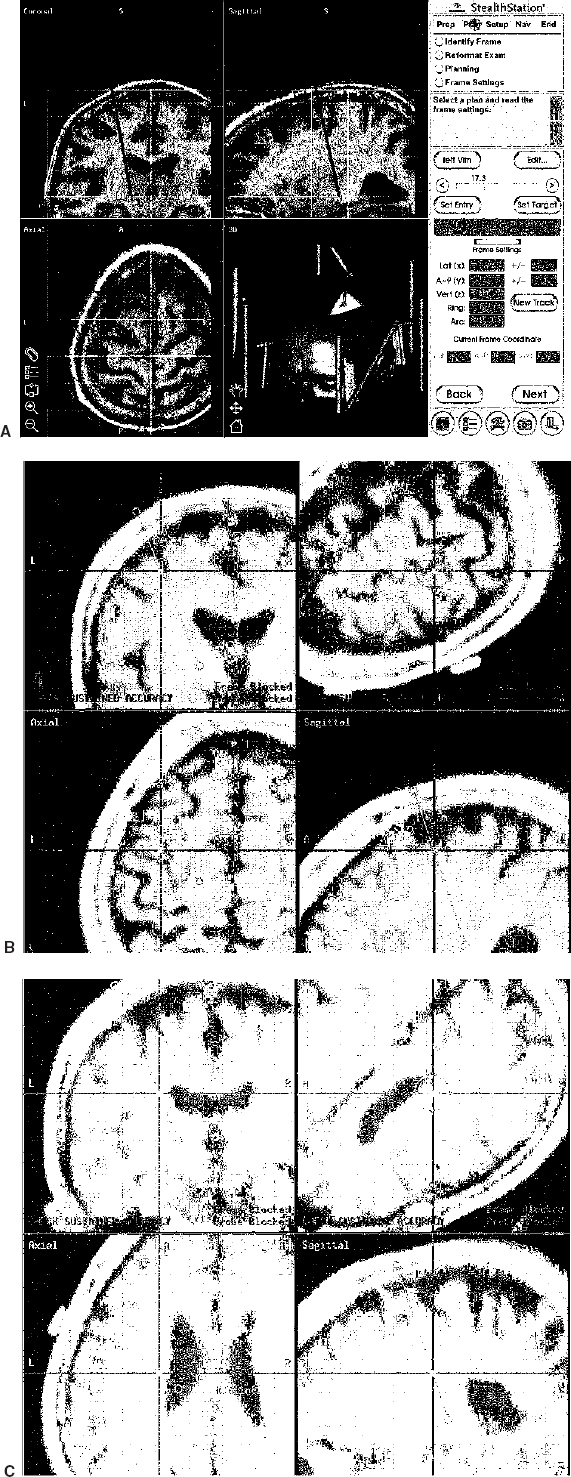
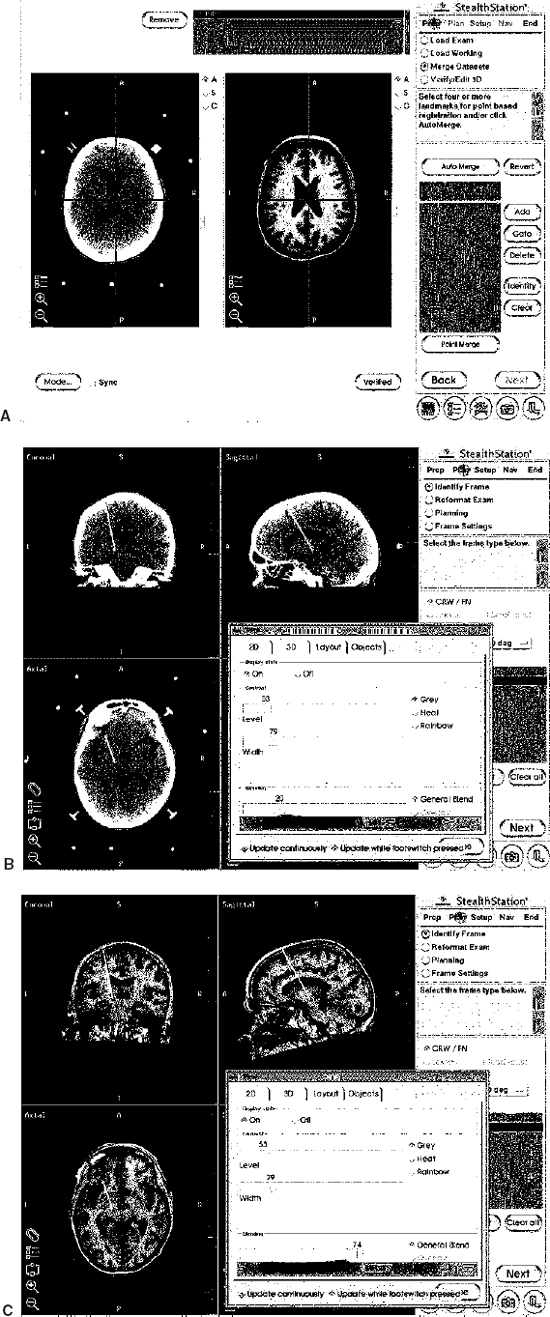
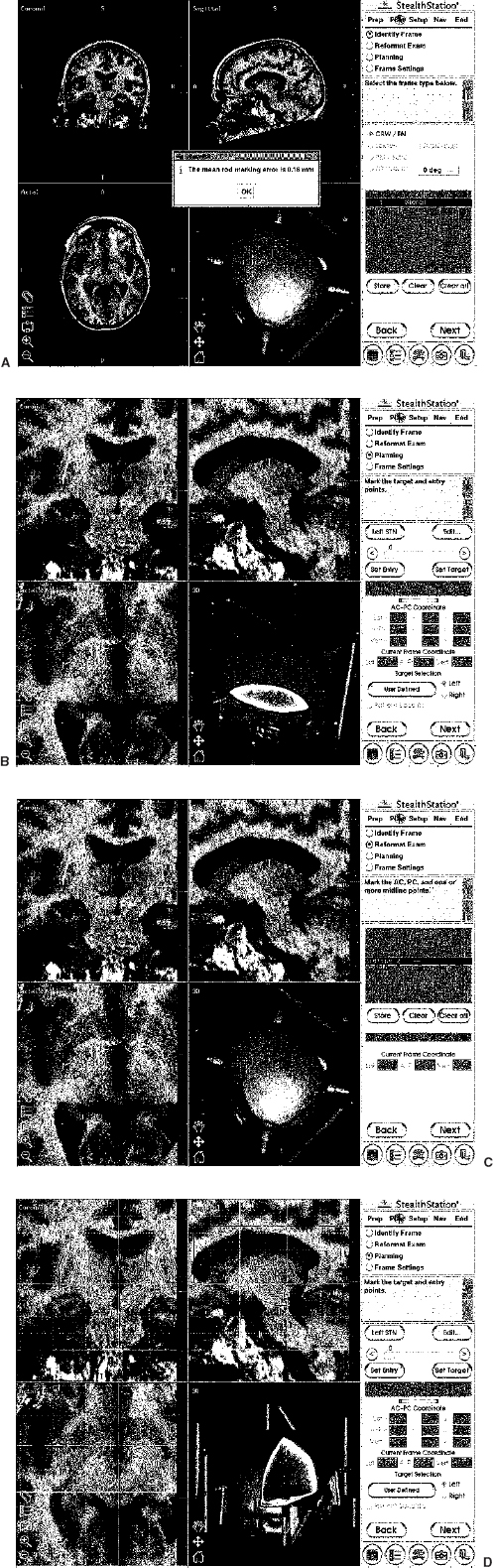
16 Stereotactic neurosurgery for movement disorders has always been challenged by the task of locating a deep brain target that might not be visible with currently used imaging techniques. In addition, the spatial anatomical variability of the subcortical structures represents a crucial factor in the determination of a target for stereotactic functional neurosurgery.1–3 Modern high-field magnetic resonance imaging (MRI) units provide great anatomical detail of the brain.4 Nonetheless, MR imaging alone is not sufficient to achieve precise localization of all targets used for neurosurgical treatment of movement disorders. To overcome this difficulty, different techniques have been used, including the use of stereotactic brain atlases. Recently, several groups5–13,15–25,27–30 have implemented these in the electronic form. Although most of these systems are highly sophisticated in contour overlay, they may lack precision in the sense that such overlays assume that the brains of patients contain proportionally the same relationship between the anatomical structures. In addition, the size of subcortical nuclei and subnuclei, which varies across normal individuals, can vary greatly in the presence of degenerative diseases or as a function of age.2,3,22 Thus, the accuracy of using atlases as adjuvant for targeting deep nuclei is still debated. Computer technology has been used at increasingly advanced levels to assist the neurosurgeon in the performance of stereotactic procedures.14,31 The basic role of computers in stereotactic neurosurgery is to perform the calculations that transform a point on the ventriculography or computed tomography (CT) or MR imaging film (the “image space”) into a target point in the three-dimensional coordinate system of the stereotactic frame (the “surgical space”). Most of the stereotactic systems currently in use employ a computer to help calculate the target based on a fiducial system placed on the patient’s head prior to the surgery. In addition, computers can be very helpful in displaying neurophysiological atlases, as described above. Finally, they can be used to integrate different imaging technologies with intraoperative real-time visualization of the recording electrode and/or ablative/augmentative electrode and the intraoperative microelectrode recording patterns.32,33 Thus, computer technology can assist the neurosurgeon in planning and performing stereotactic procedures for movement disorders. In addition, the preoperative and intraoperative dataset of each patient can be stored in the computer and used to compare with the patient’s postoperative images and neurological outcome. Data collated from such experiences can be used to create a probabilistic library that may be helpful in targeting future operations. In this chapter, we review the advantages and limitations of using computer-assisted technology for planning and validation of targets during neurosurgical procedures for movement disorders. The highest level of computer assistance in surgery for movement disorders is the integration of multiple coherent modalities into a unified, coherent display. Modern computer-assisted stereotactic systems integrate multiple imaging modalities, including CT, T1- and T2-weighted MR imaging sequences, and functional images. These anatomical data are correlated to real-time electrode position, scalable stereotactic atlases, and microelectrode recordings. A description of the system and technique used by the authors is given here (FrameLink, Medtronic SNT, Louisville, CO). On the morning of surgery, the MR-compatible Cosman-Roberts-Wells frame (CRW, Radionics, Boston, MA) is placed on the patient’s head in the standard fashion. A sagittal T1-weighted scout image is obtained to localize the anterior and posterior commissures. Axial images in the area of interest are then acquired in the plan of the AC-PC line [General Electric (GE) 1.5 T(Tesla); spin echo; repetition time (TR) = 800; TE = 50; thickness = 2 mm; interspace = 0, matrix 256 × 256; 2NEX (number of excitation)]. Targeting of the subthalamic nucleus, globus pallidus internalis, and ventral intermediate nucleus is performed, as previously described.32,33 The CRW coordinates are obtained in the standard fashion from the fiducials and target using the right-left anteroposterior superior-inferior (RAS) system on the GE console. An additional MR acquisition, encompassing the entire head without relationship to the AC-PC line, is obtained for the interface with the StealthStation (Medtronic SNT). Although the superior anatomical detail shown by MR images is recognized by most neurosurgeons, some surgeons prefer to minimize image distortion by using CT24 In this case, the head frame is applied prior to obtaining CT images. These are obtained using conventional stereotactic and image-guidance acquisition protocols. The MR image is typically obtained days before the surgical procedure and fused by the computer software to the CT images the day of the procedure (Fig. 16–1). The registration of the image space into the physical space is the pillar of computer-assisted image-guided or “frameless” neurosurgery. After completion of this process, the surgeon can use the surgical instruments that will be seen overlapped on the patient’s preoperative images on the computer screen.34 To allow coregistration of the frame with the image-guided frameless instruments, the frame