Fig. 7.1
Childhood strategies to improve adult cognition
It is generally agreed that in adults, the prevention, identification, and treatment of diabetes and the metabolic syndrome may prevent or delay cognitive decline. The interactions between cardiovascular and cerebrovascular disease, Alzheimer’s disease, depression, and other conditions such as tobacco use are still being delineated. For example, the presence of the apolipoprotein E (ApoE) ε4 allele (gene), involved in lipoprotein catabolism and modulation of the immune system, is predictive of both the development of Alzheimer’s disease, and non-Alzheimer’s cognitive decline as well [1]. However, we note that these causative conditions may differ between the countries where these risk factors have been identified and a low-income sub-Saharan country such as Uganda. For example, most (98–99 %) of the genetic variation in the human race exists in Africa [2, 3]. Other genes besides ApoE are likely important determinants of cognitive decline. Thus, the genetic contributions to diabetes and the metabolic syndrome can be expected to differ between African and non-African populations. Furthermore, traditional African diets are often low in saturated fats and high in fruits and vegetables and are closer to the Mediterranean diets than to the typical ‘Western’ diet which is high in animal protein and fats, and low in fruits and vegetables. Lastly, there is now good evidence that promoters of cognitive loss – diabetes, obesity, and cardiovascular disease – may evolve somewhat differently in low and middle income countries than in the better studied high income countries [4–6]. All of these argue for additional research into not only causation but also prevention for the African population.
A substantial body of literature has addressed the loss of both grey and white matter brain in the aging brain, as well as changing neuronal morphology with decreasing dendritic arbors and spines. Total brain volume loss, corrected for total intracranial volume, has been shown to be predictive for the subsequent development of cognitive impairment [7], as has volume loss of the amygdala and hippocampus [8]. In sum, studies conducted in adults has shown that diminished brain volume, and architectural damage at the cellular level via microvascular disease, contribute to cognitive loss. In thinking through the circumstances in sub-Saharan Africa which may lead to cognitive loss, it is important that this central pathophysiological mechanism be considered. As outlined below, the iconic illnesses we discuss below are likely to all affect cognition through this well understood set of pathways. A consequence of this is that the biological plausibility for these illnesses being relevant is quite high.
Once cognitive impairment is present, care of the individual includes the prevention of infectious disease which becomes more common as dementia progresses. These include influenza and pneumococcal disease, urinary tract infections, good oral hygiene to reduce the risk of aspiration pneumonia, and skin integrity to prevent cellulitis and bedsores. This adds to the burden of disease and expenses related to care of the affected individual. Although this is not the focus of our chapter, the spectrum of such conditions may also differ between Western countries and Africa, and childhood infections, vaccinations, and immunological experience may prove important.
Prevention and treatment of cognitive loss is still an evolving topic. Dietary interventions such as the consumption of fruits and vegetables with high levels of anti-oxidants, omega(n)-3 (or ω-3) fatty acids, low intake of saturated fats, moderate alcohol intake, and regular exercise are believed to reduce the risk of cognitive loss by acting against a number of the underlying causal conditions, such as metabolic syndrome or diabetes. A systematic review by Plassman et al. [9] examined studies of factors in five domains: nutritional; medical, social, economic or behavioral; toxic environmental exposures; and genetic. They identified the consumption of ω-3 fatty acids, vegetables and a Mediterranean diet, physical exercise, and cognitive engagement as having at least some level of supportive evidence as ways to reduce risk. (We note that many vegetables and components of the Mediterranean diet contain alpha-linoleic acid (ALA), which can be converted to eicosapentaenoic acid (EPA) and docaosahexaenoic acid (DHA), which are the two ω-3 fatty acids found in fatty fish). Recent studies of cognitive impairment in children in high-income countries have tended to focus on the adverse effects of the heavy metal lead (Pb); social neglect and abuse; and childhood chemotherapy. We note that heavy metal contamination is frequently present in both high and low-income countries, and (for example) that there is no “safe” level of exposure to lead since even low levels of lead exposure can lead to permanent cognitive damage [10, 11]. However, in low and middle income countries, such as Uganda, other threats to childhood cognition exist. These include specific dietary deficiencies such as iron deficiency anemia, early childhood stunting, and other forms of undernutrition, as well as diseases such as malaria. Over 160 million children globally are stunted, and iron-deficiency anemia is one of the most common nutritional disorders. Murray and colleagues estimated in 2012 that 207 million cases of malaria occurred, primarily in pregnant women and children in sub-Saharan Africa [12] There are well documented effects of anemia on cognition; stunting is accompanied by microcephaly (abnormally low brain volume) and decreased synaptic complexity; and both severe and asymptomatic malaria are now being linked to adverse cognitive outcomes.
