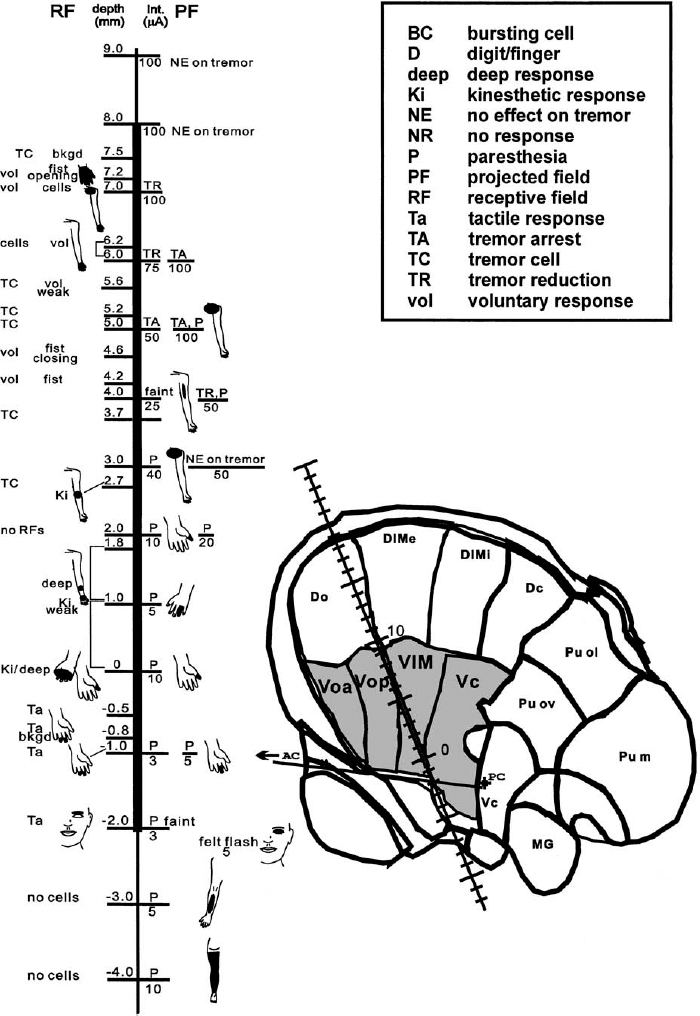
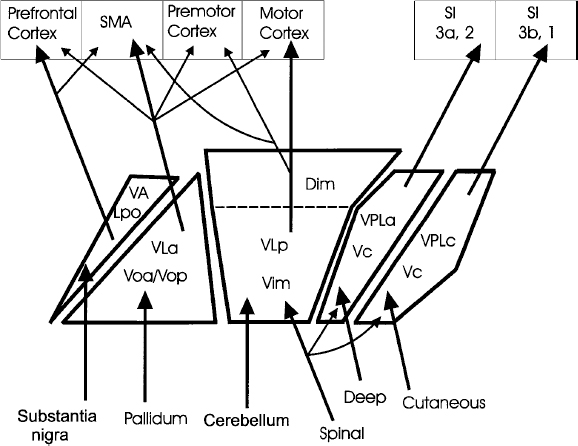
7 The electrophysiological characteristics of the thalamus have been used for close to 40 years as an aid in target localization. Although many groups rely entirely on the sensory and motor effects of stimulation through large-tipped electrodes (macrostimulation) for functional localization, the use of microelectrode recordings of neuronal activity provides more definitive data and higher resolution. The combination of microelectrode recordings and stimulation (microstimulation) as used by our group provides additional physiological information that aids in the precise localization of the target site. Microelectrode recording in the ventral tier of thalamic nuclei can readily identify the tactile relay nucleus, the ventral caudal nucleus (Vc), and its anterior border with the ventral medial nucleus (Vim), as well as the ventral border of the thalamus. Furthermore, the recordings allow one to identify regions of the motor thalamus containing neurons responsive to limb and jaw movements and neurons firing in synchrony with tremor (tremor cells). These regions are considered to be good markers for the target for a lesion or DBS electrode insertion aimed at reducing tremor. The major nuclei that comprise the ventral lateral part of the thalamus are the ventral oral anterior and posterior (Voa and Vop), the Vim, and the Vc (terminology of Hassler1 and Schaltenbrand and Wahren atlas2) (Fig. 7–1). Anterior and lateral to these nuclei lie the reticular nucleus of the thalamus and the internal capsule. There is an ongoing debate about terminology and exact definitions of the thalamic nuclei in this region (e.g., see Macchi and Jones3 and Jones4). This partly stems from the difficulties in extrapolating the terminology used in the primate thalamus, where most of the recent anatomical tracer and projection studies have been done,3–5 to the human thalamus. Hassler’s terminology for the human thalamus has become established in the clinical field, as it is widely adopted in the main stereotactic atlases used in surgery.2 The Vc is the major termination site of the medial lemniscus and projects to the primary somatosensory cortex. This region probably also includes anteriorly and dorsally a thin shell containing neurons responding to somatic inputs arising in muscle, joints, and deep tissues and which project to area 3a of the somatosensory cortex.4 The cells anterior to the Vc receive cerebellar (and some spinothalamic tract) input. There is some disagreement, however, as to whether this cerebellar receiving zone is limited to Vim or also includes some or all of Vop.4–6 Anterior to the cerebellar receiving area, the neurons receive inputs from the basal ganglia, in particular from the globus pallidus internus. This region is the Voa and possibly all of the Vop. The physiological determination of the locations of the thalamic nuclei by intraoperative MER is dependent on a cumulative acquisition of information of various types, as explained later, as well as integration of the data from multiple tracks through the region. These data, in conjunction with the image-guided stereotactic localization and its predicted location, are used to further increase confidence in establishing the nucleus from which the recording is obtained. Among the important differentiating electrophysiological data in the Voa, Vop, Vim, and Vc area are the neuronal firing rates and patterns of discharge, the density of neurons and the amplitudes of the extracellularly recorded action potentials (spikes), the responses of the cells to voluntary and passive movements and to superficial and deep mechanical stimuli, the synchrony of the neuronal firing rate to tremor, if any, and the sensory and/or motor effects of microstimulation. Of note is that some of these characteristics may be different depending on the disorder and state of arousal of the patient. FIGURE 7–1 Schematic diagram showing the thalamic ventral tier of nuclei and their major inputs and outputs. Details on the technical aspects of microelectrode recordings during stereotactic neurosurgery can be found in Dostrovsky7 and Lenz et al.8 The microelectrode should be slowly advanced through the thalamus, and changes in activity with depth should be noted. These comprise both changes in background “noise” that signify white versus gray matter and the amount of activity in the cellular regions of gray matter. This overall activity is a function of the density of the cells, the sizes of the action potentials, and the degree of spontaneous activity. It is also dependent on the electrode tip size, as very small tips will tend to emphasize the activity arising from a few cells close by and provide little information on the overall activity of neurons in the vicinity of the electrode tip. The overall activity is best monitored aurally by feeding the amplifier output (preferably with a background noise suppression circuit) into a loudspeaker and visually by observing the microelectrode signals at fairly slow sweep speed (e.g., 100–500 ms/division) on a digital oscilloscope or computer monitor. When a well-isolated neuron is encountered, its firing should be observed for a period of at least 30 sec. Assuming the recording is stable, it is useful to note the approximate firing rate and firing pattern in the absence of movement and stimulation. If the patient has ongoing tremor, it is important to note if the cell fires in synchrony with the tremor. It is then necessary to determine if the cell can be activated by passive or voluntary movements of the main joints, including the jaw, tongue movements, or mechanical stimuli applied to skin or deep tissues. Finally, at intervals of ~1 mm, one should determine the effects of stimulation (300 Hz, 1 sec trains of up to 100 μA). If the patient has tremor, the effects of stimulation on tremor should also be determined. The patient should also be asked to report any sensations induced by the stimulation, and the threshold for inducing these should be determined. We always choose as our target for the first trajectory a site halfway between the anterior and posterior borders of Vc and a few millimeters above the bottom of the nucleus to maximize the chance of finding the tactile responsive neurons of Vc on the first trajectory. This is because the Vc is the most easily recognized of the ventral tier of nuclei. Subsequent electrode tracks are then positioned on the basis of the findings of the first track and the desired location of the final target (Figs. 7–2 and 7–3). Because most frontal trajectories enter the thalamus at an angle between 60 and 80 degrees, a microelectrode targeted at the ventral half of Vc is likely to hit the upper part of the Vop first, then the Vim, and finally the Vc (Fig. 7–2). The first neural activity recorded (when starting ~10–15 mm from the target) is frequently of neurons firing in a very characteristic bursting pattern and which do not have a receptive field (Fig. 7–4A). The density of spontaneously active neurons in this region is fairly low. Such neurons are probably more frequently encountered when the patient is drowsy, but many will still fire in this bursting pattern even when the patient is alert and making movements or responding to questions. Microstimulation in this region is generally without any effect. These neurons are located in the dorsal Vop/Vim or in Dim. Below this region one generally encounters increased density of neurons firing more rapidly and regularly. The density of neurons and spike amplitudes are higher in Vim and Vc than in Vop and even lower in Voa. In these regions the neurons frequently do have receptive fields, and stimulation in Vim and Vc will usually produce effects. These characteristics are described in greater detail later. As the electrode is advanced down ventrally and posteriorly through the thalamus, the first neurons encountered that respond to movements are usually voluntary cells. These cells respond only or preferentially to voluntary movements. They probably fire in advance of their related contralateral movement, and some have RFs of a kinesthetic nature consisting of a passive movement that opposes that of the related voluntary act.9 They are predominantly present in the Vop and Voa. Raeva et al10,11 demonstrated that voluntary cells in the Voa and Vop do not respond to verbal command but rather to the voluntary act itself. In the operating room, where one is limited in time, it is sometimes difficult to determine whether a neuron responding to both voluntary and passive movements is a voluntary cell or a kinesthetic cell if the responses are similar. Voluntary cells with receptive fields involved in tremor movements will also usually fire rhythmically in synchrony with the tremor and are termed tremor cells. Stimulation in the region where voluntary cells are encountered is usually without effect, but if the patient has tremor during the stimulation, tremor reduction or arrest may occur at some sites. The stimulation parameters optimal for producing tremor arrest or paresthesia are 300 Hz, 1 sec trains of 0.1 or 0.2 ms pulse width. Stimulation current is usually limited to a maximum of ~100 μA when using high-impedance microelectrodes. As the electrode progresses posteriorly, the incidence of neurons responding to passive movements increases. Recording in the human Vim region reveals a preponderance of cells that respond to proprioceptive stimuli, the so-called kinesthetic cells. These cells change their firing rate on passive movement across a joint or deep pressure or brisk tap on a muscle or skin (see example in Fig. 7–4B). Most kinesthetic cells will also change their firing rate during voluntary movements; this is attributed to the proprioceptive stimuli produced by the active movement. However, the responses to voluntary movements are similar to or lower than those of a similar movement produced passively. If the patient has tremor involving the receptive field of kinesthetic cells, then the cells will usually fire in rhythmic bursts time-locked to the tremor12 (Fig. 7–4C). Such tremor cells are usually very readily observed in patients with parkinsonian tremor because of the continuous and frequently pronounced tremor. In contrast, in essential tremor, which is characterized by a postural kinetic tremor, the tremor must be induced. Characteristically, these tremor cells cease firing rhythmically when the tremor arrests. The kinesthetic neurons usually are found in the thalamic region corresponding to Vim and extending for ~2 mm anterior to the tactile border. It is generally assumed that all these neurons are in the cerebellar receiving area of the motor thalamus and as such project to motor cortical regions, in particular to area 4 (the primary motor cortex).13,14 However, it is very likely, on the basis of findings in cats and monkeys, that the kinesthetic/deep neurons lying immediately adjacent (anterior and possibly dorsal) to the tactile responsive neurons are in the lemniscal receiving area and project to area 3a of the primary somatosensory cortex. According to Jones,4 these neurons are still part of Vc, and in fact he has suggested that they are located in a shell corresponding to Hassler’s Vcae (ventral caudal anterior external). This shell is the recipient of deep medial lemniscal afferents, and its neurons in monkeys were shown to project to sensory areas 3a and 2.15 The tactile receiving area is located in the central part of VPL, which corresponds to Vcep.3,4 Electrophysiologically the kinesthetic cells in this “proprioceptive shell” are very sensitive to light pressure on subcutaneous tissue and muscles and because of their high sensitivity are also activated by movements.16 Although the sensitivity and response magnitude decrease for Ki cells located more anteriorly, it is difficult to determine a clear boundary between these putative Vc kinesthetic cells and those of Vim.
Electrophysiological Findings in Vim and Vc
HAZEM EL-TAHAWY, ANDRES M. LOZANO, AND JONATHAN O. DOSTROVSKY
General Methods
Background Activity
Neuronal Activity
Findings along a Typical Electrode Trajectory
Voluntary Cells
Kinesthetic Cells
Neupsy Key
Fastest Neupsy Insight Engine