Fig. 6.1
Counting chamber. Nucleus-containing cells are counted within the double-lined space. Cells lying on the upper or left border are counted and on the lower or right border are not counted. The result is divided by 3 to obtain the cell count per μL
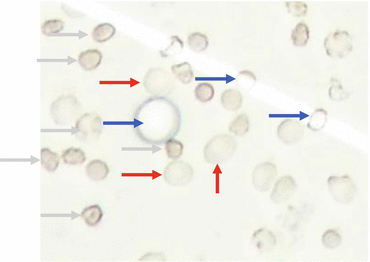
Fig. 6.2
Cell count. Red, erythrocyte: no nucleus, double-ring structure, grey, leucocyte. Irregular border of cytoplasm, inside visible structure. Blue, liposomes: round form, regular, slim border, optically empty
A total cell count of up to 4 cells per microlitre is normal; 5 or more is pathological. In cases of strong artificial contamination, 1 leucocyte per 1,000 erythrocytes can be subtracted from the total cell count as a rule of thumb.
Automated cell count has been propagated by several manufacturers of cytometers designed for blood cells and is used by some laboratories. These machines provide a reasonable cell count only in case of marked pleocytosis, where an exact result is less relevant. With normal cell counts or mild pleocytosis, where precise assessment is required to differentiate between normal and elevated counts, the result of automated cell counting is far less exact. Moreover, contamination with peripheral blood interferes with the results of automated cell counting. Automated analyses of CSF cell counts should therefore be regarded with caution and approved personally by an experienced technician (Strik et al. 2005; Heller et al. 2008).
6.5 Cell Differentiation
Differentiation of the different types of physiological and pathological CSF cells requires preparation and staining. Two different methods of preparation are used: sedimentation with the Sayk chamber and centrifugation with the cytospin method (Lehmitz and Kleine 1995). No clear recommendation can be made for either method, since the selection rather relies on personal preference and being familiar with one method. The sedimentation method is said to have a better efficiency of cell collection and to avoid preparation artefacts (Kolmel 1977; Gondos and King 1976). In our experience, however, cell preservation appears to be better with the cytospin method, probably because the preparation is faster and avoids autolysis (Hansen et al. 1974; Woodruff 1973). Centrifugation artefacts are rare and easy to detect and will not lead to misinterpretation. The technique of cytospin preparation is described in Fig. 6.3.
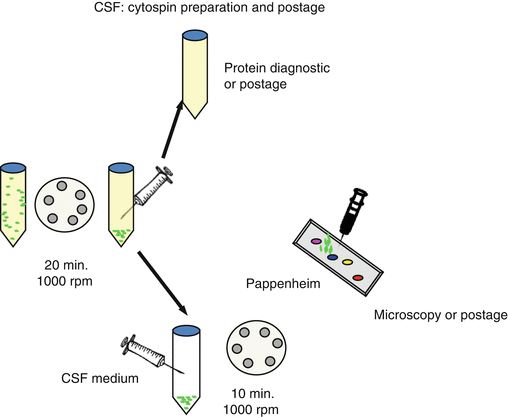

Fig. 6.3
Cytospin preparation. The CSF sample is centrifuged 20 min. with 1000 rpm. The supernatant is used for protein analysis. The remaining cells are resuspended with CSF medium containing fetal calf serum and buffer. Subsequently, the cell-containing medium is centrifuged for 10 min. at 1000 rpm on the slide. After air-drying, the preparation can be stained with May-Gruenwald – Giemsa (Pappenheim) or stored or posted for reference evaluation
Staining of the prepared cells is usually done with the Pappenheim method, a combination of May-Grünwald and Giemsa staining (Koelmel 2003). Basophilic structures are stained dark violet, eosinophilic in pink. Erythrocytes are stained in grey-shaded pink and haemosiderin in dark blue.
6.6 Physiological Cell Types
Leucocytes and monocytes (Fig. 6.4) are part of a physiological cell composition (Oehmichen 1976; Kluge et al. 2006; Worofka et al. 1997). Single polymorphonuclear, basophilic or eosinophilic granulocytes can be present. Single erythrocytes are often intermingled as a contamination artefact during lumbar puncture (Table 6.1).
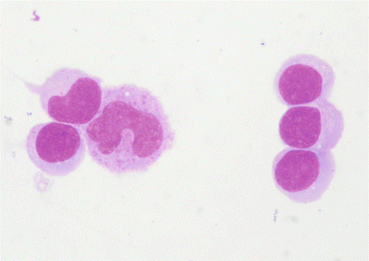