We believe a common thread relates brain development in utero and in early childhood to adult cognition and to the preservation of cognitive function in old age. We posit that combatting common childhood conditions, such as those outlined below, will lead to improved adult cognition and productivity. This, in turn, will decrease the risk that these individuals, as adults, will suffer from premature and age-associated cognitive declines given their improved brain reserves.
Stunting, Environmental Enteropathy, and the Gut Microbiome
Stunting is a physical manifestation of undernutrition which results in a child’s height being 2 standard deviations or more below the median height for the child’s age. Stunting is linked to poor cognitive development in childhood and adolescence, diminished motor development, and lower IQ. Stunted children perform less well than normally nourished children in school and demonstrate poorer cognition. In general, stunted children receive fewer years of schooling and demonstrate less productivity once they achieve adulthood [13–16]. Risk factors for stunting are many – they include undernutrition of the pregnant mother; inadequately nutritious diets; a lack of exclusive breast feeding and socio-economic status and wealth; poor water and sanitation; and many other factors which have been summarized in the recent 2013 Lancet series [17, 18]. New to the discussion of causality is the gut microbial community, or the gut microbiota. This community forms a ‘microbiome,’ or ecological community which contains both normal and pathogenic organisms. (Strictly speaking, the microbiome is the collective genomic structure of the microbiota, but the terms are sometimes used interchangeably). The gut microbiome of normal children is distinctly different from that of malnourished or obese children, and is influenced by environmental exposures.
The presence of stunting is assessed by measuring a child’s height (or length) and matching the measured height for age to that of a reference population of normally nourished children of the same age. Given the normal Gaussian variation of height, one expects 95.45 % of children to be within 2 standard deviations of the mean, and ~2.275 % of normal children to meet the definition of stunted. Similarly, 99.73 % of children should be within 3 standard deviations of the mean, with only 0.135 % of normal children having a height for age score 3 deviations below the mean. (The latter is the definition for severe stunting). The Uganda Demographic and Health Survey (DHS) conducted in 2011, however, reported that 33 % of Ugandan children under 5 years of age are stunted, and 14 % are severely stunted [19].
Stunting is accompanied by microcephaly, defined as an head circumference 2 or more standard deviations below the age-adjusted mean. Microcephaly is related to reduced brain volume. Brain growth is most rapid during the last trimester of pregnancy and the first year of life. Malnutrition results in smaller brain size, decreased brain myelinization, DNA and neurotransmitter content, and less cortical dendritic growth [20–22]. Prospective birth cohort data from diverse sites around the world have shown consistent relationships between early childhood stunting, and decreased cognition, worse economic productivity and achievement, and adult health outcomes [23–25]. It is difficult to imagine a disorder more ripe for attention that childhood stunting when it comes to adult cognition.
Transgenerational effects of stunting have been identified. The adverse effects appear early and continue into adulthood. For example, stunted girls grow up to become short women. Their birth canals are proportionately smaller, and the risk of obstructed labor increases as maternal height declines. In some countries, a practice of “eating down” (moderating food intake) in pregnancy exists, in recognition than a larger baby will cause a more difficult delivery, especially in the setting of small maternal size [26]. Maternal height is inversely associated with child mortality, stunting, underweight and wasting, based upon an analysis of 109 DHS surveys conducted in 54 low and middle income countries [27]. This study included data of 2,661,519 children born to 751,912 mothers after exclusion of outliers or incomplete case records. For every 1 cm increase in height, the risks of these outcomes significantly decreased. Short stature mothers (<145 cm) had a ~40 % greater risk of a child dying than women >160 cm, and the prevalence of stunting and underweight was doubled in the offspring of the shortest group after adjustment for confounding variables such as socioeconomic and educational variables.