needs to be registered with the image space and the physical space. The preoperative images acquired as described above are therefore transferred to the StealthStation by Ethernet, CD-ROM, or (digital audio tape) DAT tape. The computer software renders the previously acquired axial images and reformatted coronal, sagittal, and three-dimensional images. The dialogue box on the computer screen prompts the request of inputting the type of stereotactic frame used. After this information is entered, the stereotactic fiducials (i.e., rods) are entered by clicking on each of them with the computer mouse on only one axial image (Fig. 16–2A). At the end of this process, the CRW coordinates of any given point within the image space are automatically calculated and displayed in the dialogue box when clicking with the computer mouse on a chosen point (Fig. 16–2B). At this point, the target could be selected by using conventional stereotactic measurements on the computer images. Alternatively, the previously calculated CRW coordinates could be typed into the dialogue box. Additionally, the software permits an automated choice of the target using the dialogue box “User Defined” (Fig. 16–2B). FIGURE 16–1 Image fusion. Intraoperative photographs of the computer screen during the image fusion process. (A). First the CT dataset acquired with the stereotactic frame is loaded on the computer (left panel), then the MR image (right panel). After merging the two studies, the surgeon can change the blending function on the dialogue box to obtain images that are more CT-based (B) or more MR-based (C). FIGURE 16–2 Software interface. Intraoperative photographs of the computer screen during the software interface between frame-based and frameless technology. (A). To allow coregistration of the frame with the image-guided frameless instruments, the frame needs to be registered with the image space and the physical space. This is first identified using the dialogue box. (B). At the end of this process, the Cosman-Roberts-Wells (CRW) coordinates of any given point within the image space are automatically calculated and displayed in the dialogue box when clicking with the computer mouse on a chosen point. The CRW and AC-PC coordinates of the courser on the screen (arrow) are indicated on the dialogue box. (C). The first step necessary to overlap and size the Schaltenbrand-Wahren atlas to the patient’s images requires manual recognition of the AC, PC, and three midline points on the patient’s MR image. This is accomplished by clicking with the computer mouse on the appropriate anatomical structures. (D). At the end of this process, the previously selected target based on CRW coordinates is seen in relationship to the anatomical representation of the Schaltenbrand-Wahren atlas. The digitized Schaltenbrand-Wahren atlas is then sized to the patient’s MR image. The first step necessary in this process requires manual recognition of the AC, PC, and three midline points on the patient’s MR image. This is performed by clicking with the computer mouse on the appropriate anatomical structures (Figs. 16–2C) of each required point. At the end of this process, the atlas contours are then displayed on the three-planar images. Additional manual fitting of the atlas contour is achieved by manipulating the Talairach grid to size the known structures, such as the ventricular system. When the atlas sizing process is completed, the previously selected target based on CRW coordinates is seen in relationship to the anatomical representation of the Schaltenbrand-Wahren atlas (Figs. 16–2D). The explosive development of information technology and computer graphics presents a tremendous opportunity to create new tools for surgical training, planning, and procedures. Typically, for all stereotactic procedures, an entry point and a target point have been chosen based on anatomical landmarks or images information. With the latest computer software, however, after these two points are selected, it is possible to develop a surgical plan using “minimally invasive”; strategies. The ideal plan for depth electrode placement should avoid the involvement of sulci and the ventricular system to minimize cortical hemorrhage and shift related to CSF diversion, respectively. The entry point is first chosen on the triplanar images according to conventional criteria.32,33 After the entry point is selected, the software displays the trajectory of the planned pathway. This can be seen on the triplanar images as well as on images reformatted in the plan of the electrode, the “probe eye’s view” (Fig. 16–3). By moving the cursor on the screen in the dialogue box, the surgeon can perform virtual surgery and observe the electrode advancing in the brain on the computer images. Based on the feedback received by scrolling through the images, the trajectory can be modified until satisfactory. In the authors’ experience, preoperative planning is a very important step because it not only allows optimization of the placement of the deep brain electrode but also has the potential to reduce morbidity. Typically, surgical planning and virtual surgery are performed as the patient is positioned on the operating room table and intravenous anesthesia and monitoring are prepared.
Computer Technology as an Adjuvant for Target Location and Validation
ISABELLE M. GERMANO AND DONALD J. WEISZ
Technical Overview
Images Acquisition and Fusion
Software Interface
Atlas to Patient Registration
Surgical Plan and Virtual Surgery
Neupsy Key
Fastest Neupsy Insight Engine