Diagnosis and Differential Diagnosis
POSITIVE DIAGNOSIS
History Taking
“Careful and detailed history taking remains the cornerstone of accurate diagnosis” (O’Donohoe, 1985). History taking should be performed in a methodical and comprehensive manner, but flexibility in practical modalities is an absolute requirement when the clinician is dealing with children. The questions asked should be adapted to each individual, and the doctor should take advantage of the opportunities that arise in conversation rather than following a preestablished order. All relevant points should be covered by the end of the session.
In children, the history is usually obtained from the parents, but data from patients, even when they are young, may be useful because subjective feelings during seizures may be of great importance. Data from other witnesses has to be integrated as well; professional witnesses, such as doctors and nurses, are sometimes less valuable than lay persons because of their preconceived ideas about what a seizure should be like. The history taking may require more than one session because new memories and questions may arise after the first questioning. Care should be taken not to accept previous accounts, even those from professionals, without a critical examination of them, and the clinician must attempt to verify and critically consider all witness accounts (Stephenson, 1990).
Now, a home videotape of the episodes may be available, and such a resource is invaluable. The parents should be encouraged to attempt to obtain one; however, many important points cannot be decided on this basis alone because of the inherent problems with home videotaping, including angles and obstructing persons, and because consciousness cannot be assessed. Careful history taking, therefore, remains essential. As Jeavons (1983b) noted, “the diagnosis is as good as the history.”
The questioning should seek a history of both the complaint (seizures) and the person. The circumstances surrounding the occurrence of the attacks and the possible precipitating factors are of great importance. Seizures regularly precipitated by stimuli such as pain and emotion strongly suggest nonepileptic phenomena. A full description of the seizures, including aura, reconstitution of the whole ictal sequence, and postictal phenomena, is essential. The physician must adapt his or her language to the cultural level of the parents, and he or she should also ask the parents to act out or mimic the attacks (sometimes, he or she can help them by demonstrating the types of movements for which he or she is searching), which may be of considerable benefit.
The manner of asking questions is essential. The questions should be precise without leading the parents, and providing the parents with multiple terms from which they can select those that seem applicable to the description of their child’s seizures can often be useful. Beginning with descriptions of the last attack and the worst attack is often important (Aicardi and Taylor, 1998).
Questions regarding the person with seizures should include his or her developmental history, a detailed drug history, a determination of the impact of seizures on the life of the child and the family, and their level of understanding of the disease.
Neurologic and General Examination
Overview
A complete neurologic and general examination is mandatory. It should especially involve the skin, on which stigmata such as achromic spots, hemangiomas, or other abnormalities may be essential diagnostic clues; the eyes; and anthropometric data, particularly head circumference. Neurologic signs, principally those of cerebral palsy, are important for the diagnosis and prognosis. A rapid assessment of the patient’s competence both cognitively and socially is also essential.
Electroencephalography
Electroencephalographic (EEG) data are important for confirming the epileptic nature of seizures and, if possible, for making a precise diagnosis of epilepsy syndrome. Some patterns, such as the foci of delta waves suggestive of a structural lesion or the bilateral
spike-waves suggestive of idiopathic origin, may help in the search for a cause. However, one must remember that epilepsy is a clinical disorder, so its diagnosis does not depend primarily on EEG findings, no matter how important they may be.
spike-waves suggestive of idiopathic origin, may help in the search for a cause. However, one must remember that epilepsy is a clinical disorder, so its diagnosis does not depend primarily on EEG findings, no matter how important they may be.
In most cases, simple, “standard” EEG recordings are sufficient. The current techniques of digital recording allow reformatting, as well as having other practical advantages. In children, all tracings should include sleep, even though, in most cases, the recording of sleep onset and the first phases of sleep may be sufficient. Other routine facilitating maneuvers include intermittent photic stimulation (IPS) (Kasteleijn-Nolst Trenité et al., 1987, 1999; Harding and Jeavons, 1994; Wilkins and Lindsay, 1985) and hyperventilation (Blume, 1982). Sleep and the use of routine stimulants increase the positivity rate of routine EEG from 60% to roughly 90% (Mizrahi, 1984; Gastaut and Broughton, 1972).