Maternal overweight and obesity is becoming more common globally just as childhood overweight and obesity, and these appear linked, almost paradoxically, to early childhood undernutrition via epigenetic programming, a form of metabolic imprinting [28, 29]. By this we mean that nutritional gaps in utero predispose the stunted infant to permanent changes in homeostatic mechanisms which favor the later development diabetes, obesity, and the metabolic syndrome [30, 31]. For example, in support of this thesis, low birth weight infants are at elevated risk of childhood insulin resistance [25, 32], an hallmark of diabetes and metabolic syndrome, and hypertension and plasma triglycerides, after adjustment for current size of the child at 9–10 years of age [33]. This form of permanent adaptation to the conditions suffered in utero or in early childhood differs from accommodative adaptation, where a person’s metabolism changes to fit a temporary situation and eventually reverts back to a prior set point.
How, then, do these relate to environmental enteropathy and the intestinal microbiome? Scientists is the 1960s and 1970s identified a condition first termed tropical and then environmental enteropathy (EE), as it is widely prevalent in tropical countries where environmental contamination is ubiquitous [34, 35] (Yet another term has been proposed, “acquired environmental enteric dysfunction” [36]). It is characterized by asymptomatic villous atrophy, affecting nutrient absorption, and an inflamed, leaky intestine. This enteric dysfunction is characterized by increased caloric needs to maintain growth, the chronic anemia seen with inflammatory conditions, and increased gut permeability. In a series of landmark studies, Lunn and other co-workers in the 1990s showed that the single best predictor of stunting was gut permeability – an hallmark of EE [37]. Current thinking is that constant exposure to fecal-oral contamination, and repeated episodes of infectious gastroenteritis, “lead to a perpetual state of small bowel injury” [38]. Humphrey cogently synthesized this work going back to the 1960s forward and developed a simple yet accurate story line: EE is caused by pathogenic fecal bacteria ingested by young children living in unsanitary conditions [39]. In turn, the elevated metabolic demands of enteropathy and state of chronic inflammation lead to growth retardation, potentially through inflammation-mediated inhibition of the growth factor-insulin-like growth factor axis [40]. Recent cross-sectional data from Bangladesh shows significant relationships between fecal environmental contamination (based upon water testing and an household inspection), the presence of environmental enteropathy, and stunting [41]. The implication is clear: protect young children from the fecally contaminated environment, and you prevent stunting and other forms of malnutrition, and potentially the adverse adult consequences as well [42]. One needs clean water and sanitation – toilets – to accomplish this.
The final piece to this evolving picture is the discovery that the microbiome of malnourished children is not only abnormal, it can even promote malnutrition. Smith et al. in 2013 [43] reported on studies in 317 genetically-identical twin pairs in Malawi where one twin was normally nourished and the other had kwashiorkor, a form of stunting that includes body edema (a puffiness from fluids that leak out of the blood vessels and are not reabsorbed). The microbiomes of the normal and malnourished twins were markedly different, and in the case of the malnourished children less mature (the microbiota changes as a child gets older). Furthermore, nutritional supplements designed to rehabilitate the malnourished children helped them gain weight but did not change their microbiomes. Shockingly, when the microbiomes of malnourished children were transferred into germ free mice fed a Malawian diet, the mice lost ~ a third of their body weight in 18 days. In contrast, the transplanted microbiomes of their better nourished siblings did not cause any weight loss in mice. Metabolic analysis suggest that Biophilia wadsworthia, a sulfur-consuming bacterium found in the altered microbiota of malnourished children, consumes sulfur-containing amino acids which are already deficient in their diets. (This common anaerobic organism in the human microbiota is the only known intestinal organism to use taurine, a derivative of the amino acid cysteine, as its source for sulphite, its final electron acceptor for respiration. Taurine is essentially absent from vegan diets) [44]. Along the same lines, they found evidence that the microbiome was selectively interfering in the harvesting of dietary energy via the aerobic Krebs citric acid (tricarboxylic acid, TCA) cycle. Because it is well established that our gut flora reflects both our diet and our environment, we posit that an adequate diet and clean environment should prevent this circumstance.
In summary, we see that stunting is the product of under-nutrition (of both the mother and the child), specific poor feeding practices and micronutrient deficits, a lack of clean water and sanitation, the development of environmental enteropathy and the presence of an abnormal microbiome. It is of no little interest that obesity is associated with an abnormal microbiome, which if replaced through a fecal transplant leads to improved insulin sensitivity. Metabolic syndrome, diabetes, and obesity are characterized by low-grade inflammation, as well as increased gut permeability (reviewed in Shen et al. 2013) [45]. Figure 7.2 relates these findings to the microbiomes of normal and obese individuals.
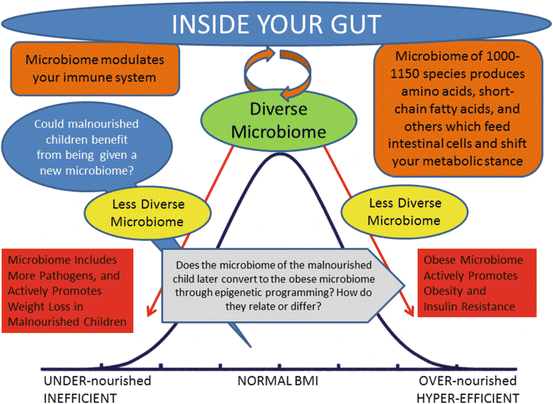

Fig. 7.2
The gut microbiome contributes to both stunting and obesity, which may be related through epigenetic programming. Both the malnourished and obese microbiomes are less diverse than the microbiomes of normally nourished individuals
Iron-Deficiency Anemia (IDA)
Anemia is one of the leading nutritional disorders globally, and the third largest cause of disability. The majority of anemia in children is due to iron deficiency, often compounded by infectious diseases such as malaria and hookworm infection which either destroy or consume red blood cells [46]. It has been repetitively identified as a predictor of decreased cognitive performance [47]. A number of studies have identified iron deficiency anemia (IDA) in infancy and childhood as long-term predictors of decreased cognition, even if treated with iron supplementation (reviewed in Beard and Connor 2003) [48]. Iron is required for brain myelinization, neurotransmitter (gamma-aminobutyric acid, GABA) metabolism, and oxygen delivery to the brain. Children with IDA have cognitive and motor deficits and delayed socio-emotional and neurophysiologic development compared to children without IDA (reviewed in Lozoff et al. 2006) [49]. The majority, but not all, of studies assessing cognition before and after treatment with iron have reported persistent deficits as well.
In an important long-term study of Costa Rican children, 185 children were identified between the ages of 12 and 23 months. Iron status was assessed and all children received iron therapy, with uniformly excellent responses and resolution of anemia. However, formerly IDA children tested in adolescence and at 19 years of age had persistent motor impairment, need for school grade repetition, depression and anxiety, inattention, impaired executive function, and longer visual-evoked potential latency times when compared to peers who had not had IDA [50, 51]. Dramatically, in the low socioeconomic group, mean IQ was 70.4 in those who had had chronic IDA versus 95.3 in those without IDA. In middle-class children, the respective IQ scores were 93.1 versus 102.8. No evidence of a “catch-up” in cognition was visible for children with chronic iron deficiency in infancy despite iron repletion at the time of study enrollment.
Iron deficiency anemia can be prevented in many ways. These include a diet with sufficient iron and the other necessary cofactors required for its uptake, iron fortification of staple foods, and the prevention or treatment of diseases such as malaria and hookworm.
Malaria
Cerebral malaria is a severe neurological manifestation of infection with Plasmodium falciparum, the causative agent of falciparum malaria. Coma and seizures are both hallmarks of cerebral malaria. In Uganda, infection with this species of malaria parasite is the most common form of malaria [52]. Unlike other species of malaria, falciparum parasites developing in parasitized red blood cells (pRBCs) attach to endothelial cells lining the vasculature in a process called sequestration. This can lead to reduced local brain perfusion, depriving brain cells of glucose and oxygen, in multiple small areas of the brain. The adherence of pRBCs induces inflammatory changes including cytokine release, endothelial cell activation and death of nearby endothelium, neurons and glia [53]. The blood-brain barrier can be disrupted, and intracranial hypertension is common. Both cerebral malaria, and severe malarial anemia, have now been linked to long-term cognitive impairment as assessed at 1 year after hospitalization in studies conducted in Uganda [54]. Epilepsy develops in about 10 % of children, sometimes after a number of years, and the cumulative incidence increases over time [55]. Severe malaria is also associated with acquired language disorders in a similar percentage (~12 %) of survivors [56].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