Fig. 6.4
Small- to medium-sized lymphocytes, round with thin cytoplasm (right and lower left) and monocytes with a broader, less homogeneous cytoplasm. Typical ratio of 2:1
Table 6.1
Cell types within the CSF
Physiological cells |
Lymphocytes and monocytes, ratio 3:1–4:1 |
Single granulocytes or erythrocytes |
Alterations of physiological cells |
Lymphocytes: inflammatory changes, lymphoid cells, plasma cells |
Monocytes: activated monocytes (small vacuoles), macrophages (larger vacuoles, probably visible object of phagocytosis like cells, erythrocytes, infectious agents) |
Cells lining the CSF space and rarely occurring cells |
Cartilage cells |
Bone marrow cells |
Ependymal, plexus, leptomeningeal cells |
Multinucleated giant cells |
Inflammatory reactions |
Lymphocytic cell composition |
Granulocytic cell composition |
Monocytic and macrophage reaction – reparative phase |
Haemorrhagic changes |
Erythrophages, masked erythrophages, siderophages, haematoidin-containing macrophages |
Neoplastic diseases |
Neoplastic morphology (see Table 6.2) |
Pigmented melanoma cells |
Blast-like cells in haematological malignancy |
Signet ring cells in adenocarcinoma |
Leucocytes are characteristically small to intermediate sized, approx. 1–1.5 times as large as an erythrocyte. They are round with round nuclei and have regularly shaped borders. Monocytes are more irregularly formed, often beanlike shaped, and can contain small vacuoles which are interpreted as a sign of unspecific activation. Erythrocytes are small and round and have a double-ring structure because they lack a nucleus.
Granulocytes can sporadically occur in a normal cell composition without being a sign of a pathological process. Most often, polymorphonuclear granulocytes are seen, small round cells with highly irregular, often intersected nuclei (Fig. 6.5). Less frequently, eosinophilic (Fig. 6.6) or basophilic granulocytes are found.
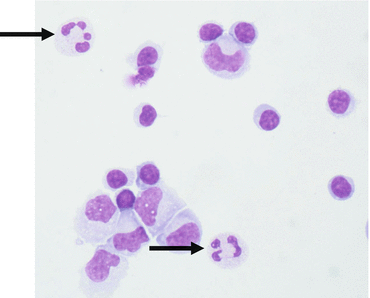
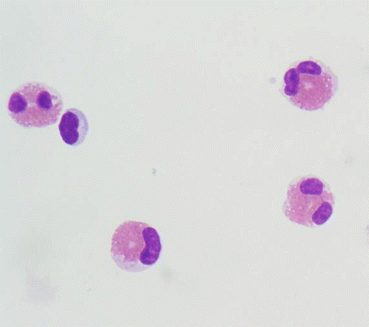

Fig. 6.5
Normal cell composition with single polymorphonuclear granulocytes (arrows)

