• Congestive heart failure (class III/IV) and/or known severe left ventricular dysfunction LVEF <30 %
• Open heart surgery within 6 weeks
• Recent MI (>24 h and <4 weeks)
• Unstable angina (CCS class III/IV)
• Coexistent severe coronary artery disease requiring carotid and coronary revascularization
• Severe pulmonary disease (FEV <1.0)
• Contralateral carotid occlusion
• Contralateral laryngeal nerve palsy
• Post-cervical radiation treatment
• Previous CEA (i.e., recurrent stenosis)
• High cervical ICA lesions (C2 or higher)
• CCA lesions below the clavicle
• Severe tandem lesions
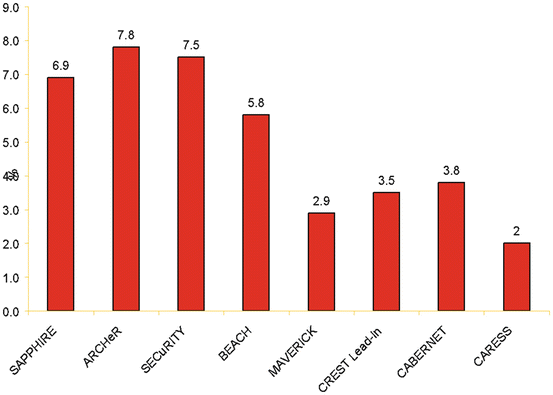
Fig. 4.1
Bar chart illustrating the steady reduction in procedural morbidity and mortality over time
Following the development of stent and then filter technology, a clinical trial comparing CEA and CAS was needed. Protected stenting (i.e., stenting with the use an EPD) had not been validated outside of registries [2–4]. Several small and poorly conducted trials were initiated [5, 6], but to date the only large randomized trial of protected CAS vs. CEA in high-surgical-risk patients has been completed: the Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) study [7]. This non-inferiority study evaluated the Cordis PRECISE™ stent and the ANGIOGUARD™ filter and was sponsored by the manufacturer Cordis Endovascular Inc. The primary study endpoint of stroke, myocardial infarction, or death at 30 days and ipsilateral stroke to 1 year was 12.2 % with CAS and 20.1 % with CEA, p = 0.05 for non-inferiority. The 30-day perioperative stroke/MI/death was lower in the CAS group as compared with the CEA group, 4.4 % vs. 9.9 %, respectively, but the difference was not statistically significant (p = 0.06) in the on-treatment analysis. The CEA complication rates were much higher than those noted in the “low-risk” studies NASCET (5.8 %) and ACAS (<3 %) [1, 8]. The 3-year results continued to show non-inferiority of CAS to CEA with a cumulative major adverse event rate (stroke, death, or MI) of 20.1 % in the stenting arm and 30.3 % in the CEA arm (p = 0.231). Additionally, the need for reoperation at 1 year was significantly lower in the CAS group than in the CEA group, 0.7 % vs. 4.6 %, respectively, p = 0.04.
In addition to the randomized data from SAPPHIRE, two data sets from large post-marketing studies, CAPTURE and CASES-PMS [9, 10], show continued good outcomes with CAS in high-surgical-risk patients. Both of these registries consisted of real-world experience with commercially available stent and embolic prevention systems as well as independent neurological adjudication of outcomes and events.
The much larger follow-up registry, the SAPPHIRE Worldwide Registry, has enrolled more than 15,000 patients since October 2006. The periprocedural results were presented at the Transcatheter Cardiovascular Therapeutics (TCT) conference on October 23, 2012, on the first 15,003 patients, 4,569 of whom (30 %) were symptomatic and 10,433 (70 %) were asymptomatic. The 30-day stroke/MI/death rate was 4.5 % (death 1.2 %, MI 0.6 %, stroke 3.3 %). There was a significant difference in the “NASCET” 30-day endpoint (stroke/death) between symptomatic (5.6 %) and asymptomatic (3.5 %) patients (p < 0.0001). This registry also confirmed that patients 75 years of age and older had a higher complication rate (5.6 % vs. 2.9 %) compared to younger patients (p < 0.0001). These results compare favorably with the results of the CEA arm in the SAPPHIRE trial, the only randomized data set to define the outcomes of CEA in this patient population. These results also compare favorably with ACAS and ACST surgical results of approximately 3 % 30-day stroke/death in low-surgical-risk patients who are also at lower risk of stroke or death perioperatively. Based on these data, it is clear that in the high-risk patient with symptomatic ICA stenosis, CAS with EPD is the treatment of choice.
The SAPPHIRE study results clearly showed that CEA carries a markedly elevated risk to asymptomatic high-risk patients. Although it appears that in asymptomatic high-surgical-risk patients CAS has similar complication rates to low-surgical-risk CEA patients, the overall benefit in the high-surgical-risk population is not clear, and therefore, a definitive statement cannot be made on the best treatment option for these patients. For some of these patients, medical therapy may be the best treatment. A clinical trial evaluating CAS vs. medical therapy in asymptomatic high-risk patients may help to answer this question.
Four recent studies have greatly clouded the issue of CEA vs. CAS in standard-surgical-risk patients. The CaRESS trial was a non-randomized study with “real-world allocation” of 397 primarily asymptomatic patients that found no statistical difference in death/stroke/MI at 30 days (4.4 % vs. 2.1 %) or 1 year (14.3 % vs. 10.9 %) with CEA compared to protected CAS, respectively [11]. The SPACE trial was a randomized comparison of CEA vs. CAS in 1,183 symptomatic patients [12]. Not surprisingly, since less than 30 % of patients were treated with embolic prevention devices contrary to the accepted standard of care, there was no difference in outcomes at 30 days (6.34 % vs. 6.84 %, p = 0.09). This study effectively replicated the results of the earlier CAVATAS trial and adds no new data except to confirm that CAS without embolic prevention devices is not safe. The most problematic study was the EVA-3S study of 527 randomized patients with symptomatic stenosis [13]. This study was conducted with poor standardization of CAS technique including the inconsistent use of dual antiplatelet therapy, incomplete use of EPD, no angiographic exclusion criteria for CAS patients but with high-risk exclusion for CEA patients, and most importantly very low CAS operator experience with some operators having performed only five cases prior to randomizing patients. Not surprisingly, the complication rates were unacceptably high in the CAS arm compared to the CEA arm, 9.6 % vs. 3.9 %, respectively. The final study was the International Carotid Stenting Study (ICSS), which was a randomized trial of CEA vs. CAS in symptomatic normal-surgical-risk patients [14]. In that trial the use of EPD was the discretion of the operator and approximately 20 % of patients were treated without an EPD. Also operators did not have to have extensive experience in performing CAS. They could be supervised by an experienced operator, and experience was defined having performed 50 stent procedures anywhere in the body, of which a minimum of 10 were required to be carotid artery procedures. That trial showed that the 30-day stroke/MI/death rate was 8.5 % with CAS and 5.1 % with CEA (p = 0.004).
The most important trial of CAS, the Carotid Revascularization Endarterectomy vs. Stent Trial (CREST), was actually started in 2000 but took 10 years to complete [15]. The initial purpose of CREST was to compare protected CAS vs. CEA in low-surgical-risk symptomatic patients, but due to slow enrollment it was expanded to include asymptomatic low-surgical-risk patients in 2005. Both the National Institutes of Health (NIH) and Guidant (now Abbott Vascular) sponsored CREST. It was designed as a 2,500-patient superiority trial with equal randomization between CEA and protected CAS using the Accunet™ embolic prevention device (“whenever feasible”) and Acculink™ carotid stenting system (Abbott Vascular Inc.). Symptomatic patients with a carotid bifurcation stenosis ≥50 % in severity on angiography, ≥70 % on ultrasonography, or ≥70 % on computed tomography angiography or magnetic resonance angiography were enrolled. Asymptomatic patients were enrolled with a stenosis ≥60 % by angiography, ≥70 % on ultrasonography, or ≥80 % on computed tomography angiography or magnetic resonance angiography if the stenosis on ultrasonography was 50–69 %. In addition to the exclusion of high-surgical-risk patients, the study excluded patients who had contraindications to CAS such as severe tortuosity, extensive or diffuse atherosclerotic disease involving the aortic arch and proximal common carotid artery, an intraluminal filling defect, ipsilateral intracranial or extracranial arterial stenosis more severe than the lesion to be treated, and occlusion or “string sign” >1 cm of the ipsilateral common or ICA. Aspirin in the CEA arm and dual antiplatelet therapy (aspirin 325 mg plus clopidogrel 75 mg or ticlopidine) in the CAS arm were mandated for at least 30 days, with aspirin in all patients thereafter.
Importantly, the study included a rigorous vetting of interventionists with a lead-in/credentialing phase of approximately 20 patients per interventionist [16]. In fact only 225 (52 %) of 429 interventionists were approved for randomization. Those who were refused outright had a median case experience of 12, range 1–56; these operators would have qualified for EVA-3S and ICSS.
The study primary endpoint consisted of the composite of any periprocedural (i.e., within 30 days) stroke, MI, or death and ipsilateral stroke within 4 years of randomization. Patients underwent independent neurological evaluations.
A total of 2,522 patients were enrolled (1,271 CAS and 1,251 CEA) with a median follow-up of 2.5 years. Approximately 5.4 % of the CAS patients and 8.8 % of the CEA patients were lost to follow-up or withdrew consent. The patients were very well matched other than a slightly higher preponderance of patients with dyslipidemia in the CEA group (85.8 % vs. 82.9 %, p = 0.05) and more smoking in the CAS group during follow-up (21.8 % vs. 13.8 %, p = 0.03). The median time to treatment from randomization was similar (6 days for CAS and 7 days for CEA). The majority of the CEA were performed under general anesthesia (90 %) and most had a patch (62.4 %) or shunt (56.7 %). The overwhelming majority of CAS were performed with embolic protection device (96.1 %), and most had pre-dilatation before stenting (67.7 %). There were a high rate (12.1 %) of CAS patients not taking dual antiplatelet agents for the full 4 weeks and a high rate of no aspirin use among CEA patients (8.9 %).
CREST showed no difference in the primary study endpoint of stroke/MI/death within 30 days [CAS 5.2 ± 0.6 vs. CEA 4.5 ± 0.6, hazard ratio (HR) 1.18 (0.82 to 1.68), p = 0.38] or up to 4 years [CAS 7.2 ± 0.8 vs. CEA 6.8 ± 0.8, HR 1.11 (0.81 to 1.51), p = 0.51]. There was no difference in the individual endpoint of periprocedural death (CAS 0.7 ± 0.2 vs. CEA 0.3 ± 0.2, p = 0.18), but there was a difference for any periprocedural stroke [CAS 4.1 ± 0.6 vs. CEA 2.3 ± 0.4, HR 1.79 (1.14 to 2.82), p = 0.01] or MI [CAS 1.1 ± 0.3 vs. CEA 2.3 ± 0.4, HR 0.50 (0.26 to 0.94), p = 0.03]. Following the periprocedural period, the incidence of ipsilateral stroke was similar (CEA 2.4 % vs. CAS 2.0 %, p = 0.85) as was the risk of fatal stroke (CAS N = 7 vs. CEA N = 6). There was no difference in the primary endpoint during the perioperative period among symptomatic patients [CAS 6.7 % vs. CEA 5.4 %, HR 1.26 (0.81–1.96)] or asymptomatic patients [CAS 3.5 % vs. CEA 3.6 %, HR 1.02 (0.55 to 1.86)]. There was no interaction between sex and symptomatic status and treatment effect although there was an interaction between age and efficacy (p = 0.02). The crossover point for age was at approximately 70 years with greater efficacy with CAS in younger patients and greater efficacy with CEA for older patients. The risk of cranial neuropathy was significantly higher in the CEA group (0.3 % vs. 4.7 %).
The CREST trial, the first trial to compare protected CAS vs. CEA in standard-surgical-risk symptomatic and asymptomatic patients, has shown that both procedures are equivalent in perioperative morbidity and mortality as well as long-term stroke prevention. There was a clear difference however in the risk of perioperative stroke with an increased risk in the endovascular group; most of these strokes were minor.
Conversely, there was a higher risk of MI in the surgical group. Importantly, the 30-day outcomes were similar for both procedures to the accepted thresholds for clinical benefit compared to medical therapy, i.e., <6 % for symptomatic (6 % stroke/death with CREST CAS and 3.2 % for CREST CEA) and <3 % for asymptomatic patients (in CREST the rate of 30-day stroke/death with CAS was 2.3 % for ACAS-eligible patients). It is important to note that the stroke rate declined over time in the CREST trial and if the results from the latter half of the study were utilized there would have been no difference in stroke rate with CEA. This highlights the importance of case experience and improved patient selection.
These results contradict the results of the three randomized European trials discussed earlier [12–14]. The EVA-3S, ICSS, and SPACE trial results have greatly reduced the enthusiasm for CAS and have blocked the expansion of CMS coverage for CAS. Taken at face value, this cooling of enthusiasm is understandable; however, all randomized trials are not created equal, and the results of these trials have to be reconciled with those of the CREST trial and the registries. As with the early trials of CEA, the differences in outcomes have to do with patient and operator selection as well as procedural techniques. Several editorials have highlighted the many limitations of these trials and (Table 4.2) highlighted the differences and possible explanations for the differing results.
Table 4.2
Comparison of CAS trial protocols
SAPPHIRE | CAVATAS | EVA-3S | SPACE | ICS | CREST | ||
|---|---|---|---|---|---|---|---|
Stenting | ✓ | X | ✓ | ✓ | ✓ | ✓ | |
EPD mandatory | ✓ | X | ✓ | X | X | ✓ | |
Experienced operators | ✓ | X | X | X | X | ✓ | |
Dual antiplatelet Tx | ✓ | X | X | ✓ | ✓? | ✓ | |
Angiographic exclusions | ✓ | Xa | X | X | ✓ | ✓ | |
Independent neurologist | ✓ | ✓?b | ✓ | ✓ | ✓?b | ✓ | |
No general anesthesia | ✓? | ✓? | X | X | ✓? | √ | |
Symptomatic definition | 3 months | >6 months | 4 months | 6 months | 12 months | 6 months | |
Angiography in all patients | X | X | X | X | X | X |
Carotid artery stenting can be performed in nearly all patients (98.6 % in SAPPHIRE). The remainder may be better treated medically or surgically (Table 4.3 lists the relative contraindications to CAS). There are two groups of patients for whom the ideal therapy is unknown. The first are patients who have an intraluminal filling defect (i.e., thrombus) within the stenotic segment. In NASCET these patients had an 18–22 % risk of perioperative stroke. Such patients have not been enrolled in the trials of CAS, and it is generally agreed that they may also have a high stroke risk with CAS. In these patients a short period of anticoagulation may be appropriate followed by CEA or CAS when the thrombus resolves. The other and far larger group of patients is those over the age of 80. These patients were mostly excluded from the trials of CEA and are known to have a higher perioperative complication rate than younger patients. With CAS, however, the elderly appear to have a higher rate of complications [9, 15]. In the CREST trial lead-in phase (N = 1,246), octogenarians had a 12.1 % 30-day stroke/death rate. At this time therefore a conclusion cannot be drawn on the optimal treatment for octogenarians, but medical therapy alone should be given strong consideration since CEA also carries a nearly 12 % complication rate in those over age 75.
Table 4.3
Contraindications to CAS
Severe vascular tortuosity |
Poor arterial access |
Coagulation or platelet disorder that precludes adequate antithrombotic agent use |
Severe, circumferential target lesion calcification |
Target lesion length >15 mm |
The long-term patency of the two procedures appears comparable. In a 2-year follow-up period of the CREST population, severe restenosis and occlusion were infrequent (approximately 6 %), and rates were similar between the CEA and CAS groups [17].
There are several issues that have not yet been addressed by the published results, e.g., newer embolic prevention devices and stents are available; might one or several of them be associated with lower stroke rates? There have been debates about the type of stent used with some suggestion that closed-cell stents are associated with lower periprocedural stroke [18]. Proximal occlusion EPDs have also been touted as superior at stroke prevention, but they have some limitations such as larger bore femoral access and increased probability of intolerance to the occlusion of antegrade flow. In a large single-center registry of 1,300 patients treated with proximal occlusion, the 30-day stroke/death rate was 1.38 % with independent neurological assessment at 24 h and 30 days [19]. In a meta-analysis of 2,397 patients from 6 independent databases, Bersin et al. found that the composite of stroke/MI/death occurred in 2.25 % of cases [20]. So while these data are tantalizing, there are no definitive randomized trial data that show one type of EPD device is superior to another. Clinicians should, in the author’s opinion, become familiar with one or two devices/approaches and use them exclusively until there are definitive data on superiority of one approach or device over another.
Also studies on which other factors predict complications are also needed. One such study pooled data on 2,104 patients from four Cordis Endovascular Inc.-sponsored registries [21]. In that analysis the median age was 74 years (24 % >80 years), 36 % were female and 24.2 % of the patients were symptomatic. Multivariate predictors of the 4.2 % neurological deaths or strokes included older age (continuous), African-American race, angiographically visible thrombus in symptomatic patients, procedural use of glycoprotein IIb/IIIa inhibitor, procedural transient ischemic attack, final residual stenosis >30 %, and periprocedural use of protamine or vasopressors.
Of particular interest is that in symptomatic patients, the risk of a neurological event declined with increasing time between incident event and CAS [22]. The issue of timing of CAS in symptomatic patients has been a major unanswered question. The vast clinical experience with CEA has clearly shown that earlier intervention is superior to delayed intervention in preventing recurrent ischemic stroke but comes at the cost of increased intracerebral hemorrhage [23]. The fear of reperfusion/hyperperfusion ICH is perhaps more justified with CAS since patients are treated with dual antiplatelet agents and are theoretically more likely to have ICH. The available literature has not corroborated those fears. To the contrary with adequate blood pressure control, the risk of the hyperperfusion syndrome can be mitigated [24, 25]. Early CAS can also be performed safely in selected patients [26].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








