and Héctor H. García3, 4
(1)
School of Medicine, Universidad Espíritu Santo, Santo, Ecuador
(2)
Department of Neurological Sciences, Hospital-Clinica Kennedy, Guayaquil, Ecuador
(3)
Cysticercosis Unit, Instituto Nacional de Ciencias Neurológicas, Lima, Peru
(4)
School of Sciences, Universidad Peruana Cayetano Heredia, Lima, Peru
Abstract
As previously discussed in this book, Taenia solium is endemic in vast parts of the world, in particular, in regions where pigs are raised as domestic animals in rural areas. The resulting disease, neurocysticercosis, is also present in non-endemic regions because of travel and immigration. Taeniasis/cysticercosis exerts a significant economic impact worldwide, most of which is associated with human neurocysticercosis, although there are other associated costs related to porcine cysticercosis (Bhattarai et al. 2012; Praet et al. 2009; Torgerson and Macpherson 2011). The substantial contribution of neurocysticercosis to the burden of neurological disease in Latin America has been estimated as between 23,512 and 39,186 symptomatic neurocysticercosis cases in Peru, and approximately, 400,000 in Latin America (Bern et al. 1999). Even in non-endemic regions, immigrants with neurocysticercosis may become a significant economic burden for health systems. General costs related to neurocysticercosis in California, USA, have been estimated to be $17 million, with hospital costs above $5 million (Croker et al. 2012).
As previously discussed in this book, Taenia solium is endemic in vast parts of the world, in particular, in regions where pigs are raised as domestic animals in rural areas. The resulting disease, neurocysticercosis, is also present in non-endemic regions because of travel and immigration. Taeniasis/cysticercosis exerts a significant economic impact worldwide, most of which is associated with human neurocysticercosis, although there are other associated costs related to porcine cysticercosis (Bhattarai et al. 2012; Praet et al. 2009; Torgerson and Macpherson 2011). The substantial contribution of neurocysticercosis to the burden of neurological disease in Latin America has been estimated as between 23,512 and 39,186 symptomatic neurocysticercosis cases in Peru, and approximately, 400,000 in Latin America (Bern et al. 1999). Even in non-endemic regions, immigrants with neurocysticercosis may become a significant economic burden for health systems. General costs related to neurocysticercosis in California, USA, have been estimated to be $17 million, with hospital costs above $5 million (Croker et al. 2012).
In disease-endemic regions, neurocysticercosis has been blamed to account for approximately one third of all epilepsies (Del Brutto et al. 2005; Montano et al. 2005; Ndimubanzi et al. 2010) after accounting for the baseline prevalence of serological markers and CT abnormalities in asymptomatic individuals. This proportion is quite consistent among studies (Ndimubanzi et al. 2010). From all causes of epilepsy, only a few are amenable to strategies for their decrease in the medium to long term, and even fewer could be eliminated. At the population level, the numbers of seizure cases related to motor vehicle accidents respond to enforcing legislation, perinatal epilepsies respond to improved prenatal control and better delivery conditions, stroke-associated seizures respond to secondary prevention (Newton and García 2012). None of these conditions can be foreseen to disappear. Conversely, if transmission of Taenia solium is eliminated, up to 30 % of all epilepsy cases could be prevented.
Being such a significant public health problem, actions on control and potential elimination of Taenia solium are urgently required. Actively intervening to control Taenia solium is not a new idea. Control interventions have been tested since 1985, and the International Task Force for Disease Eradication listed Taenia solium taeniasis/cysticercosis as potentially eradicable (International Task Force for Disease Eradication 1993), mainly because of the lack of contributing wild reservoirs, pig as an easily targetable intermediate host, and the availability of effective treatments for taeniasis (and now for pig cysticercosis), among other reasons. With this, taeniasis cysticercosis joined a reduced number of diseases targeted for elimination and potential eradication.
10.1 Principles of Control
10.1.1 Treatment of Taeniasis
The only source of infection for humans and pigs is the human tapeworm carrier. Moreover, the adult Taenia solium tapeworm has a very high biotic potential, meaning that a single worm can infect many hosts in its surroundings. For all these reasons the tapeworm carrier is the main target of control interventions. Tapeworm carriers may be treated and cured either by case diagnosis and individualized treatment or by mass treatment of the human population with a single oral dose of either niclosamide (2 g in adults) or praziquantel (5–10 mg/kg). Diagnosis of taeniasis is usually made by stool microscopy after concentration by sedimentation, with poor sensitivity (under 70 %) (García et al. 2007a). Detection of tapeworm antigens in stools (coproantigen detection), described by Allan et al. (1990), is a much more sensitive technique but is not widely available beyond research settings.
Mass treatment is logistically easier and likely much cheaper. However, there is a theoretical risk that the administration of antihelminthic drugs to an individual who has silent neurocysticercosis could trigger seizures or other neurological symptoms. This has been reported in a few individuals after a single dose of praziquantel (Flisser et al. 1993; Johnson 1986; Torres et al. 1988) or after a single dose of the commonly used antihelminthic albendazole (García et al. 2007b; Ramos-Zuniga et al. 2013). This seems to be a rare event since praziquantel is routinely administered in mass-treatment campaigns for schistosomiasis in Africa, some of which are also endemic for cysticercosis, and reports of neurological side effects are rare (Johnson 1986). Niclosamide does not have this drawback but it is poorly available in many parts of the drug (as it also happens with praziquantel). Adding mass niclosamide or praziquantel treatment to existing mass deworming programs has been suggested as a highly cost-effective control strategy (Alexander et al. 2011).
A series of experiences with mass chemotherapy have been performed in several countries. Between 1985 and 1987, Cruz et al. (1989) de-parasitized over 10,000 individuals in Loja, Ecuador, using a single dose of 5 mg/kg of praziquantel. They found 1.6 % of taeniasis by patient’s self-report and published a reduction in porcine cysticercosis after the program. This program, however, was interrupted soon after its implementation, and a few years after stopping control, transmission returned to levels close to baseline. Díaz Camacho et al. (1991) performed mass praziquantel treatment in a village of 559 individuals in Sinaloa, Mexico. The authors reported no taeniasis cases or cysticercosis-infected pigs 1 year after the intervention and noted a marked clustering around tapeworm carriers. However, 1 year after the intervention, porcine cysticercosis detected by tongue examination had raised from 6 to 11 % (Keilbach et al. 1989). From 1991 to 1996, Sarti et al. (2000) worked in three villages in Morelos, Mexico. In one of them, mass chemotherapy with 5 mg/kg praziquantel was 67 % effective to treat human taeniasis. The prevalence of taeniasis dropped by half, and there was also a long-term drop in porcine seroprevalence and a decrease in late-onset seizures. Health education was tested in one of these villages and shown to decrease transmission of taeniasis/cysticercosis very fast after the intervention (Sarti et al. 1997). In the third community, where both health education and mass praziquantel treatment were administered together, the prevalence of human taeniasis did not show an impact along time, but porcine prevalence decreased gradually (Pawlowski et al. 2005).
Between 1994 and 1996, Allan et al. (1997) used mass niclosamide therapy in approximately 2,000 people in two villages in Guatemala and dropped the prevalence of taeniasis from 3.5 to 1 %, accompanied by a marked drop in pig seroprevalence. More recently, an educational intervention was also tested in 42 villages (827 households) in Mbulu district, Tanzania. It resulted in improved knowledge of the disease without apparent changes in observed risk practices; however, the incidence of porcine cysticercosis was significantly reduced in the intervention arm (Ngowi et al. 2008). Wide efforts in China using health education, mass chemotherapy, and environment management are reported to have reduced porcine cysticercosis by 97 % and human taeniasis by 91 % along 6 years (Wu et al. 2012).
10.1.2 Treatment of Infected Pigs
Treatment of the human tapeworm carrier ignores the porcine reservoir. Mathematical modeling strongly suggests that driving disease transmission to an extinction state would require multiple sustained interventions in the human population, for a very long time (Gonzalez 1997). Treatment of infected pigs would avoid future taeniasis infections and increase the likelihood of elimination. Flisser and coworkers (1990a, b) demonstrated the efficacy of praziquantel to eliminate muscle cysts in pigs. Tellez-Giron et al. (1981) reported 100 % efficacy with 10 days of flubendazole, Gonzalez et al. (1995) reported good efficacy of multiple doses of albendazole, and Peniche-Cardenas et al. (2002) reported similar effects using 8 days of intramuscular albendazole sulfoxide. Most of these regimes require multiple doses which is highly impractical in field conditions. Oxfendazole, given as a single oral dose of 30 mg/kg, is close to 100 % effective to destroy muscle cysts; it is not expensive and has no major side effects. Death of cysts usually occurs along a few weeks. The withdrawal period of oxfendazole at this dose was calculated to be 17 days. Oxfendazole treatment of pigs at 4 and 9 months of age as a single control measure was tested in two groups of 54 pigs each and 108 controls in Mozambique; significant decreases in pig infection were found, more marked when pigs were treated at 9 months of age (Pondja et al. 2012).
Combined human and pig treatment was applied by the Cysticercosis Working Group in Peru in ten villages in Huancayo, at the Peruvian central highlands. The effect was measured in pig seroincidence and demonstrated a significant decrease followed by a rapid return to levels close to baseline, quick after the intervention was stopped (García et al. 2006).
10.1.3 Pig Vaccination
A third major pathway of intervention is the immunization of the porcine population. Molinari et al. (1993) performed a large trial of pig immunization using a crude cysticercal extract in 3,295 animals, a proportion of which were vaccinated twice. No infected animals were reported 1 year after the intervention, although it is unclear how infection status of pigs was determined. Others have demonstrated diverse degrees of protection using antigens from the scolex, the entire cyst, or from the oncosphere (Nascimento et al. 1995; Plancarte et al. 1999; Verastegui et al. 2002). Improved pig vaccines were later developed. TSOL18 is an oncospheral recombinant protein identified and developed in Australia. Two doses of TSOL18 vaccine provide almost absolute protection to further infection (Flisser et al. 2004; Gonzalez et al. 2005), a protection that was later confirmed in field trials (Assana et al. 2010). A vaccine using synthetic peptides of Taenia crassiceps antigens has been produced and applied in Mexico. This vaccine seems to obtain reductions of up to 98.7 % in the numbers of parasites but only 53 % in the proportion of infected pigs (Huerta et al. 2002). DNA vaccines have also been shown to provide diverse degrees of protection, up to 93 % (Guo et al. 2004, 2007; Wang et al. 2003).
10.1.4 Other Potential Points of Intervention
A time-honored form to detect infected pigs in the field is to examine their tongues to see cysticerci nodules (Fig. 10.1). However, not all infected animals have a positive tongue examination. Inspection of carcasses at slaughterhouses is also a classic standard in control which, nevertheless, has limited value to detect porcine cysticercosis in endemic regions. First of all, most cysticercosis infected pigs will have less than five or ten cysts in the entire carcass, so opening the carcass and performing a few cuts in the larger muscles is likely insufficient to detect these few cysts. Furthermore, peasants will be reluctant to bring animals at risk of being infected with cysticercosis because of wrong confiscation policies with no reimbursement to the affected peasant (Cysticercosis Working Group in Peru 1993; Gemmell et al. 1983).
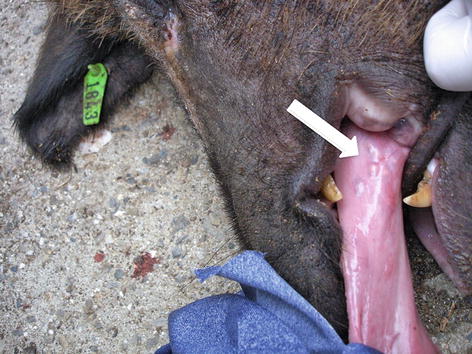

Fig. 10.1
Tongue of infected pig showing nodules of cysticerci (arrow)
As noted, a major problem in rural villages of developing countries is the fact that pigs are allowed to roam free and the close relationship that they have with humans (Fig. 10.2). So, pigs get access to human feces in places with poor sewage disposal and may become infected. Confinement of pigs (pig corralling) is a logical measure (Fig. 10.3). It however collides with the requirement of investment from the farmer to feed corralled pigs, something not required if pigs are left to roam and pasture. In survival economies, farmers will be unlikely to comply with sustained corralling because of these economic reasons (Cysticercosis Working Group in Peru 1993; García et al. 2007a). Anecdotally, sometimes open latrines are placed in the backyard where pigs are corralled, allowing the cycle to complete.



Fig. 10.2
Pig and man living together. This close relationship is the main responsible for the high endemicity of cysticercosis in rural villages of developing countries (Reproduced with permission from Pathogens & Global Health. Neurocysticercosis special issue, August 2012, cover photo, Oscar H. Del Brutto and Hector H. García, Guest Editors)

Fig. 10.3
Pig corralling in a rural village of coastal Ecuador (Atahualpa). This simple measure prevents pigs to be in contact with human feces and may help to interrupt the life cycle of Taenia solium
Sotelo et al. (1986) reported in 1986 that freezing of infected pork will kill cysticerci in a day at −24 °C or in 4 days at −5 °C. Meat processing by gamma radiation at high doses inhibits the ability of cysticerci to evaginate. Cysticerci irradiated at lower doses may evaginate, but when they are fed to hamsters as a tapeworm infection model, they seem to have an impaired development in the hamster gut (Verster et al. 1976).
Vaccinating the human host to prevent intestinal taeniasis infection has been proposed as a potential control measure (Flisser et al. 2010; Leon-Cabrera et al. 2009), and early experimental models in rodents seem to show a degree of protection induced by Taenia solium calreticulin (Flisser et al. 2010). So far, no attempts have been made to produce a human cysticercosis vaccine.
10.2 Current Status of Taenia solium Control
In an interesting trial in Cameroon, Assana et al. (2010) used two rounds of porcine TSOL18 vaccination at 3, 4, and 7 months of age plus oxfendazole treatment at the time of the second immunization and found no infected animals in the intervention group 5 months after the last immunization, compared to 20 % in controls; this trial did not intervene in the human population. In Tumbes, Peru, a large-scale control program has been implemented using repeated rounds of human mass treatment with niclosamide, pig treatment with oxfendazole, and pig vaccination with the TSOL18 vaccine, with apparently focal elimination of transmission (Mahanty and García 2010). In the Tumbes program, coproantigen detection was widely applied to detect treatment failures after mass treatment. In the settings of poorly effective regimes (effectiveness of 5 mg/kg of praziquantel in mass campaigns has been reported to be as low as 67 %), a field-applicable coproantigen test could complement treatment efficacy. Coproantigen levels become negative as soon as a few days after successful taeniasis treatment (Bustos et al. 2012).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