Fig. 6.6
Multiple eosinophilic granulocytes, one small lymphocyte
A normal cell composition consists primarily of lymphocytes and monocytes with a relation of 3:1–4:1 and probably single granulocytes and artificially contaminated erythrocytes.
6.7 Cell Types Lining the CSF Spaces
Several cell types that are located in close vicinity to the CSF spaces can occur in the CSF (Worofka et al. 1997):
Erythrocytes, which contaminate the CSF sample during the puncture, have already been mentioned (Fig. 6.7).
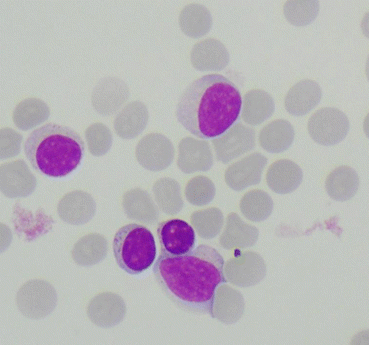

Fig. 6.7
Lymphocytes and peripheral blood. Erythrocytes are small and stain greyish-red. The absence of phagocytosis indicates that the erythrocytes result from artificial contamination
Cartilage cells can be intermingled when the puncture is too deep and reaches the vertebral disk. Cartilage cells can be somewhat larger than physiological CSF cells and dark basophilic (Fig. 6.8). A less experienced cytologist may mistake these cells as malignant cells.
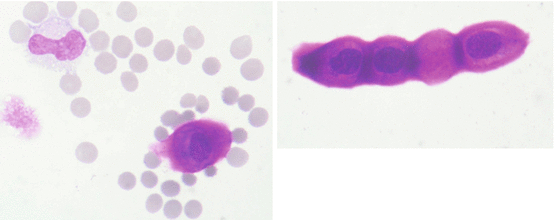

Fig. 6.8
Left: cartilage cell with typical dark basophilic staining of the nucleus and cytoplasm (lower right), monocyte (upper left), autolytic cell (middle left) and artificial blood contamination. Right: cartilage cells lined up
Bone marrow cells can contaminate the CSF during the puncture if blood-forming bone is affected. Therefore, blood precursor cells like erythroblasts or myeloblasts should be clearly identified. One hallmark to detect bone marrow contamination as opposed to malignant proliferation is the occurrence of cells from different lineages in different stages of development, while neoplastic proliferation will affect only one lineage and one certain stage of development. In case of bone marrow contamination in haematological malignancies, blasts in the CSF are not a proof of neoplastic meningitis, since artificial contamination deriving from the bone marrow cannot be differentiated from neoplastic affection of the CSF.
Other cell types that may enter the CSF space include choroid plexus (Fig. 6.9), ependymal (Fig. 6.10) and leptomeningeal cells (Fig. 6.11). Ependymal cells are up to approx. two times larger than leucocytes; their eosinophilic cytoplasm and regular appearance make them relatively easy to identify. Plexus cells are relatively small, round and basophilic and usually occur in agglomerations. Leptomeningeal cells are smaller than small lymphocytes and bean shaped and always occur in agglomerations of multiple cells.
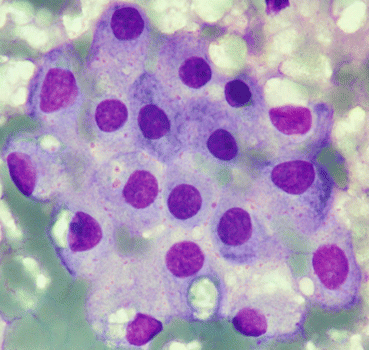
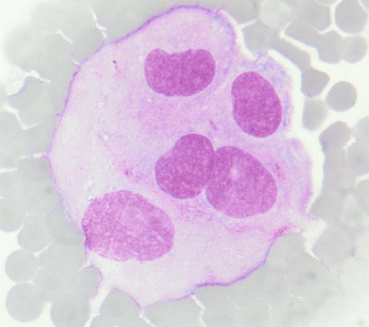

Fig. 6.9
Plexus cells with round nucleus and basophilic cytoplasm. Background of detritus with confluent erythrocytes

Fig. 6.10




Ependymal cells: large, eosinophilic, bean-shaped nucleus, loose cytoplasm. Peripheral blood in the background
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






