Chapter 172 Degenerative Disease and Infection
Role of Imaging
Degenerative Disease
The term degeneration is commonly applied to a variety of pathologic and imaging manifestations of spine morphology, reflecting the complexity of the process and gaps in our current knowledge. The distribution of axial load is responsible for the typical locations of spine degeneration. C5-6 and C6-7 levels are involved in most cases, because they are the sites of lordosis reversal. The thoracic spine, because of its lesser mobility, is less often affected by degeneration. L4-5 and L5-S1 are the most commonly affected levels because they are the sites of the highest dynamic and static loads.1,2 Because degenerative spine sequelae are among the leading causes of functional incapacity and chronic disability, further research in the epidemiology, pathology, imaging, biomechanics, and therapeutics is warranted.
Low Back Pain
The natural history of low back pain (LBP) with or without radiculopathy is not clearly understood. Traditional practice suggests that imaging has no role in evaluating back pain unless associated signs and symptoms are suggestive of a potentially treatable condition related to trauma, instability, infection, or malignancy. Foregoing imaging in these circumstances may not seriously affect the outcome in most patients and, in fact, may improve conservative management by avoiding the detection of confounding altered morphology. Because back pain is typically a recurrent problem, physicians and patients should benefit from information related to patient group stratification, prognosis for recovery from acute episodes, and likelihood of recurrence.3
Low Back Pain with Radiculopathy
Patients with LBP with radiculopathy constitute a relatively less diverse group in terms of etiology, and the symptoms usually suggest nerve root compression. Multiple authors4–6 suggest that an imaging study is indicated for the evaluation of back pain and associated sciatica when (1) true radicular symptoms are present; (2) there is evidence of nerve root irritation on physical examination (i.e., positive straight-leg raise test); and/or (3) “conservative management” of 4 to 6 weeks’ duration has failed. Earlier imaging is considered appropriate if clinical features raise concern regarding malignancy or infectious pathology or if neurologic findings worsen during observation. These recommendations are based on several studies of the successful nonoperative treatment of sciatica.7–10 Thus, imaging is recommended only for the remaining minority of patients with persistent signs and symptoms who are felt to be surgical candidates or in whom diagnostic uncertainty remains.
Role of Early Imaging
The use of diagnostic imaging earlier in the course of a patient’s symptoms is supported by the following evidence: (1) patients with herniated discs treated surgically have better short-term outcomes than those treated conservatively10,11; (2) the earlier the surgery the better the outcome12; and (3) surgery is cost-effective compared with conservative management (though this varies with the type of surgery).13,14 However, the potential reduction in morbidity from early intervention requires the identification of accurate prognostic indicators, be they clinical or imaging based. Certainly, more than morphology is a factor. As has been suggested by Postacchini,15 two needs likely require consideration: (1) protracted conservative treatment may lengthen the time off from work and reduce the chances of successful surgical treatment; and (2) surgery should be avoided in patients with a herniated disc that may become asymptomatic or even disappear within a few months of onset. Thus, the possible benefits of imaging for degenerative diseases of the spine, other than as a presurgical tool, have never been carefully documented and quantified. Predicting which patients will benefit from imaging is a complicated and controversial subject. Its use as a preintervention planning tool is critical, but its employment for diagnostic information per se is less well accepted and the subject of much debate. Nevertheless, if imaging could be employed to prospectively determine which patients will not do well with surgery or other types of therapy, it would be of great value. Conversely, its use to identify patients undergoing prolonged conservative treatment who require more aggressive therapy (e.g., surgery) might be equally beneficial. This would save the cost of lost work, medical expenses, and personal discomfort. Clearly, there is little consensus, either within or among specialties, on the use of diagnostic tests for patients with back pain.16 The diagnostic evaluation depends heavily on individual physicians, their specialties, and patient socioeconomics, in addition to the patient’s symptoms.16,17 On the other hand, patients with underlying risk factors (“red flags”), mainly, infection, trauma, and malignancy, warrant early imaging for timely intervention.
Counting Reference
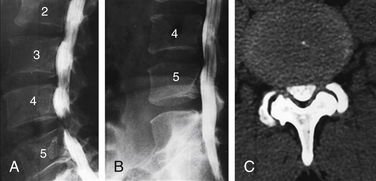
The topic is discussed in detail in Chapter 175. It is extremely important that counting reference is clearly stated in the spine imaging report. This is to avoid wrong level surgeries with potential medico-legal implications. The two major factors responsible for discrepancy are (1) a transitional vertebra, which can be a sacralized L5 or lumbarized S1, and (2) variations in the number of rib-bearing vertebrae. Lumbosacral transitional vertebra (LSTV) is a common finding in the general population. Identification of LSTV on imaging is the essential first step in the interpretation of lumbar and thoracic imaging.18 The coronal MRI localizer image of the thoracic and lumbar spine can be useful because L4-5 is generally at the level of the iliac crest. Some prefer to use a vitamin E back-skin marker at an overlapping level in the thoracic spine, which, however, requires imaging of the entire spine. The iliolumbar ligament is easily identified on axial MRI and can be used as a marker of L5.19 There is a greater tendency of counting the lowest lumbar-type intervertebral disc space as L5-S1. Nevertheless, it is crucial that these approaches and variations be specified and clearly communicated in the report.