Both hyperventilation and photic stimulation are effective mostly in generalized epilepsies and in approximately one-third of focal epilepsies. However, hyperventilation should be avoided in patients with sickle cell disease, moyamoya disease, and strokes because of the increased risk of aggravating brain hypoxia (Lin et al., 2000; Allen et al., 1976). More sophisticated techniques, such as polygraphy with simultaneous recording of the electromyography, an electrocardiogram (ECG), a respirogram, and an electrooculogram, can provide valuable data in selected patients, especially for precisely classifying seizures or for their differential diagnosis.
Additional nonconventional electrodes (nasoethmoidal, pharyngeal, and sphenoidal) are widely used in some centers for the presurgical investigation of patients with complex partial seizures (Oxbury et al., 2000; Lüders et al., 1989b; Wieser and Elger, 1987), but they are used less often in children (Alvarez et al., 1983). Closely spaced additional electrodes may also be useful for better localizing and defining the electrical field of transients (Blume, 1982).
The correct recognition of epileptiform transients is a crucial prerequisite for an EEG-established diagnosis. A number of EEG graphical elements may resemble these more or less closely. Epileptiform transients include the so-called psychomotor variant pattern, six-per-second spike-waves and 14-6 positive spikes, wicket spikes, and small sharp waves (Blume, 1982). Hypnagogic bursts (Alvarez et al., 1983) are probably the most commonly misdiagnosed EEG pattern in children younger than 5 years, in whom they are often interpreted as bursts of spike-waves.
In about 10% of patients with epilepsy, EEG with routine stimulations and short sleep recording is negative. The sensitivity can be increased by long-duration (24 to 48 hours) recording (particularly overnight or nap records) and sleep deprivation (Dalla Bernardina et al., 1985b; Degen and Degen, 1984).
Long-duration records can be obtained at home with the use of EEG cassettes (Ebersole, 1998; Crandall et al., 1983; Ebersole and Leroy, 1983) or telemetry, or they may be conducted in the laboratory. Invasive EEG techniques are not considered in this text. Video-EEG recording has become an essential tool because it allows the clinician to review clinical phenomena that are often difficult to analyze because of their brevity, as well as to correlate them with the simultaneous EEG study. This method is routinely used in presurgical evaluation. However, no interaction with unattended patients is possible, and, even if the EEG correlates permit the clinician to assign significance to relatively trivial events, the limitations mentioned for videotapes apply to video-EEG as well.
The specificity of interictal EEG is relatively low, especially when stimulations are used. IPS gives a positive result in the form of photoparoxysmal reaction in 5% to 8% of children (Eeg-Olofsson et al., 1971), and sleep may also decrease the specificity and induce graphical elements that can simulate epileptic paroxysms in the absence of epilepsy, making the interpretation of sleep figures and their differentiation from true paroxysms more difficult.
Various types of EEG epileptiform paroxysms can be observed in the interictal EEGs of 5% to 8% of nonepileptic children when an adequate sleep record is obtained. These paroxysmal abnormalities can include generalized spike-wave discharges (9% in the series by Eeg-Olofsson et al. [1971]) and focal spikes or sharp waves that closely resemble those in children with rolandic epilepsy. Cavazzuti et al. (1980) found such spikes in 2.4% of school children from 6 to 14 years of age in Modena, Italy. Eeg-Olofsson et al. (1971) found spike foci in 1.9% of a sample of well children in Stockholm, Sweden. At follow-up, only a small proportion of those children in whom the EEG paroxysms were discovered but who had no clinical history of seizures later developed epilepsy (Cavazzuti et al., 1980). Such abnormalities may be a marker of a genetic susceptibility to seizures. Thus, “characteristic” EEG paroxysms are several times more common than all forms of epilepsy. The coincidence of finding such common EEG abnormalities with equally common clinical paroxysmal events, such as syncope, breath-holding spells (BHSs), or migraine, is, on a purely statistical basis, not unexpected. This
precludes making a diagnosis of epilepsy based solely on the EEG in the absence of convincing clinical manifestations (see “Differential Diagnosis”). Conversely, a normal interictal EEG does not exclude the diagnosis of epilepsy, when the clinical history is convincing.
precludes making a diagnosis of epilepsy based solely on the EEG in the absence of convincing clinical manifestations (see “Differential Diagnosis”). Conversely, a normal interictal EEG does not exclude the diagnosis of epilepsy, when the clinical history is convincing.
The ictal EEG is obviously much more specific. It is pathognomonic when it is associated with the clinical phenomena. In a few cases, however, paroxysmal discharges closely resembling those of seizures may be recorded (e.g., patients who have experienced severe head trauma) (Courjon and Mauguière, 1982). Less rarely, the ictal discharges may be atypical, thus making them difficult to interpret. This may be the case for ictal flattening or for low-amplitude rapid rhythms, which may easily escape recognition.
Finally, genuine epileptic seizures can occur without EEG paroxysms being detectable on the scalp records. This may occur, for example, in cases of frontal epilepsy (Picard et al., 2000; Oldani et al., 1998; Scheffer et al., 1995a) or in other focal seizures originating in parts of the brain that are remote from the electrodes or that are deeply located (Blume, 1982). Therefore, even a negative ictal EEG does not absolutely rule out epilepsy, although it should certainly lead to the consideration of other possibilities.
The occurrence of abnormal EEG discharges in the absence of simultaneous clinical seizures raises a delicate issue. Some of these discharges have been associated with detectable disturbances of cognition or behavior or both (Massa et al., 2001; Binnie, 1990; Kasteleijn-Nolst Trenité et al., 1988, 1990; Binnie et al., 1981, 1987). Precise neuropsychologic testing has demonstrated disruptions of attention, especially with discharges of generalized spike-waves, and memory or of other cognitive functions with focal spikes that are closely correlated with the EEG paroxysms. Such phenomena, which are termed transient cognitive impairments, could be regarded as minimal electroclinical seizures. However, the practical significance of this phenomenon must still be fully assessed, although, in some cases, it has been demonstrated to interfere with the patient’s activities (Aarts et al., 1984).
A similar problem can be raised by the association of intense and continuous (or almost continuous) paroxysmal EEG activity with lasting cognitive or behavioral dysfunction or deterioration even in the absence of seizures. Some investigators have proposed that this type of deterioration may be regarded as a form of prolonged “seizure” or status and that it may be the direct consequence of the epileptic disturbance that is expressed by the EEG abnormalities (Deonna, 1996). Examples include continuous spike-waves of slow sleep (or electrical status epilepticus of slow sleep) and Landau-Kleffner syndrome (Beaumanoir, 1992). Hypsarrhythmia and intense spike-wave activity in Lennox-Gastaut syndrome have been similarly interpreted by many investigators (see Chapter 3).
A close relationship between cognitive disturbances and EEG discharges is suggested by the coinciding times of occurrence of both phenomena, the possible parallels between the intensity of EEG and clinical anomalies, and the observation that the clinical anomalies do not seem to disappear as long as the EEG paroxysms have not cleared (Aicardi, 1999a; Engel and Pedley, 1998; Deonna, 1996; Morrell et al., 1995). Deonna (1996) suggested using the term cognitive epilepsy to describe such occurrences. The significance of these is of practical importance because efforts at controlling the EEG activity might also check the neuropsychologic disturbances, even though this has not been demonstrated. This dilemma is further discussed in Chapters 3, 4, and 11.
Other Laboratory Tests
Other laboratory tests play only a minor role in the diagnosis of seizures and epilepsy. Prolactin is released following generalized (Fisher et al., 1991) and some focal (Bilo et al., 1988; Molaie et al., 1988; Sperling et al., 1986b) seizures, but this does not occur with pseudoseizures. Thus, determining the level of serum prolactin within an hour of a seizure can help establish its true nature. However, hypoxic attacks can also result in prolactin release, so the practical value of the test is limited. An increase in creatine kinase levels is seen after generalized seizures only, and it is related to muscle activity.
DIFFERENTIAL DIAGNOSIS
Definition of Epilepsy and Diagnosis Issues
This section is concerned with differentiating epileptic seizures and epilepsy from other nonepileptic, paroxysmal clinical events. Epilepsy is characterized by the repetition of unprovoked seizures, so, even though the diagnosis of an epileptic seizure does not establish that of epilepsy, it is crucially important. A diagnosis of epilepsy requires that the repeated seizures are unprovoked (Engel and Pedley, 2001). A commonly used definition of repetition is the occurrence of two or more events that are not part of the same episode. This epidemiologic definition is of relatively little help in clinical practice. The notion of chronicity that is associated with epilepsy implies a
“sufficient” duration, which clearly is arbitrary. Conversely, some single clinical episodes occurring in association with characteristic EEG patterns (e.g., in rolandic epilepsy) may well be considered to fulfill, for all practical purposes, the major criteria for epilepsy if not for treatment.
“sufficient” duration, which clearly is arbitrary. Conversely, some single clinical episodes occurring in association with characteristic EEG patterns (e.g., in rolandic epilepsy) may well be considered to fulfill, for all practical purposes, the major criteria for epilepsy if not for treatment.
The requirement of unprovoked seizures also is in need of more precision. Some triggers, such as photic stimulation, and a vast number of precipitating factors, such as reading or cognitive or language activities, are conventionally included in the group of the so-called reflex epilepsies, whereas others, such as fever, are not, even though they demonstrate the individual’s special sensitivity to seizures.
The differential diagnosis of epilepsy is of utmost importance because of its personal and social consequences for the affected individual, so it must be made only on solid evidence. The differential diagnosis of individual epilepsy syndromes is briefly considered in the preceding chapters. The present discussion is limited to considerations relevant to the diagnosis of epilepsy in general.
Epilepsy is, unfortunately, grossly overdiagnosed. Jeavons (1983b) found that 20% to 25% of patients referred to his epilepsy clinic did not have epileptic seizures. Likewise, Metrick et al. (1991) reported that 10% to 20% of children who had been referred with a diagnosis of refractory epilepsy actually had nonepileptic attacks. Because an incorrect label of epilepsy can have profound and regrettable consequences on a child’s schooling, social life, and employment, such misdiagnoses must be avoided.
Jeavons (1983b) listed the most common misdiagnoses in 200 patients. Syncopes accounted for 44% of errors, psychiatric disturbances for 20%, BHSs for 11%, migraine and night terrors for 6% each, and miscellaneous conditions for 11%. The main reasons for misdiagnosis were an inadequate history, the presence of a family history of epilepsy or a history of febrile seizures, an “abnormal EEG,” and misinterpretation of clonic movements or incontinence as being inherently epileptic. Jeavons (1983b) also stressed the obvious but often overlooked fact that nonepileptic seizures may occur in epileptic patients who are being treated and, therefore, that any seizure in a “known epileptic” should not necessarily be interpreted as a recurrence. In a series composed entirely of children, Robinson (1984) found that the diagnosis of epilepsy was doubtful in 94 of 201 patients referred to his outpatient clinic with a possible diagnosis of epilepsy, and 75 children were proved to have some other kind of episodic attack. The most common nonepileptic paroxysmal disorders in his series were BHSs, fainting, migraine, and dizziness or vertigo. Metrick et al. (1991) found that the most common cause of a misdiagnosis of epilepsy in children was an unusual reaction to stimuli that included an arrest of ongoing activity, stereotypic behavior, and/or abnormal movements. The main types of nonepileptic attacks are considered in “Main Differential Diagnoses” below (Pranzatelli and Pedley, 1991; Stephenson, 1990; Aicardi, 1988c).
Underdiagnosis of epilepsy is much less common. However, the wrong diagnosis of nonepileptic seizures can be made in those patients who have atypical manifestations that are wrongly thought to be of psychogenic origin, who exhibit some abnormal paroxysmal movements, or who, as in cases of familial frontal epilepsy, present with nocturnal seizures that, not infrequently, are interpreted to be parasomnias (Oldani et al., 1998). In a few cases, the possible diagnosis of epilepsy may not be considered in patients presenting with mainly cognitive or behavioral symptoms; however, a sleep EEG may reveal intense paroxysmal activity. Some of these have rare, subtle seizures, and others may not have any seizures at all. The term cognitive epilepsy has been proposed for such cases (Deonna, 1996), emphasizing the notion that cognitive and/or behavioral manifestations can be regarded to be the direct consequence of the epileptic activity and that they represent an equivalent to seizures. Even though some uncertainty still exists about the exact significance of such cases because both EEG changes and clinical manifestations may also be regarded as being due to the same basic dysfunction that is as yet undetermined (Holmes et al., 1981), the recognition of these EEG abnormalities is of definite diagnostic and possibly therapeutic value.
Some abnormal paroxysmal motor events may not be recognized as epileptic because of their unusual characteristics. This may be true for some patients with seizures in the supplementary motor area (Kaplan, 1975), in whom these may be regarded as psychogenic pseudoseizures (Kawazawa et al., 1985), and for some with nocturnal familial frontal epilepsy, which is often misdiagnosed as parasomnias or behavior disorders (Oldani et al., 1996, 1998).
Main Differential Diagnoses
Anoxic Seizures
Although the use of the term anoxic seizures is not correct because some degree of oxygenation does persist,
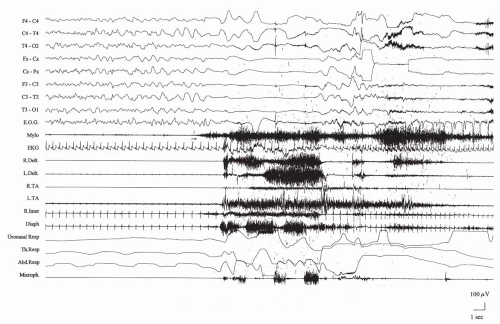
it is commonly used and thus is employed here. Anoxic seizures comprise a heterogeneous group of paroxysmal events that include a loss or attenuation of consciousness and variable other sensorimotor phenomena. Cortical anoxia that is sufficient to provoke syncope may occur under a number of the following circumstances: bradycardia with a heart rate of less than 40 beats per minute or tachycardia with more than 150 beats per minute, asystole of more than 4 seconds, systolic pressure of less than 50 mm Hg, or a jugular venous O2 concentration of less than 20 mm Hg. Obstructive apnea may occasionally be seen in children with a palatal malformation (Fig. 21.1).
it is commonly used and thus is employed here. Anoxic seizures comprise a heterogeneous group of paroxysmal events that include a loss or attenuation of consciousness and variable other sensorimotor phenomena. Cortical anoxia that is sufficient to provoke syncope may occur under a number of the following circumstances: bradycardia with a heart rate of less than 40 beats per minute or tachycardia with more than 150 beats per minute, asystole of more than 4 seconds, systolic pressure of less than 50 mm Hg, or a jugular venous O2 concentration of less than 20 mm Hg. Obstructive apnea may occasionally be seen in children with a palatal malformation (Fig. 21.1).
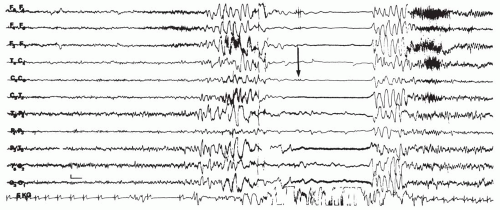
Severe decorticate rigidity, when it ensues, is caused by the loss of corticoreticular inhibition of certain brainstem structures involved in the control of muscle tone (Gastaut, 1974). On the EEG, increasing degrees of cortical anoxia produce slowing of the frequency of the dominant cortical rhythms. If the decorticate state is reached (convulsive syncope), complete electrical silence with an isoelectric recording occurs. Convulsive syncope seems to be more common than generally assumed; it has been observed in 42% of prospectively studied blood donors who fainted (Lin et al., 1982) and in an even higher proportion of young adults who fainted as a result of the Valsalva maneuver (Schmidt and Lampert, 1990). With the resumption of sufficient blood flow, the slow waves reappear; they may be synchronous with a few jerks of the limbs (Gastaut, 1974; Lombroso and Lerman, 1967) (Fig. 21.2). Normal rhythms return rapidly, and very little postictal abnormality is observed.
Reflex Anoxic Seizures and Breath-Holding Spells
Reflex anoxic seizures (RASs) and BHSs are probably pathophysiologically distinct phenomena. RASs are usually the result of a temporary asystole that is of reflex origin, and BHSs feature cyanosis and grunting that suggest the presence of expiratory apnea with intrapulmonary blood shunting and a mismatch of ventilation and perfusion (Stephenson, 1990; Southall et al., 1985). The clinical differences between them can, however, be minimal if they even exist. RASs and BHSs may often coexist in the same child, with differing mechanisms for different episodes; there is no absolute proof that they differ essentially (Stephenson, 2001).
The overall frequency of RASs and BHSs, which occur in about 4% of children, is high. Lombroso and Lerman (1967) found that 19% of cases were of the pallid type, probably due to the reflex anoxic mechanism, and 76% were cyanotic, which is suggestive of BHSs. The onset mostly occurs in infants and toddlers between 6 and 18 months of age, but a very early onset in the first days of life can be seen.
Reflex Anoxic Seizures.
Classic RASs are typically precipitated by pain, often due to a bump on the head. The toddler may begin to cry, but the crying is rapidly interrupted by a loss of consciousness and tone, resulting in a fall that may be slow and progressive or abrupt. When the asystole lasts more than a few seconds, hypertonia of the trunk and limbs sets in and opisthotonus, a rare manifestation of epileptic seizures, is common. A few jerks and eye deviation, either upward or downward, are common (Engel and Pedley, 1998; Lempert et al., 1994; Lin et al., 1982). The child is pale, and, occasionally, the parent may note the absence of pulse. After a variable period (usually 30 to 60 seconds), the child returns to consciousness, and the hypertonia resolves. In some cases, a genuine, usually clonic, epileptic seizure immediately follows the tonic episode (Stephenson, 2001; Battaglia et al., 1989; Aicardi et al., 1988). Such seizures, which have been termed anoxic epileptic seizures, may be of long duration, and cases of resulting status epilepticus have been recorded (Emery, 1990; Stephenson, 1990). Conversely, a true epileptic seizure may result in severe hypoxia as a consequence of the interference of the discharge with the respiratory control mechanisms; this is the so-called epileptic anoxic seizure (Stephenson, 1983). However, this rarely causes a misdiagnosis of epilepsy.
Atypical features such as lateral body incurvation and postictal confusion are common. In older children, auras, which usually consist of an unpleasant feeling of discomfort but which may occasionally be quite complex, including hallucinations and automatisms that are sometimes elaborate, may complicate the diagnostic problem (Stephenson, 2001).
The consistent presence of a precipitating factor is a major characteristic, but this may be difficult to
elicit; it often escapes detection with perfunctory questioning. Older children and adolescents tend to be reluctant to tell “strange stories,” thus adding to the difficulties. Contrary to the common belief, RASs can occur not only when an individual is standing or sitting but also when one is lying down, so the individual’s position does not rule out the diagnosis.
elicit; it often escapes detection with perfunctory questioning. Older children and adolescents tend to be reluctant to tell “strange stories,” thus adding to the difficulties. Contrary to the common belief, RASs can occur not only when an individual is standing or sitting but also when one is lying down, so the individual’s position does not rule out the diagnosis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree