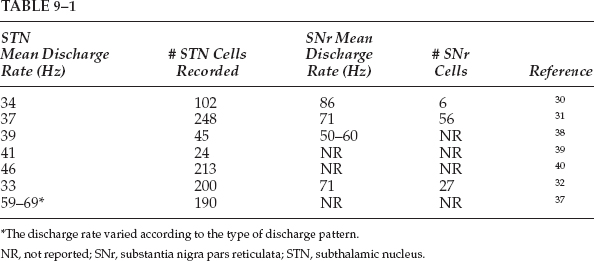
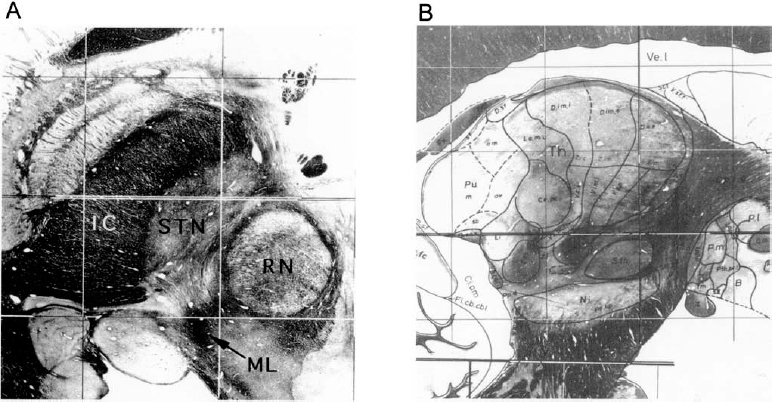
9 The subthalamic nucleus and the substantia nigra pars reticulata are basal ganglia structures that play important roles in the control of frontal cortical functions, including movement. Interest in the electrophysiology of the STN and SNr is prompted by their rapidly emerging role in the surgical treatment of Parkinson’s disease and other disorders of the central nervous system. An understanding of the electrophysiological characteristics of the STN and SNr is important for successful surgical navigation in these areas. The STN is located at the midbrain–diencephalic junction, ventral to the thalamus. It is bordered by the zona incerta superiorly, the cerebral peduncle anterolaterally, and the red nucleus posteromedially. The SNr is in the upper midbrain, immediately ventral to the STN (Fig. 9–1). In humans, the STN measures ~7 mm in both the rostrocaudal axis and the mediolateral axis. The basal ganglia nuclei form parts of loop circuits connecting them with functionally related areas of the cortex and the thalamus. Basal ganglia–thalamocortical loops serve nonmotor as well as motor functions. The STN and SNr have extensive connections to other structures within these loops. Of the two nuclei, the STN has more divergent outputs. The primary input structure of the basal ganglia is the striatum, and its primary outputs are the internal segment of the globus pallidus and the SNr. Starting in the late 1980s, the internal connections in the basal ganglia motor subcircuit were conceptualized as forming two distinct pathways: a “direct” striatum to the GPi/SNr connection and an “indirect” path in which striatum projects to the external segment of the globus pallidus, the GPe projects to the STN, and the STN projects to the GPi/SNr.1 The STN, therefore, connects input structures of the basal ganglia with their outputs via the indirect pathway, and the SNr is one of the two output structures of the basal ganglia (along with the GPi) where direct and indirect pathways converge. In addition to its inhibitory afferents from the GPe, the STN receives strong afferent excitatory projections from various frontal areas of the cortex.2,3 This simple conceptualization has been evolving, although there is not yet a new formulation that is widely accepted. For example, Parent and Hazrati3 found that the anatomic territory of the STN receiving the major GPe projection does not overlap with the territory giving rise to the efferent projection to the GPi or SNr, calling into question the concept of the indirect pathway. Other anatomic data, however, continue to support the existence of the indirect pathway.4 In addition, there are likely to be multiple indirect pathways from the striatum to the GPi, including one from the striatum to the GPe to the GPi.5 Although the STN is currently the more popular surgical target for treatment of PD, an appreciation of the physiology of the SNr is also highly relevant to surgical treatment of movement disorders. The SNr forms the ventral border of the STN, and thus its recognition is important for surgical navigation in the STN region. In addition, as one of the paired output nuclei of the basal ganglia, it is possible that interruption of the SNr, like its “sister” nucleus, the GPi, could prove beneficial in movement disorders.6 Finally, the SNr is anatomically juxtaposed with the substantia nigra pars compacta (SNc), and thus could serve as a “signpost” marking the region of the depleted SNc during surgical procedures aimed at the delivery of growth factors or biologics to the SNc for PD.7 It has long been known that damage to the region of the STN in humans can produce a hyperkinetic state. Experimental STN lesions in otherwise normal rhesus monkeys have been shown to produce hemiballism.8 More recently it has been shown that the STN plays a major role in the pathophysiology of Parkinson’s disease. According to the standard model of parkinsonian pathophysiology,1 decreased striatal dopamine levels result in reduced activity in the GPe, disinhibition of the STN, and an increase in the excitatory output of the STN to the GPi. The overall effect is an increase in the inhibitory signal from the GPi to the thalamus and a suppression of activity in cortical areas involved in motor control, resulting in a hypokinetic state. This model predicts that inactivation of the STN should help to “normalize” basal ganglia output and partially compensate for the effects of striatal dopamine loss. Strong evidence for this has come from studies of primates rendered parkinsonian by injection of MPTP. In this model, STN lesions reversed all motor disturbances.9 Unilateral lesions of the STN have been used in the treatment of PD with moderate efficacy.10,11 The introduction of chronic high-frequency DBS as a surgical alternative to lesioning has created further interest in the STN as a target in PD.12,13,14 The development of intractable hyperkinetic states with STN lesioning or DBS in PD has been observed but is unexpectedly rare.15 The parkinsonian state probably raises the threshold for the development of hyperkinesias after lesions of the STN, possibly due to the decreased activity in the direct striatopallidal projection.16 Surgery of the STN/SNr region may also be relevant to the treatment of epilepsy. An area of the dorsal midbrain, the dorsal midbrain antiepileptic zone (DMAEZ), has been shown to modulate seizure activity in several rat models of epilepsy.17 The DMAEZ is under tonic inhibition by the SNr, which in turn is under tonic excitation by the STN. Inactivation of the STN, along with the resulting suppression of SNr activity, appears to disinhibit the DMAEZ and thereby produces an antiepileptic effect.18,19 Based on this evidence, STN-DBS for intractable epilepsy is under investigation in humans. In addition to their skeletal motor function, the basal ganglia participate in limbic, associative, and oculomotor functions. For each of these modalities, there exists a loop circuit connecting functionally related areas of the cortex, basal ganglia, and thalamus.20 In general, the circuits subserving these different functions appear to be anatomically segregated, although there may be interconnections between the loops.21 The principle of the segregation of motor from nonmotor circuits is critically important for surgical navigation within the basal ganglia because interventions for Parkinson’s disease should ideally selectively target the motor area of the relevant nuclei, avoiding influence on the nonmotor areas. For example, unexpected stimulation-induced effects on mood have been reported with STN-DBS,22,23 which may relate to the spread of stimulation beyond the motor territory of the STN into its nonmotor regions, or into nonmotor regions of the SNr. Motor and nonmotor areas of the STN and SNr have been distinguished based on their pattern of connectivity to related structures, as well as from electrophysiological studies. Most of this work has been in primates. For the STN, the dorsolateral part of the nucleus appears to represent its motor territory, based on the dorsolateral termination of the major corticosubthalamic projection from the primary motor cortex.3,24,25 In addition, the dorsolateral STN projects to the motor territory of the GPe. The premotor cortices (Brodmann’s area 6) project to more medial and ventral areas than the primary motor cortex efferents and define the medial extent of the motor territory.3,24–27 The ventral STN is likely to be an association area rather than a region involved in motor control, based on the fact that it projects primarily to the association areas of the GPi and SNr, as well as to the caudate nucleus, a part of the striatum involved in the association loop circuit.3 The medial tip of the STN is considered limbic territory, based on its anatomic projection to the limbic (ventral) pallidum.28 An oculomotor area of the STN has also been defined. In a primate study involving the performance of a visuo-occulomotor task, approximately one third of the neurons assessed showed discharge modulation related to eye fixation, saccades, and visual stimuli. Oculomotor-related neurons were primarily located in the ventral part of the nucleus.29 The boundaries between the motor and nonmotor subdivisions of the STN have not been studied extensively in humans, but human electrophysiologic studies indicate that cells whose discharge is modulated by limb movements are found dorsolaterally,30–32 not ventrally or medially, as expected from primate data. Limbic or association areas of the human STN may be relatively larger than in primates. Within the SNr, the topography of the oculomotor circuitry is better delineated than that of the skeletomotor territory. Transynaptic retrograde tracing techniques have shown that the area of the SNr projecting to frontal eye fields (via the thalamus) is lateral and caudal within the nucleus; this area is thus considered the oculomotor territory.28 The nature and location of the skeletomotor subterritory of the SNr are somewhat controversial because transynaptic tracing has not shown a skeletomotor cortical area that is associated with SNr.28 Other types of experimental evidence, however, indicate the presence of a skeletomotor circuit within the central and lateral parts of the nucleus. This is based both on the termination of striatonigral fibers that arise in the motor part of the striatum33,34 and on electrophysiological studies showing that cells responsive to movement occur in the central and lateral areas.34,35 However, in the centrolateral SNr, there may be anatomic convergence of motor and nonmotor circuits.33 Thus, the general organizing principle that motor and nonmotor circuits of the basal ganglia are anatomically segregated may not be absolutely true in all regions. Wichmann et al36 have studied the spontaneous discharge characteristics of the primate motor STN in the normal and parkinsonian (MPTP-treated) state. In the normal state, the mean resting discharge rate was 19 ± 10 Hz. Based on an analysis of autocorrelograms, 69% of the neurons showed burst discharges, 2% showed 4 to 8 Hz oscillations, and 0.7% showed 8 to 20 Hz oscillations. After treatment with MPTP until parkinsonian signs appeared, the mean resting discharge rate increased to 26 ± 15 Hz; 79% of the cells discharged in bursts, 16% had a tremor frequency oscillatory pattern, and 10% exhibited 8 to 20 Hz oscillations.36 Thus, the induction of the parkinsonian state is associated with an increase in the mean firing rate, as well as a greater tendency to discharge in rhythmic and non-rhythmic bursts. Several groups have reported the spontaneous discharge rate of human parkinsonian STN during surgery for PD (Table 9–1). With the exception of one study,37 these measurements are all fairly consistent and place most cells in the 30 to 50 Hz range. Rhythmic oscillatory discharge has been recorded in the human parkinsonian STN. Many groups have reported the occurrence of oscillatory discharge in the tremor-frequency range.30–32,41,42 Oscillations at higher frequencies also have been studied. Levy et al41 reported on 213 STN cells and found 4% of cells with tremor-frequency oscillations, 20% with higher frequency oscillation (> 10 Hz), and 6% of cells exhibiting both tremor and higher frequency oscillatory activity. Using simultaneous recordings from multiple electrodes, they found that pairs of cells showing oscillatory activity tended to fire synchronously. Although comparable data from nonparkinsonian humans is not yet available, these results are consistent with the theory that parkinsonism is associated with an increase in synchronous firing between neuronal pairs over certain frequency ranges, as has been shown in the dopamine-depleted primate for the globus pallidus43 and the striatum.44
Electrophysiological Findings in STN and SNr
PHILIP V. THEODOSOPOULOS, ROBERT S. TURNER, AND PHILIP A. STARR
The STN and SNr in Movement Disorders
STN/SNr in Epilepsy
Motor versus Nonmotor Functions of STN/SNr
Spontaneous Discharge Characteristics: Normal and Parkinsonian
Neupsy Key
Fastest Neupsy Insight Engine