Plain Radiography
Plain radiography (PR) is still a very useful and cost-effective diagnostic tool by virtue of its availability and efficiency.20 PR is generally used in trauma, for screening of degenerative disease, and for evaluation of sagittal translation using flexion-extension views. PR can aid MRI in characterization of disc disease by better demonstration of vacuum phenomena, disc calcification, bridging osteophytosis, and dorsal longitudinal calcification (though less common in the thoracolumbar spine). Conventional screen-film imaging has been nearly completely replaced by digital acquisition modalities, using either imaging plates (computed radiography, CR) or flat-panel detectors (direct radiography, DR).
Routine Magnetic Resonance Imaging
Unlike CT and conventional radiography, which depend on information related to electron density, proton MR signals are influenced by the T1 and T2 relaxation time, proton density, and motion (both macro and micro) to provide greater tissue contrast. Thus, its role may go beyond gross anatomic appraisal to actual tissue characterization of pathology and biochemical change. The routine sequences and their significance are as follows: (1) sagittal T1 to evaluate bone marrow infiltration and end-plate bone marrow degenerative changes; (2) sagittal T2 to evaluate disc and end-plate degeneration, thecal sac extrinsic impression, the conus medullaris, and the cauda equina morphology; and (3) sagittal STIR to detect bone marrow and paraspinal soft tissue edema. Axial T1 and T2 basically confirm the findings on sagittal imaging and provide a better assessment of central canal/thecal sac narrowing, cauda equina abnormalities, facet disease, and lateral disc herniations. The major degrading factors are patients’ gross motion, positioning, body habitus, and instrumentation.
Magnetic Resonance Myelography
MR myelography (MRM) is currently not practiced routinely in the United States for two major reasons: (1) Intrathecal gadolinium is not yet approved by the FDA, and (2) MRM has no significant added advantage compared with a CT myelogram or conventional MRI.21,22 MRM is, however, comparably accurate in detecting CSF leaks in patients with intracranial hypotension compared with radioisotope cisternography.23
Open and Large-Bore Magnetic Resonance Imaging
The open-design MRI systems enable imaging of those who are claustrophobic and overweight and offer the potential of dynamic imaging. The major penalty is some compromise of the image quality due to decreased signal-to-noise ratio. However, the diagnostic information is satisfactory in most cases.24,25 Closed high-magnetic-field scanners with short magnets and wide-bore tubes offer patients improved comfort and lessen anxiety.26
Dynamic Imaging and Upright Magnetic Resonance Imaging
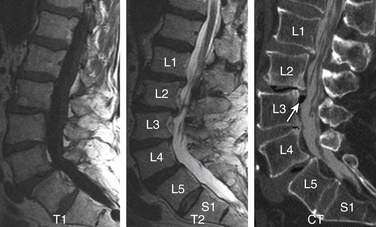
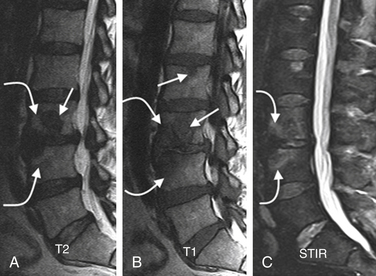
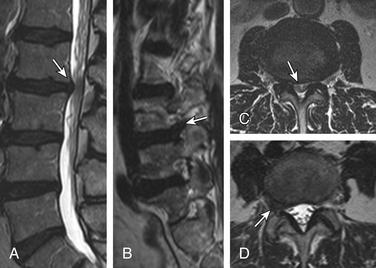
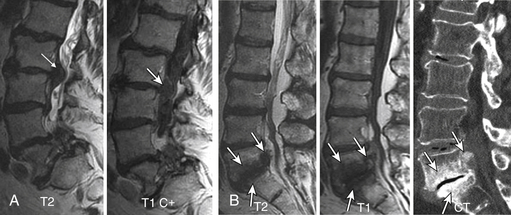
Sagittal segmental lumbar motion is traditionally assessed with lateral flexion-extension radiographs. These dynamic studies often demonstrate a decrease in the slip percentage between the vertebral segments with extension and an increase with forward flexion. Ironically, the degree of spondylolisthesis can actually improve on the lateral film taken on the operating table in an anesthetized patient.27 Somewhat analogous to the flexion-extension imaging, supine-prone imaging of the lumbar spine can also be more sensitive by showing a higher degree of listhesis in the prone position.28 Position-dependent stenosis can be demonstrated on the lateral myelogram in flexion-extension postures, which would otherwise have been underestimated on the follow-up CT myelogram29 (Fig. 172-1). Ben-Galim and Reitman in a study of a small group of patients showed position-dependent severe spinal canal stenosis on CT myelography with grade I or II spondylolisthesis. Additionally, they showed fluid-filled distended facet joints, a marker of positional translation.30 Imaging findings on supine MRI may correlate poorly with clinical findings. This may be related to the positional dependence of spinal stenosis, which reflects dynamic changes in soft tissue structures. Therefore, it is potentially valuable to evaluate spinal disorders under mechanical loading. Hydraulic axial-loading devices have also been tried.31 Upright MRI in different postures can reproduce the positions that bring about their symptoms and may unmask findings that are not visible with routine supine imaging. Small disc herniations and ventral spondylolisthesis can be unmasked, and disc herniations can be increased in size on upright MRI compared with recumbent MRI.32 Assessment of the degree of spinal stability in the degenerative and postoperative lumbar spine can also be performed.33,34 Kinetic magnetic resonance images (kMRIs) in axially loaded, upright-neutral, flexion-extension positions can also accentuate the size of the disc herniations and play a potential problem-solving role in radiculopathy with negative supine static imaging.35 Clearly, axial loading changes anatomic relations. The significance of the findings in terms of therapeutic decision making is not very clear.36
Morphologic Alterations on Imaging
Standardized reporting is a better means of communication and consistent reporting, as well as a useful source of database research. Given the complexity, inconsistency, and overlapping of terminology used to discuss the spine over the years, standardized report generation in spine imaging is needed the most to describe the morphologic alterations in the spine. A consensus-based terminology recommended by Millette et al.37 is used in this chapter. It must be clearly understood that these terms are descriptive only and, in and of themselves, independent of the test. It cannot be overemphasized that these terms do not imply knowledge of etiology, symptoms, prognosis, or need for treatment. Recently, Carrinno et al.38 showed moderate reliability in the interpretation of qualitative nondisc contour degenerative findings on lumbar MRI.
