The histopathological changes, particularly the development of NFTs, occur in a characteristic sequence in various regions of the brain. The first alterations appear in the transentorhinal cortex (Braak stages 1 and 2); later the limbic regions become involved (Braak stages 3 and 4). In the late stages of the disease, isocortical regions of the brain are also affected by neurofibrillary changes (Braak stages 5 and 6). Histopathological studies on many brains have shown that the entire course of the disease may take up to 50 years. About 30 years may pass between the first changes in the transentorhinal region and the first clinical symptoms.
CSF analysis. Until recently, CSF analysis has only been carried out to exclude acute or chronic inflammation. It has now been established by many studies that the CSF of patients with Alzheimer’s disease contains:
• Decreased levels of Aβ peptide1–42 (< 450 pg/mL).
• Elevated levels of total tau protein (> 450 pg/mL).
Pitfalls of CSF analysis in Alzheimer’s disease
Decreased levels of Aβ peptide1–42 and elevated levels of tau protein may also occur in other forms of dementia. Furthermore, about 20% of patients with Alzheimer’s disease have normal levels of Aβ peptide1–42 and tau protein. The interpretation of Aβ1–42 levels is further hampered by the fact that in some studies the levels depended on the ApoE ε4 gene dose. These studies revealed that lower Aβ1–42 levels are already found in nondemented persons carrying one ApoE ε4 allele. Interestingly, higher Aβ1–42 levels have been reported in test persons taking insulin. Whether we will have to work with ApoE ε4-specific and medication-specific reference levels in the future is currently under debate.
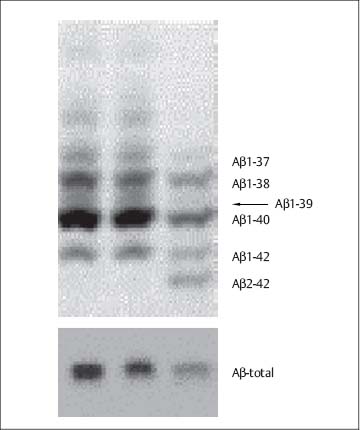
Fig. 11.1 Separation of Aβ peptides in urea-based SDS-PAGE/immunoblot according to Wiltfang (Lewczuk et al., 2004; Maler et al. 2007) (top); this procedure permits quantitative detection of Aβ peptides in the CSF. For comparison, total Aβ fraction in a conventional SDS immunoblot (bottom).
Recent studies suggest that calculating the ratio Aβ1–42/Aβ1–40 is more important for differential diagnosis than determining only Aβ1–42. This has been confirmed for the Aβ1–42/Aβ1–39 ratio in our own investigations into distinguishing Alzheimer’s disease from Creutzfeldt-Jakob disease. In that study, we used a special urea gel electrophoresis in contrast to the conventional ELISA procedures which can only measure Aβ1–42 and Aβ1–40 (Fig. 11.1). However, further studies are still needed before this ratio can be recommended for general use. Determination of phosphorylated isoforms of tau protein should also increase the diagnostic sensitivity for Alzheimer’s disease versus other forms of dementia, although here, too, further studies are needed before generalized use of this marker can be recommended.
References
Braak H, Braak E. Neuropathological staging of Alzheimer-related changes. Acta Neuropathol 1991;82:239–259
Hüll H, Bauer J. Demenzen. In: Berlit P, ed. Klinische Neurologie. Heidelberg: Springer; 1999:829–856
Lewczuk P, Esselmann H, Bibl M, et al. Electrophoretic separation on amyloid beta peptides in plasma. Electrophoresis 2004;25:3336–3343
Maler JM, Klafki HW, Paul S, et al. Urea-based two-dimensional electrophoresis of beta-amyloid peptides in human plasma: evidence for novel Abeta species. Proteomics 2007;7:3815–3820
Further Reading
Kretzschmar HA, Neumann M. Neuropathological diagnosis of neurodegenerative and dementia diseases. Pathologe 2000;21:364–374
Mirra SS, Heyman A, McKeel D, et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer’s disease. Neurology 1991;41:479–486
Lewy Body Dementia
Epidemiology
Lewy body dementia (LBD) is still an underdiagnosed disease. Recent studies suggest that, at 15–35% of cases, it is the second most common form of dementia after Alzheimer’s disease (McKeith et al., 2000).
Clinical Features
Core symptoms. The core symptoms are (Table 11.4):
• Distinct fluctuations in cognitive abilities, particularly in attention.
• Visual hallucinations.
• Spontaneous extrapyramidal motor symptoms.
In addition, recurrent falls, syncope, depression, REM sleep behavior disorder, and pronounced sensitivity to treatment with neuroleptics are often seen.
Fluctuations. One of the core symptoms of LBD is the fluctuation of cognitive deficits and attention. Due to the parkinsonian symptoms, the diagnosis is often Parkinson’s disease or Parkinson’s disease with dementia. For differential diagnosis, it is suggested that if dementia occurs within 12 months alongside parkinsonian symptoms, a diagnosis of LBD should be made. Creutzfeldt-Jakob disease is frequently considered in the differential diagnosis on the basis of the rapid development and the additional focal neurological symptoms.
Diagnosis
CSF analysis. Elevated tau protein levels and decreased Aβ peptide1–42 levels may be found. Therefore, these markers do not permit a distinction between LBD and Alzheimer’s disease in any given case. Recently, it has been reported that elevated levels of heart-type fatty-acid-binding protein (h-FABP) were detected in the serum of LBD patients. In a small study, this marker distinguished between Alzheimer’s disease and LBD (Steinacker et al., 2004). It should be pointed out that the elevation of this marker may imply cardiac involvement in the development of LBD; at least this supposition is confirmed by some neuropathological investigations.
Although no LBD-specific genetic markers have been identified, LBD, like Alzheimer’s disease, occurs more frequently in patients carrying the ApoE ε4 allele.
Neuroimaging. CT and MRI may reveal generalized atrophy with prominent changes in the frontal lobe.
Neuropathology. The disease is characterized by eosinophilic cytoplasmic inclusions called Lewy bodies. During the further course of the disease, Lewy bodies are found not—as in Parkinson’s disease—in the brain stem alone, but also in the cortex and neocortex. Lewy bodies are easily detected by immunohistochemical staining for α-synuclein, a protein constituent of Lewy bodies. Apart from these Lewy bodies, the pathology is to a variable extent typical of Alzheimer’s disease; this is in agreement with the neuropathological diagnostic criteria for LBD.
References
McKeith IG, Ballard CG, Perry RH, et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology 2000;54:1050–1058
Steinacker P, Mollenhauer B, Bibl M, et al. Heart fatty acid binding protein as a potential diagnostic marker for neurodegenerative diseases. Neurosci Lett 2004;370:36–39
Further Reading
Holmes C, Cairns N, Lantos P, Mann A. Validity of current clinical criteria for Alzheimer’s disease, vascular dementia and dementia with Lewy bodies. Br J Psychiatry 1999;174:45–50
Kretzschmar HA, Neumann M. Neuropathological diagnosis of neurodegenerative and dementia diseases. Pathologe 2000;21:364–374
Mc Keith IG, Mintzer J, Aarsland D, et al. Dementia with Lewy bodies. Lancet Neurol 2004;3:19–28
Merdes AR, Hansen LA, Jeste DV, et al. Influence of Alzheimer pathology on clinical diagnostic accuracy in dementia with Lewy bodies. Neurology 2003;60:1586–1590
Spillantini MG, Schmidt ML, Lee VM, et al. Alpha-synuclein in Lewy bodies. Nature 1997;388:39–40
Frontotemporal Lobar Degeneration
Definition and Classification of Frontotemporal Lobar Degeneration
The term frontotemporal lobar degeneration (FTLD) covers three clinical syndromes (Benke and Donnemiller, 2002; Snowden et al., 2002):
• Frontotemporal dementia (FTD)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree