14 Brain Death and Organ Donation Jennifer A. Frontera Brain death, which is equivalent to legal death, is defined as the irreversible loss of brain and brainstem function. There is no universal policy governing the declaration of brain death, and protocols vary from hospital to hospital, although many states have issued guidelines. The declaration of pediatric brain death follows different parameters, and adult brain death will be the focus of this chapter. In the United States, top etiologies for brain death include subarachnoid hemorrhage, traumatic brain injury, intracerebral hemorrhage, and hypoxic ischemic injury. According to the United Network for Organ Sharing (UNOS), there were over 98,000 patients awaiting transplant in 2007, yet only 14,000 donors. In 2005, 18 patients died each day awaiting transplant.1 Although historically most organ donations have come after neurologic death, donation after cardiac death (DCD) and tissue donation (cornea, skin, bone, and musculoskeletal tissue) are also possible. Patients with terminal injuries and intent to withdraw life support are potential donors if cardiac death is expected within 60 minutes of withdrawal of support (DCD). For DCD, withdrawal of life-sustaining measures is performed in the operating room. Typically, morphine and benzodiazepines are administered for comfort after withdrawal of support. Final decisions over suitability of a donor should be arbitrated by the regional organ donor center, not the physician (Table 14.1). Because the progression from brain death to somatic death results in the loss of 10 to 20% of potential donors, intensive monitoring and care are needed to preserve organs for donation.2
| Contraindication | Notes |
| Multisystem organ failure due to sepsis | |
| History of cancer | EXCEPT: Skin cancer other than melanoma, certain primary brain tumors, remote prostate cancer |
| Viral infections | HIV, HTLV-1 and 2, rabies, reactive HbsAg, measles, West Nile virus, SARS, adenovirus, enterovirus, parvovirus, active HSV, VZV, EBV, viral encephalitis/meningitis Hepatitis B or C organs can be transplanted into recipients with the same virus CMV+ organs can be transplanted-better success if recipient is prophylaxed |
| Bacterial infections | Tuberculosis, gangrenous bowel, bowel perforation, intra-abdominal sepsis |
| Fungal infections | Active Cryptococcus, Aspergillus, Histoplasma, Coccidioides, Candidemia, invasive yeast infections |
| Parasitic infections | Leishmania, Trypanosoma, Strongyloides, malaria |
| Prion disease | CJD, vCJD, fatal familial insomnia, Gerstmann-Sträussler-Scheinker syndrome |
Abbreviations: CJD, Creutzfeldt-Jakob disease; CMV, cytomegalovirus; EBV, Epstein-Barr virus; HbsAg, hepatitis B surface antigen; HIV, human immunodeficiency virus; HSV, herpes simplex virus; HTLV-1, human T-lymphotropic virus 1; SARS, severe acute respiratory syndrome; vCJD, variant CJD; VZV, varicella zoster virus.
History and Examination
History
Certain clinical criteria are necessary prior to the diagnosis of brain death:
- A diagnosis compatible with brain death (e.g., intracranial hemorrhage [ICH], subarachnoid hemorrhage [SAH])
- Exclusion of confounding factors: no severe electrolyte, endocrine, or acid-base abnormalities
- Exclusion of intoxicating drugs, poisoning, or neuromuscular blockade
- Midazolam has active metabolites, and propofol can accumulate. Effects can be seen for prolonged periods after discontinuation of medications. Check urine toxicology for opiates or benzodiazepines and illicit drugs. Benzodiazepines can be reversed with flumazenil and opiates with naloxone. There is no urine or blood test for propofol.
- Reverse neuromuscular blockade with neostigmine/glycopyrro-late; check a train-of-four stimulus to confirm reversal. On the maximal stimulus over the median nerve four sharp twitches of the thumb and fingers should be seen if neuromuscular blockade is reversed.
- If a known drug cannot be quantified, the patient should be observed for 4x the half-life of the drug prior to proceeding with the brain death examination, or a confirmatory test can be performed (see below).
- Midazolam has active metabolites, and propofol can accumulate. Effects can be seen for prolonged periods after discontinuation of medications. Check urine toxicology for opiates or benzodiazepines and illicit drugs. Benzodiazepines can be reversed with flumazenil and opiates with naloxone. There is no urine or blood test for propofol.
- Patient must not be hypothermic (core temperature <32°C) or hypotensive (systolic blood pressure [SBP] <90 mm Hg).
Physical Examination
Brain death is a clinical diagnosis based on the physical examination. In most hospitals the brain death exam can be performed by a neurologist, neurosurgeon, or other specially designated physicians. Typically, two neurologic exams performed 6 hours apart and an apnea test are required to confirm brain death.
- Vitals: Assess for hypothermia, urine output (assess for diabetes insipidus, urinary output [UOP] ≥5 cc/kg/h x 2 hours, or urine-specific gravity < 1.005), and hypotension.
Neurologic Examination
- Mental status: Patient should have no response to verbal, tactile, or painful stimuli.
- Cranial nerves (Table 14.2)
- Motor: No response to deep nail bed pressure in any extremity
- Sensory: No motor or hemodynamic (tachycardia) response to pain.
- Reflexes: Brisk reflexes do not exclude brain death. A variety of exam findings, including C-spine reflexes (seen in 44–75% of patients)3,4 can be compatible with brain death (Table 14.3).
| Brainstem Reflex | Cranial Nerves Tested | Response |
| Pupillary reflex | Afferent: II, Efferent: III | Midposition fixed (4 mm) or fixed and dilated (9 mm) |
| Corneal reflex | Afferent: V, Efferent: VII | No blink bilaterally to limbus stimuli |
| Oculocephalic/ doll’s eye | Afferent: VIII, Efferent: III, IV, VI | Eyes move with head as head is turned (no doll’s eye effect) |
| Oculovestibular/ cold calorics | Afferent: V, VIII, Efferent: III, VI | No eye movement bilaterally when 60 cc of cold water instilled on intact tympanic membrane bilaterally (5 minutes apart) |
| Cough/gag | Afferent: IX, X, Efferent: X | Absent cough/gag |
| • Triple flexion | • Undulating toe (flexion/extension) | • Sweating, flushing, tachycardia, blood pressure swings |
| • Babinski | • Finger flexor movement | • Opisthotonus |
| • Lazarus (looks like Moro reflex) | • Semirhythmic facial spasm (due to denervated seventh nerve) | • Tonic neck flexion (passive neck movement induces fencer posturing or stereotyped trunk or extremity movement) |
Differential Diagnosis
- Brain death
- Locked-in syndrome (pontine ischemia or hemorrhage): Evaluate for command following with vertical eye movement or blinking.
- Drug intoxication, neuromuscular blockade
Life-Threatening Diagnoses Not to Miss
- Hypothermia: Temperature must be ≥32°C to proceed with brain death testing.
- Metabolic disarray (i.e., acidosis)
- Endocrine abnormalities: Hypothyroidism/myxedema coma
Diagnostic Evaluation
Apnea Test
Complications occur in 25% of patients undergoing an apnea test, most often in patients with inadequate preoxygenation, acid-base or electrolyte abnormalities, or preexisting arrhythmia.5 To avoid such complications, normalize pH, preoxygenate to PO2 >200 mm Hg, and ensure that core temperature >36.5°C. CO2 rises roughly 3 mm Hg per minute. Follow steps shown in Table 14.4.
| 1. | Check baseline ABG. A baseline pCO2 ≥40 mm Hg makes it more likely that the apnea test will reach target pCO2 levels. |
| 2. | While the patient is connected to an O2 sat monitor and BP monitor, disconnect the ventilator and use 100% 6 L/min flow-by. |
| 3. | A physician designated to declare brain death monitors for chest excursion or respiratory effort after ventilator disconnect. |
| 4. | Check an ABG after 8 min. |
| 5. | If pCO2 ≥60 mm Hg or increased by 20 mm Hg over baseline pCO2 and no respiratory movement observed: the apnea test supports brain death. |
| 6. | Abort the test and reconnect the ventilator if the SBP < 90 mm Hg, the patient has a significant desaturation, or an arrhythmia develops. Draw an immediate ABG. |
| 7. | If the patient cannot tolerate an apnea test, a confirmatory test must be performed. |
Abbreviations: ABG, arterial blood gas; BP, blood pressure; sat, saturation; SBP, systolic blood pressure.
Confirmatory Tests
Confirmatory tests are necessary when there is severe facial or C-spine trauma (do not perform doll’s eye maneuver on patients with C-spine injury), surgical pupils or preexisting pupillary abnormality, sleep apnea or chronic obstructive pulmonary disease (COPD) with chronic CO2 retention or toxic levels of sedatives, aminoglycosides, tricyclic antidepressants, anticholinergics, chemotherapeutics, or neuromuscular blockade. Examples of confirmatory tests include:
- Digital subtraction angiography (DSA): Gold standard—absence of intracranial blood flow at the level of carotid bifurcation or circle of Willis. The external carotid artery typically fills. Bilateral anterior circulation and posterior circulation injections are required.
- Computed tomography angiography (CTA) or magnetic resonance angiography (MRA): similar to above.
- Transcranial Doppler ultrasound: Isolated systolic flow (no diastolic flow) or reverberating flow are compatible with brain death. Both bilateral anterior circulation and posterior circulation must be insonated and reveal isolated systolic or reverberating flow. The sensitivity of TCD for brain death is 91 to 99%, and the specificity is 100%. Absent temporal windows occur in 10% of the population.6
- Electroencephalography (EEG)-electrical-cerebral silence: Criteria include >2 microvolt sensitivity, 1 to 30 Hz, minimum of eight electrodes 10 cm apart, impedance 100 to 10,000 O, and 30 minutes of recording. Due to frequent ICU artifact, it is difficult to use EEG to confirm brain death.
- Radionucleotide study (technetium single-photon emission computed tomography [SPECT]): Empty lightbulb sign. No radionucleotide uptake intracranially.
- Somatosensory evoked potential (SSEP): Absent N20-P22 bilaterally; less useful in patients with neuropathy or primary brainstem pathology.
- Electroencephalography (EEG)-electrical-cerebral silence: Criteria include >2 microvolt sensitivity, 1 to 30 Hz, minimum of eight electrodes 10 cm apart, impedance 100 to 10,000 O, and 30 minutes of recording. Due to frequent ICU artifact, it is difficult to use EEG to confirm brain death.
Notification of Next of Kin
The physician is responsible for informing the next of kin of the patient’s prognosis, though consent is not needed to perform a brain death examination. Once a patient is declared brain dead, the patient is legally dead and can be extubated unless the patients next of kin has consented to organ donation. Reasonable accommodations should be made for the family’s religious and moral beliefs, and life support should be continued for a reasonable amount of time based on family wishes. Brain-dead patients can survive typically for only 1 to 2 weeks after brain death is declared (97% suffer cardiac death within 7 days).7
Notification of Organ Donor Services
Regional organ donor networks should be informed when:
- Impending brain death is suspected
- Withdrawal of care is planned
- After all deaths for potential cornea or tissue donation
Organ donor representatives, rather than the primary physician team, should reach out to the family for potential donation to avoid a conflict of interests. This is typically done after the second brain death examination and apnea test have been performed.
Treatment and Care for the Potential Organ Donor
Sympathetic Surge
The final stage of brain death is typically medullary-level death, at which point a sympathetic surge occurs with subsequent hypertension, and potentially, cardiac stunning with myocyte necrosis and troponin leak. Intracranial pressure (ICP) is typically elevated, and a Cushing’s triad (elevated ICP, bradycardia, and abnormal respiratory pattern) may be seen. Spinal cord ischemia coincides with herniation, resulting in deactivation of the sympathetic nervous system and hypotension.
Pan-Hypopituitary State
With brain death comes a pan-hypopituitary state with concomitant diabetes insipidus and hypotension. Hormonal therapy addresses this pan-hypopituitary state and has been shown to reduce cardiovascular lability, electrocardiogram (ECG) abnormalities, and acid-base disturbances. Hormonal therapy has been shown to improve transplant rates and graft function. Patients refractory to volume and vasopressors have been shown to have a significant response to hormonal therapy such that >50% can be weaned off other vasopressors.8 In a study of 10,292 brain-dead donors, hormonal therapy with thyroxine, insulin, and pitressin yielded 22.5% more organs with significant increases in kidney, heart, liver, lung, and pancreas donation.9 Graft loss and graft dysfunction posttransplantation are significantly lower in organs that were managed preoperatively with hormonal therapy.10 Means of employing hormonal therapy are outlined below.
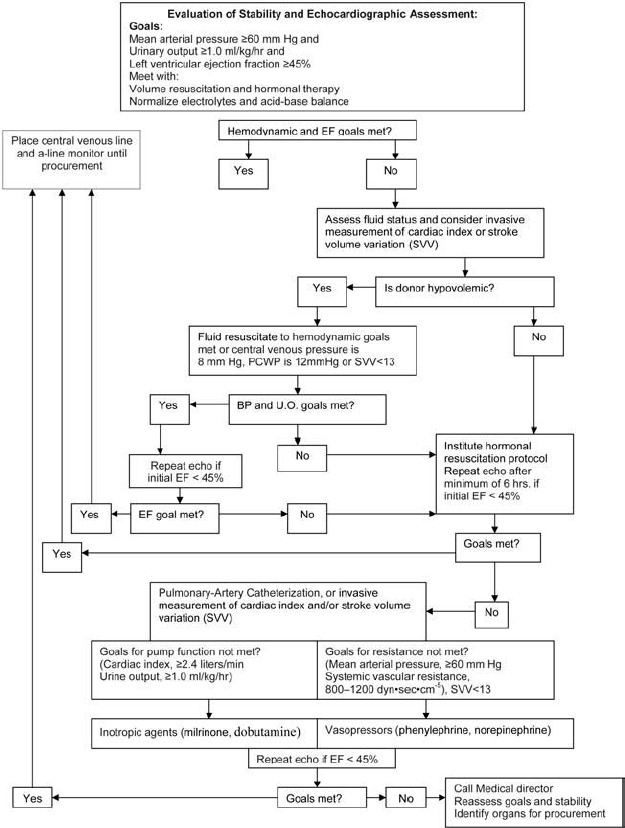
Flowsheet of Organ-Sparing Therapy
Organ-sparing therapy should be initiated as soon as brain death is evident or impending brain death is suspected (Fig. 14.1).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