Disc Degeneration
The term disc degeneration is used for a variety of changes, including any or all of the following: real or apparent desiccation, fibrosis, narrowing of the disc space, bulging, fissuring or mucinous degeneration of the anulus, osteophytes of the vertebral apophyses, and end-plate/adjacent marrow changes. MRI and CT show disc space narrowing, T2-weighted signal intensity loss from the intervertebral disc, presence of fissures, fluid, vacuum changes and calcification within the intervertebral disc, ligamentous signal changes, marrow signal changes, and osteophytosis. These alterations result in disc herniation, malalignment, and spinal canal stenosis. Conventional theory suggests that aging and degeneration are very similar processes, albeit occurring at different rates. Historically, the two processes have been characterized morphologically and termed as spondylosis deformans and spondylosis osteochondrosis (deteriorated disc), respectively. The former affects the anulus fibrosus and adjacent apophyses, whereas the latter mainly affects the nucleus pulposus and the vertebral body end plates, particularly fissuring (numerous tears) of the anulus fibrosus. Scientific evidence suggests that spondylosis deformans is the consequence of normal aging, whereas intervertebral osteochondrosis represents a pathologic, though not necessarily symptomatic, process.39 Anteriolateral osteophytosis is a reliable marker of spondylosis deformans, with a 100% incidence above 40 years.39 Disc degeneration and aging are multifactorial processes that encompass a wide spectrum of changes and sequelae, of which the radial tear is but one. Disc degeneration may be explained primarily by genetic influences and complex unpredictable interactions of unidentified factors.
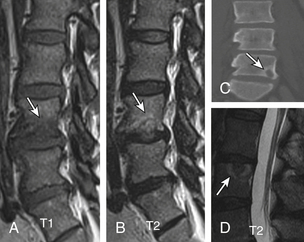
The nucleus pulposus is eccentrically located and more closely related to the dorsal surface of the intervertebral disc. With degeneration and aging, type II collagen increases outwardly in the anulus, and water loss from the nucleus pulposus is greater than from the anulus. This results in a loss of the hydrostatic properties of the disc, with an overall reduction of hydration in both areas to about 70%. In addition to water and collagen, the other important biochemical constituents of the intervertebral disc are the proteoglycans. It is not the individual composition but the relative composition of these constituents that in turn affects the hydrostatic properties and tensile strength of the intervertebral disc. The ratio of keratin sulfate to chondroitin sulfate increases. The disc becomes progressively more fibrous and disorganized, with the end stage represented by amorphous fibrocartilage and no clear distinction between nucleus and anulus. The degenerating disc manifests as diminishing T2 signal and loss of internal lamellation. T2 measurements also correlate with glycoaminoglycan concentration rather than absolute water content. So, it is not just the absolute concentration of water but also the state of water involved in the disc degeneration. The status of the proteoglycans is thus a major determinant of disc space T2 and T1 signal39–41 (Fig. 172-2).
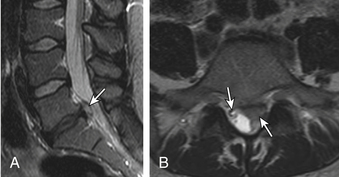
Anular Tears
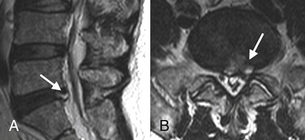
Also called anular fissures, anular tears are separations between anular fibers, avulsion of fibers from their vertebral body insertions, or breaks through fibers that extend radially, transversely, or concentrically and involve one or many layers of the anular lamellae. The term tear, or fissure, describes the spectrum of such lesions and does not imply that the lesion is a consequence of trauma. Although it has certainly been verified that anular disruption is a sequela of degeneration and is often associated with it, its role as the causal agent of disc degeneration has certainly not been proved. Anular disruption initiates a reparative process as it is replaced by dense fibrous tissue and cystic spaces.42–45 Fissuring, chondrocyte generation, and granulation tissue formation have been noted within the end plate, anulus fibrosus, and nucleus pulposus of degenerative discs, indicating attempts at healing.39 Anular tears are commonly seen in the dorsal bulging discs as focal T2 hyperintensity on T2 and STIR imaging; there may be associated small disc protrusion. Anular tears consistently show focal contrast enhancement46 (Fig. 172-3).
Disc Gas “Vacuum Phenomenon”
Radiolucent foci on CT or plain films representing gas, principally nitrogen, occur at sites of negative pressure produced by the abnormal spaces. The vacuum phenomenon within a degenerated disc is represented on spin echo images as areas of signal void. Whereas the presence of gas within the disc is usually suggestive of degenerative disease, spinal infection may (rarely) be accompanied by intradiscal or intraosseous gas.47 A protruding disc with a vacuum phenomenon can present as epidural gas foci on CT and hypointensities on MRI (see Fig. 172-2).
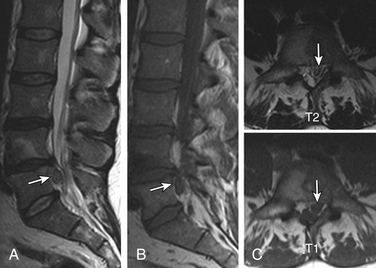
Disc Calcification and T1 Hyperintensity
Patchy, partial, or complete calcification of the disc space can be seen either as an isolated finding without degenerative stigmata or as a chronic dystrophic change in a degenerated disc. The clinical significance of a calcified disc is unknown. The disc space on CT and plain films can appears as fused, perhaps “autofused.” Calcification, depending on its biochemical status, can appear as hyperintense or hypointense on sagittal T1 imaging. Furthermore, the hyperintense T1 disc may represent an ossified disc, as can be proven by loss of signal on fat-suppressed MRIs due to lipid marrow. A calcified disc is easily demonstrable on CT (Fig. 172-4).
Degenerative Marrow Changes
The anulus fibrosus, vertebral end plate, and vertebral body have an interreactive relationship in the degenerative process, as is also evident from the model of chymopapain-treated discs.48,49 Paradiscal MR signal changes in the end plate and vertebral bodies take three forms.
Type I Changes
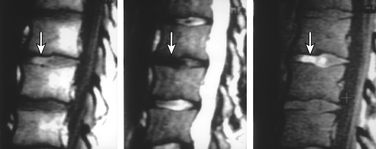
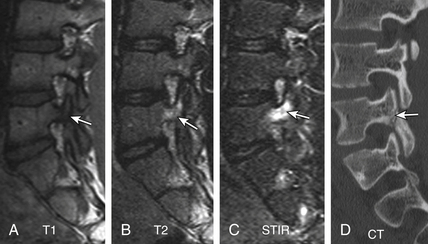
Type I changes demonstrate decreased signal intensity on T1-weighted images and increased signal intensity on T2-weighted images. Type I change is found in about 4% of nonsurgical lumbar spines, 8% in postdiscectomy discs, and 40% to 50% of chymopapain-treated discs.48–50 The last group can be used as a model of acute disc degeneration. Histopathology of type I changes demonstrates disruption and fissuring of the end plate and vascularized fibrous tissues within the adjacent marrow, accounting for prolonged T1 and T2 MR signal. Enhancement of type I vertebral body marrow changes is seen with administration of IV gadolinium. The enhancement may involve a portion of the disc itself and is presumably related to the vascularized fibrous change. The type I signal is expectedly accentuated on STIR, mimicking bone marrow edema, and may be very difficult in some cases to differentiate from discitis osteomyelitis (discussed later under the differential diagnosis of vertebral osteomyelitis; Fig. 172-5).
Type II Changes
Type II changes are represented by increased signal intensity on T1-weighted images and isointense or slightly hyperintense signal on T2-weighted images. Type II changes are identified in approximately 16% of patients on MRI.48–50 Discs with type II changes, like type I, show evidence of end-plate disruption, with fat marrow in the end plate and adjacent vertebral body resulting in T1 hyperintensity due to T1 shortening (Fig. 172-6).
Type III Changes
Type III changes are represented by a decreased signal intensity on both T1- and T2-weighted images due to extensive bony sclerosis, as evident on plain radiographs and CT. The first two types show no definite correlation with sclerosis seen at radiography, which is not surprising when one considers the histology. The sclerosis on plain radiographs is a reflection of dense woven bone within the vertebral body rather than of the marrow elements. The lack of signal in the type III change represents the relative absence of marrow in areas of advanced sclerosis (see Fig. 172-17B).
Facets, Pedicles, and Ligamentous Changes
The superior articulating process of one vertebra articulates with the inferior articulating process of the vertebra above by a synovium-lined articulation—the zygapophyseal joint. The lumbar facet joints are predisposed to arthropathy with alterations of the articular cartilage. Facet joint degeneration results in hypertrophic changes (osteophytosis), joint effusions, synovial cyst formation, and rostrocaudal subluxation. Mild to moderate facet joint osteophytosis is commonly seen in the lumbar spine at L4-5 and L5-S1 and is more suggestive of age-related change.51,52 The important ligaments of the spine include the anterior longitudinal ligament, the posterior longitudinal ligament, the paired sets of ligamenta flava (connecting the laminae of adjacent vertebrae), the intertransverse ligaments (extending between transverse processes), and the unpaired supraspinous ligament (along the tips of the spinous processes). As these ligaments normally provide stability, any alteration in the vertebral articulations can lead to ligamentous laxity with subsequent deterioration. Loss of elastic tissue, calcification and ossification, and development of bone proliferation at sites of ligamentous attachment to bone are recognized manifestations of such degeneration.
Facet and Disc Degeneration
Generally, disc degeneration dominates facet arthropathy. Some degree of facet arthritic changes usually accompanies disc degeneration. Is facet disease a sequela of disc degeneration? The answer to this question is debatable, but at least it is clear that disc degeneration is more common and generally precedes facet joint arthropathy. Due to altered mechanics, disc degeneration probably accelerates facet joint arthropathy.51–54 A systemic component is hypothesized to account for facet arthrosis since there is a high incidence of concurrent lumbar and cervical facet arthrosis.55 Osteophytosis of the facet joints is, to a degree, a counterpart of end-plate osteophytosis and specifically contributes to lateral recess and foraminal stenosis. Facet arthrosis may occur independently and be a source of symptoms on its own.56 A smaller group of lumbar spines in our experience show dominant facet and ligamentous degenerative hypertrophic changes with absent to minimal degenerative disc changes.
Facet Joint Effusions
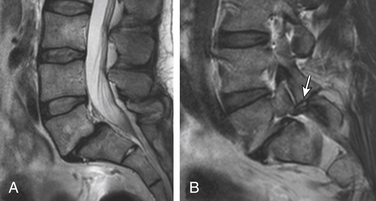
Pencil-thin homogeneous bilateral facet joint effusion is a common and normal finding in the lower lumbar spine. In an arthritic facet joint, synovial villi may become entrapped within the joint, with resulting joint effusions. STIR is the best method for demonstrating joint effusions. Large joint effusions (>1.5 mm) are highly predictive of degenerative spondylolisthesis on dynamic imaging, particularly at L4-557 (Fig. 172-7).
Synovial Cysts
Herniation of the synovium through the facet joint capsule may result in synovial cysts. In a review of patients with degenerative facet disease, synovial cysts occurred at a ventral or intraspinal location in 2.3% of cases and dorsal or extraspinal location in 7.3%.58 There is a more straightforward relationship of synovial cysts with osteoarthritis and the instability of the facet joints than degeneration of the intervertebral disc alone.57 Intraspinal synovial cysts can be symptomatic, depending on their size. Typically, synovial cysts have T2 hyperintense cystic appearance, but signal intensity can vary due to debris, hemorrhage, gas, and superimposed infection. The cyst may exhibit rim enhancement (see Fig. 172-24). Distinguishing the synovial disc from the herniated disc is not usually difficult due to its location and MR characteristics. A dorsolateral extradural impression and associated juxtafacet asymmetrical soft tissue density on CT myelogram is a nonspecific finding; asymmetrical facet joint arthritis can be helpful in such cases.
Dorsal Elements and Paraspinal Edema
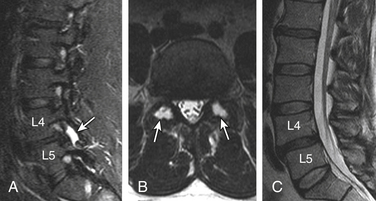
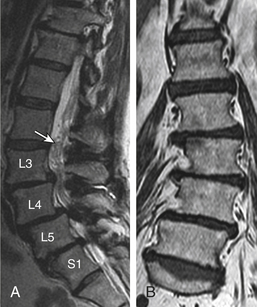
Routine use of STIR and similar sequences has resulted in increased detection of dorsal elements and paraspinal soft tissue edema.59,60 Facet joint degenerative arthritis can result in para-articular and pedicular edema, joint effusion, and para-articular soft tissue edema. In patients at high risk, it may be difficult to differentiate from infected facet joints. The signal changes and sclerosis in the pedicles can be seen secondary to facet joint arthritis and underlying pedicle or pars fractures. The greatest mechanical stress is on the pars interarticularis, followed by pedicles as shown by Sairyo et al.61 These stresses can result in a spectrum of reactive bone marrow changes in the pars and pedicles. T2 hyperintense signal in the pedicles has been shown as an early marker of spondylolysis.61 There is evidence that categories of changes in MR signal intensity, similar to those described adjacent to degenerating discs, can be seen in lumbar pedicles adjacent to a spondylolytic defect of the pars interarticularis62 (Figs. 172-8 and 172-9).
Morphologic and Functional Sequelae
Segmental Instability
Each level in the thoracolumbar spine consists of three joints: intervertebral disc space and bilateral facet joints. Degeneration of these joints results in segmental instability replacing the normal spinal motion, which can be irregular, excessive, restricted, translational, and/or angular. Segmental instability, which can cause static and dynamic stenosis, is considered a cause of LBP but is poorly defined.63 On flexion-extension plain radiography, generally about 3 mm or more of sagittal translation is equated with instability. Spondylolisthesis is a term used in static imaging and refers to intervertebral displacement—anterolisthesis, retrolisthesis, or lateral listhesis. The different types, based on etiology, include congenital, degenerative, isthmic, iatrogenic, pathologic, and traumatic (Fig. 172-10).
Grading of Spondylolisthesis
Spondylolisthesis in the sagittal plane can be graded by one of two methods, independent of the imaging modality and type of spondylolisthesis.64 The first is the method described by Meyerding.65 The anteroposterior (AP) diameter of the superior surface of the lower vertebral body is divided into quarters, and a grade of I to IV is assigned to slips of one, two, three, or four quarters of the superior vertebra, respectively. The second method, first described by Taillard,66 expresses the degree of slip as a percentage of the AP diameter of the top of the lower vertebra. Complete slip of L5 on S1 is termed spondyloptosis. The second method is favored by most authors as it is more accurately reproducible.67 Measurement of the slip and its apparent progression, however, should be viewed with caution. Studies have shown that inter- and intraobserver error of up to 15% is possible. This variation can increase if an element of rotation is present. Therefore, only a progression of greater than 20% slip can be reliably assessed.68,69
Degenerative Spondylolisthesis
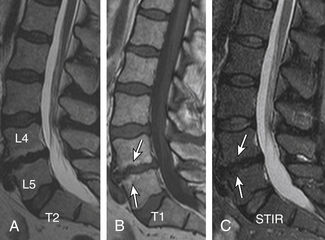
Degenerative spondylolisthesis (DS), which is usually seen with an intact pars interarticularis, is related to degenerative changes of the apophyseal joints and/or intervertebral discs and is most common at the L4-5 and L5-S1 vertebral levels. Lumbar DS is a major cause of spinal canal stenosis and is often related to low back and leg pain.70 Long-standing DS is almost invariably accompanied by disc and facet degenerative changes, which obscure the origin of the process. Discogenic DS and facetic DS cause secondary stress and stretch on the facet joints and disc spaces, respectively. The degree of sagittal listhesis is generally mild in DS relative to other types, particularly isthmic spondylolisthesis. There may be associated degenerative scoliosis and listhesis in nonsagittal planes. Type I end-plate changes, which may be more extensive and quite prominent on a STIR sequence, even without spondylolisthesis on static imaging, may represent segmental instability71 and can mimic discitis osteomyelitis, as discussed later (Fig. 172-11).
Pseudarthrosis
Nonunion of Bone Fusion
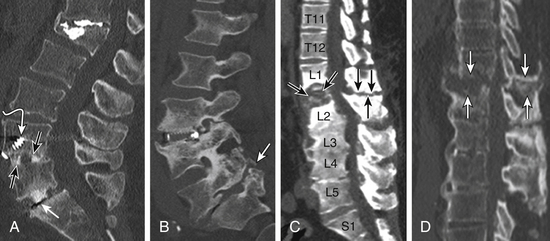
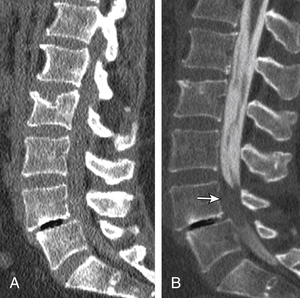
Nonunion is the result of failed spinal fusion. The failed fusion typically manifests with axial or radicular pain months to years after surgery. The common causes of this complication are inadequate surgical technique, excessive stresses across the fusion site, insufficient internal or external stabilization, and unrecognized metabolic abnormalities.72 Diagnosis is based on clinical presentation and imaging studies, after other causes of persistent pain are excluded.73 The degree of fusion and associated motion seen on flexion-extension radiographs that is indicative of solid or failed fusion remains a point of controversy due to significant interobserver variations.74 The choice of bone graft material can affect the radiographic assessment of interbody fusion. CT has higher contrast resolution and is therefore better than radiography for assessment of bone fusion. One of the main reasons for CT’s superiority is that premineralized osteoid may be functionally fused but radiographically lucent, therefore leading to underestimation on plain radiography. Bony arthrodesis is usually evident by 6 months, with evidence of bridging trabecular bone. Bridging bone is usually seen lateral to the implant and may also be noted within the implant itself. Mature solid fusion is generally present at between 12 and 24 months75,76 (Fig. 172-12).
Disc Space Failed Fusion
Failed fusion at the disc space manifests as lucency at the device margins or lucent lines through the fusion mass by about 18 to 24 months. Lucency around the fusion devices (e.g., cage or pedicular screws) represents abnormal motion and predicts delayed or failed fusion. Cystic change at the end plates adjacent to the implant is a reliable marker of failed fusion. Other signs include malpositioning and subsidence of the fusion devices74–76 (see Fig. 172-12).
Posterolateral Failed Fusion
Posterolateral failed fusion is divided into three types: Type I shows atrophy and resorption of the grafted bone; type II shows a lack in trabecular continuity of the fusion mass; and type III shows a gap either cranially or caudally between the fusion mass and lumbar matrix (which includes the transverse process and superior facet). CT is better than PR for diagnosing all types of posterolateral failed fusion, particularly type III77 (see Fig. 172-12).
Transitional or Segmental Pseudarthrosis
Segmental pseudarthrosis is usually secondary to transition of the mechanical loading to the adjacent level above the surgical fusion or fusion due to ankylosing spondyloarthropathies and advanced degenerative processes. Neuropathic spine (Charcot spine) can also result in segmental pseudarthrosis. The typical CT imaging pattern is lucency through the ventral and dorsal pillars in a transverse plane with or without associated spondylolisthesis. MRI shows corresponding hyperintense T2 (fluid) signal in the transverse plane defects and variable end-plate signal changes (see Fig. 172-12).
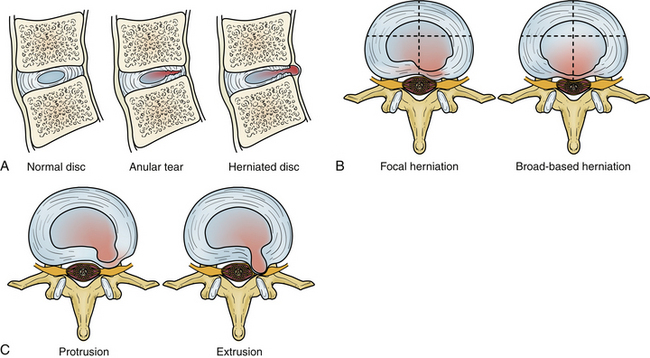
Intervertebral Disc Displacement (Disc Herniation)
Degeneration allows the disc to displace focally or diffusely in any direction beyond the end-plate margins of the disc space. The anulus fibrosus in a displaced disc tries to contain or limit the herniating nucleus pulposus. A variety of terms are used to describe disc displacement. See Figure 172-13 for diagrammatic representation of disc displacements.
Anular Disc Bulge
An anular bulge is a generalized extension, greater than 50% of the circumference of the disc tissues to a short distance (<3 mm) beyond the edges of the apophyses. A bulge is not a herniation; it is generally diffuse and may be asymmetrical. The latter usually occurs when associated lateral listhesis and scoliosis are present. The nucleus pulposus is contained by the anulus fibrosus. Grading of the disc bulging is subjective. Associated end-plate hypertrophy (spondylosis) may be present but is less prominent than in the cervical spine. Disc bulging may show crescentic calcification along its dorsal margin on sagittal CT (see Fig. 172-11A).
Protrusions
Protrusions are disc displacements in which the greatest distance in any plane between the edges of disc material beyond the disc space is less than the distance between the edges of the base in the same plane (neck is broader than dome). Protrusion is still contained by the anulus fibrosus fibers and therefore is not a true herniation. There may be an associated anular tear, which is usually evident on MRI as T2 hyperintensity, typically in the dorsal or dorsolateral disc protrusions. If the protrusion is less than 25% of the disc circumference, it is referred to as focal, and if between 25% and 50%, it is referred to as broad-based (Fig. 172-14).
Extrusions
Extrusions are disc displacements that are true herniations in which in at least one plane, any one distance between the edges of the disc material beyond the disc space, is greater than the distance between the edges of the base in the same plane (dome is broader than neck). Extrusions usually extend above or below into the suprapedicular or infrapedicular zone, resulting in further narrowing of the neck but still abutting the native disc. The signal intensity of the extruded portion may be increased on T2-weighted images. There may be associated curvilinear areas of decreased signal intensity on the T2-weighted images, which are related to torn portions of the anulus and posterior longitudinal ligament (Fig. 172-15).
Sequestration
Sequestration is displaced disc material that has completely lost any continuity with the parent disc. The extrusion is referred to as “migrated” if it has displaced from the site of extrusion, regardless of whether it is sequestrated. Sequestrated fragments can lie ventral to the posterior longitudinal ligament (especially if they have migrated behind the vertebral bodies where the posterior longitudinal ligament is not in direct opposition), dorsal to the ligament, rarely in the intradural or intraradicular space, and even in the dorsal extradural space.78 Nevertheless, penetration almost invariably occurs through the posterior longitudinal ligament; either dorsal, where it is fused with the anulus, or rostrally or caudally, where it fuses with the vertebral body margin. In the majority of patients in whom sequestrated fragments migrate behind the vertebral body, the sequestration usually lateralizes, with disc material pushing across the midline and the leading edge being smoothly capped. It has been postulated that this shape is imposed by a midline septum in the ventral epidural space. This space is largest in the lower lumbar region and is delineated dorsally by the posterior longitudinal ligament and laterally attached membranes and ventrally by the vertebral body. It is divided into two compartments by a sagittally aligned septum. Sequestrated fragments within the lateral recess and the neural foramen have been shown to produce eroded cortical bone and expansion of those spaces and, thus, should be considered in the differential diagnosis of a mass arising and expanding the neural foramen and lateral recess. Intradural disc herniation is very rare, with few cases reported.79,80 It is most frequent in the lower lumbar spine. The mechanism is thought to be the development of chronic inflammation leading to adhesions between the dura mater and posterior longitudinal ligament. The appearance on axial MRI is described as a “hawk-beak sign.”81 As the herniated disc penetrates, the ligament extends through the dura instead of pushing it away. Other possible causes are congenital connections between the ligament and the dura or previous surgery (Fig. 172-16).
Imaging Characterization of a Disc Herniation
Hyperintense zone (HIZ) in a disc has been used in correlative studies of MR and provocative discography. This nonstandard term generally represents anular tears.82,83
Compression of the nerve root within its sleeve may not correlate with patient symptoms. A grading system by Pfirrmann et al.84 had significant reliability and surgical correlation:
Grade 0 (normal): No contact of disc material with the nerve root.
Grade 1 (contact): Visible contact of disc material with the nerve root and loss of the epidural fat layer between the two.
Grade 2 (deviation): The nerve root is displaced dorsally by disc material.
Grade 3 (compression): The nerve root is compressed between disc material and the wall of the spinal canal; it may appear flattened or be indistinguishable from disc material (see Fig. 172-15B).
A herniated disc may be densely calcified, which may not be easily predictable on preoperative MRI. An isointense to hyperintense T1 signal can be helpful in such cases. CT clearly demonstrates calcification of disc herniation and ossification of the posterior longitudinal ligament (Fig. 172-17).
Intravertebral Disc Herniations (Also Known as Schmorl Nodes)
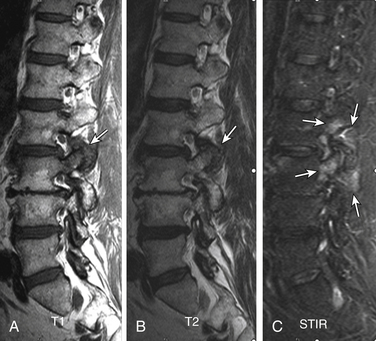
Nonacute Schmorl node (SN) intrabody herniations are common spinal abnormalities regarded as incidental observations. They have been reported in 38% to 75% of the population.85,86 Most intrabody herniations probably form after axial loading trauma, with extrusion of nuclear material through the vertebral end plate, resulting in reactive bone marrow edema. Another theory is that Schmorl nodes are the end result of ischemic necrosis beneath the cartilaginous end plate, with secondary herniation into the body of the vertebra.87 SNs are mostly well defined and have an isointense intrinsic T1/T2 signal to the disc.88 SNs have been traced to episodes of significant, sudden-onset, localized, nonradiating back pain and tenderness. Wagner et al.89 showed in symptomatic patients that 57% of cases on MRI had SNs surrounded by vertebral body marrow edema. The remaining nodes (43%) were not immediately apparent as SNs and manifested only as vertebral body edema, suggesting end-plate fracture, but did evolve into classical chronic SNs as revealed on follow-up imaging.89 Contrast enhancement demonstrated vascularized SNs, with a higher incidence of surrounding edema. They are more common in symptomatic patients.90,91 A concentric hypointense ring appearance has a high negative predictive value for the absence of underlying fracture, infection, or malignancy.88 Giant cystic SNs have been described by Hauger et al.92 as a clinicoimaging entity in a small series of six cases (Fig. 172-18; and see also Fig. 172-5).
Spinal Canal and Foraminal Stenosis
Spinal stenosis refers to any type of narrowing of the spinal canal, nerve root canals, or intervertebral foramina.93 Two broad groups have been defined: (1) acquired (usually related to degenerative changes) and (2) congenital or developmental. Developmental stenosis can be exacerbated by superimposed acquired degenerative changes. In the acquired type, there has been no association between the severity of pain and the degree of stenosis. The most common symptoms are sensory disturbances in the legs, LBP, neurogenic claudication, weakness, and relief of pain by bending forward. Clinical stenosis is perhaps a better term than spinal stenosis (as diagnosed on imaging studies). The imaging changes are in general more severe than expected from the clinical findings.94 Although there does appear to be a correlation between cross-sectional area and midsagittal measurements in patients with symptomatic spinal stenosis, measurement of canal stenosis is not generally used.39
Acquired Lumbar Spinal Stenosis
Acquired lumbar spine stenosis (LSS) is typically a sequela of hypertrophic degenerative disc and facet/ligamentous changes that can be accentuated by static or dynamic degenerative DS. Anatomically, spinal stenosis is divided into central canal, lateral canal (subarticular and lateral recess), and foraminal stenosis. Subjective visual grading of the spinal stenosis is a routine practice in interpretation of the spine imaging. Quantitative evaluation by measuring the AP diameter or cross-sectional area of the central canal is used in research studies but is not commonly done in clinical practice.95 Recent Framingham study data used a cut-off AP diameter of 12 mm for relative LSS, and 10 mm for absolute LSS.96 There is tendency to subjectively grade central spinal canal stenosis as mild, moderate, and severe based on the effacement of the thecal sac due to a combination of anular disc bulging, end-plate osteophytosis, and dorsal ligamentum laxity. Nevertheless, spinal stenosis, even of a severe grade, is meaningless if it lacks a correlation to patient symptoms (Fig. 172-19).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree