperative swelling and airway compromise (Fig. 23.1). These reports prompted the FDA to issue a public health notification in 2008 that warned of the life-threatening complications associated with rhBMP use in the cervical spine.10 However, there have also been other reports exploring the efficacious use of rhBMP in the cervical spine without a significant complication rate.11–13 Although several studies have confirmed the efficacy, appropriate dosage, and safety of rhBMP in the lumbar spine,14–19 the data regarding rhBMP use in the cervical spine is not as extensive. The limited number and inconsistent outcomes of the studies available have led the way to several key issues remaining controversial, including the following: (1) How effective is the use of rhBMP in the cervical spine and (2) what are the associated risks of rhBMP use in the cervical spine? We performed a review of the literature to determine the best available evidence that addresses these issues. The search included Medline, Scopus, and the Cochrane Collaboration Library for literature published through May 2009. The reference lists of key articles were also reviewed for any additional studies. A search for the term “bone morphogenetic protein” returned 8028 articles. A search for “cervical spine” returned 11,714 articles. When these terms were combined, the search returned 40 articles. Review articles, editorials, case reports, non-English written studies, reports available only as abstracts, and animal studies were excluded. The remaining articles were evaluated based on their abstract or full text for inclusion in this analysis. One relevant level I,20 three level II,3,4,11 and eight level III5–9,12,13,21 studies were identified. The level of evidence of the studies is summarized in Table 23.1. Only one level I study directly compares the efficacy of rh-BMP-2 use in the cervical spine with iliac crest bone graft (ICBG). Baskin et al20 performed a prospective, randomized, controlled study evaluating the efficacy of rhBMP-2 (0.6 mg per level) with ACS versus ICBG, placed inside of fibular allograft with concomitant anterior cervical plate augmentation in anterior cervical diskectomy and fusion (ACDF). The study included 33 patients, 18 in the rhBMP-2 group and 15 in the autograft group. All patients had one-or two-level cervical disk disease resulting in radiculopathy, myelopathy, or both, and had failed to see improvement after a minimum of 6 weeks of conservative therapy. At 6-, 12-, and 24-month follow-up, there was a 100% radiographic fusion rate in both groups. Although both groups achieved a significant improvement in neck disability index scores, the rhBMP-2 group had mean improvement superior to the control group at 24 months (p < 0.03). Also at 24 months, both groups had an overall neurological and neck pain success rate of 100%, patient satisfaction of 90%, and similar mean improvement on the physical and mental components of the Short Form-36 (SF-36). None of the patients developed antibodies to rhBMP-2. Two patients in the rhBMP-2 group and one in the autograft group developed bone formation immediately anterior to segments adjacent to the treated level. However, because this heterotopic bone growth developed in both groups and all three surgeries were performed by the same surgeon, the authors suggest that it is likely secondary to technique and not the use of rhBMP-2. A criticism of this study is the small sample size. Nevertheless, it was a pilot study and opened the door to subsequent larger trials studying the efficacy and safety of rhBMP-2 in the cervical spine. Fig. 23.1 Lateral radiograph of a patient presenting with complaints of dysphagia 4 days after a single-level instrumented anterior cervical diskectomy and fusion (ACDF) utilizing 2.1 mg of rhBMP-2 with allograft. Notice the significant prevertebral swelling. (B) At 2 weeks postoperatively, the swelling significantly decreased and the dysphagia resolved without any further intervention. Table 23.1 Evidence Levels of Published Studies
Bone Morphogenetic Protein in the Cervical Spine: Efficacy and Associated Risks
 Efficacy of Bone Morphogenetic Protein Use in the Cervical Spine
Efficacy of Bone Morphogenetic Protein Use in the Cervical Spine
rhBMP-2
Level I Data
Level | Number of Studies | Study Type |
|---|---|---|
I | 1 | Prospective, randomized controlled trial20 |
II | 3 | |
III | 8 |
Level II Data
Buttermann reported on a prospective, double-cohort study comparing the use of rhBMP-2 versus ICBG with fibular allograft in one-to three-level ACDF for degenerative conditions.3 The study included a total of 66 consecutive patients with a minimum 2-year follow-up who were counseled regarding the off-label use of rhBMP-2 and other alternatives to ICBG. The patients were then allowed to choose the type of graft to be used in their surgery. Thirty-six patients chose to undergo ACDF with the use of ICBG, whereas 30 patients opted for the use of rhBMP-2 with allograft (0.9 mg rhBMP-2 per level). All of the patients in the rhBMP-2 group underwent anterior cervical plate instrumentation, whereas 10 of the patients who underwent single-level ACDF in the ICBG group did not have instrumentation. Both groups showed significant improvement in all outcome measures throughout the follow-up period when compared with their preoperative status (p < 0.01). The only difference between the groups during the follow-up period was that patients in the rhBMP-2 group showed a greater improvement in disability 7 to 12 months postoperatively, but this difference between the two groups was no longer evident at 2- to 3-year follow-up. Two patients in the ICBG group and one in the rhBMP-2 group developed a pseudarthrosis; however, this difference was not statistically significant. The authors concluded that ACDF performed with rhBMP-2 is as effective as ICBG in terms of clinical outcomes and fusion rates. A weakness of this study was that it was not randomized. Patients were allowed to choose the bone graft agent in their procedure, rendering the groups not strictly comparable. There were also a relatively small number of patients in each group, resulting in an inability to detect any true differences between them.
Lanman and Hopkins performed a prospective evaluation of their early findings in a pilot study of ACDF with the use of rhBMP-2 (dosage not specified) and poly (L-lactide-co-D,Llactide) bioabsorbable implants in 20 patients.11 Patients had a one- to three-level ACDF with plate augmentation. Evidence of fusion was noted starting at 3 months postoperatively on computed tomography (CT) and plain radiographs in all patients and was consistent with findings at 6 months. This was, however, a small study with a relatively short-term follow-up without any control group or future report that illustrated the long-term results in these patients.
Vaidya et al4 prospectively examined the outcomes of 11 patients (18 levels) undergoing ACDF with an anterior locking plate using allograft and rhBMP-2 (1 mg per level), and compared those with the outcomes of 12 patients (22 levels) undergoing the same procedure with allograft and demineralized bone matrix (DBM). At a mean of 6 months, all levels in the rhBMP-2 group were solidly fused. The DBM group achieved a similar fusion rate (95.4% of levels); however, this took a substantially longer time to develop (mean of 19 months). Thirty percent of patients in the rhBMP-2 group reported increased pain between 2 and 6 weeks postoperatively, but this pain resolved between 6 weeks and 3 months. Both groups showed a significant improvement in visual analogue scale (VAS) and Oswestry Disability Index (ODI) scores, but there was no significant difference between the two groups. Weaknesses of this study include its lack of randomization and small sample size.
Level III Data
In a retrospective study by Vaidya et al,5 the authors compared the clinical and radiographic outcomes in patients undergoing ACDF and anterior cervical plate instrumentation with the use of rhBMP-2 (1 mg per level) and a polyetheretherketone (PEEK) cage (22 patients, 38 levels) versus allograft spacers and DBM (24 patients, 40 levels). All 46 cases were primary fusions for either radiculopathy or myelopathy. Although there were no statistically significant differences in neck or arm pain VAS or ODI scores between the groups at any time point, both groups showed significant improvements from their preoperative scores. All patients in the rhBMP-2 group achieved radiographic evidence of fusion at their latest follow-up, whereas one patient in the allograft group developed a nonunion. In a subsequent report by the same team evaluating the outcomes of 23 patients who underwent ACDF with the use of rhBMP-2 (~1 mg per level) and a PEEK cage, all subjects had radio-graphic evidence of solid fusion by 9 months postoperatively and showed significant improvement in neck and arm pain and cervical ODI scores.6 Both studies are weakened by their retrospective nature and small patient population. The latter study also lacked a concurrent control group.
In a review of 200 patients, Tumialán et al12 evaluated the efficacy of instrumented ACDF surgeries with PEEK spacers filled with three different dosages of rhBMP-2. This study included 24 patients that were also reported by Boakye et al,7 which served as the initial group receiving the highest dose of rhBMP-2 (2.1 mg per level). After noticing hetero-topic bone formation in three of the patients, the authors decreased the dose of rhBMP-2 to 1.05 mg per level in the next 93 patients. Subsequently, the authors chose to further decrease the dosage to 0.7 mg per level for the final 83 patients. All patients achieved solid fusion confirmed by dynamic radiographs and CT scans. Based on Odom criteria, 165 (85%) of patients’ outcomes were rated as good to excellent, 24 (12.4%) were fair, and four (2%) were poor. Among patients with myelopathy, Nurick grades improved from a preoperative mean of 1.42 to a postoperative mean of 0.26. The authors concluded that the use of the combination of these synthetic materials led to an excellent fusion rate without any significant morbidity. Although this study included a large number of patients, it was weakened by its retrospective design and lack of a concurrent control group.
rhBMP-7
Level I, II, and III Data
There are no level I, II, or III data published regarding this topic.
Summary of Data for rhBMP-2 and rhBMP-7
There are limited level I and II data directly comparing the efficacy of BMP in the cervical spine versus autogenous bone graft, the current gold standard. The best evidence available suggests that the efficacy of rhBMP-2 in the cervical spine is equivalent to autogenous bone or DBM. In a majority of reports, the rhBMP-2 groups achieved a 100% fusion rate and showed significant improvement in all clinical outcomes. In the studies with an ICBG control group, there were no significant differences in fusion rates or clinical outcomes between subjects treated with rhBMP-2 and those with ICBG. Based on the grading scale of Wright et al,22 the recommendation of using rhBMP-2 in the cervical spine would be a grade B: there is fair evidence for recommending the intervention. The data are summarized in Table 23.2.
There are currently no level I, II, or III data available regarding the efficacy of rhBMP-7 use in the cervical spine. Thus the recommendation of using rhBMP-7 in the cervical spine would be a grade I: there is insufficient evidence to make a recommendation.22
Pearls
• Level I and II evidence suggests that the efficacy of rhBMP-2 in the cervical spine is equivalent to autograft or demineralized bone matrix.
• There are no level I, II, or III data evaluating the efficacy of rhBMP-7 in the cervical spine. There is insufficient data available to make a recommendation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree