Target
Spatial resolution (mm)
Temporal resolution (S)
EEG/MEG
Neuronal current
10
0.001
fMRI
Regional changes in blood flow
4–5
1–10
PET
Oxygen consumption, glucose metabolism etc.
5–10
30–1,000
SPECT
Changes in blood flow change
10
60–1,000
NIRS (near-infrared spectroscopy)
Changes in blood flow change
8–
0.01–
32.2 Transmission of Pain Signals at the Spinal Cord Level and Efforts to Visualize This Phenomenon
Pain impulses traveling to the spinal cord from the periphery via primary afferent fibers are transmitted to spinothalamic tract cells, which are a type of cell in the spinal dorsal horn, and subsequently transmitted as pain signals to the brain via the ventrolateral pathway on the opposite side. In contrast to the brain, which ultimately experiences pain through various modulations, the spinal cord receives fewer of these modulations and tends to directly reflect nerve response to outside stimulation. For this reason, a method of inserting an electrode directly into the dorsal horn of the spinal cord to record neural activities of spinothalamic tract cells has been widely applied in animal studies, and this particular method has been used to elucidate mechanisms of pain exacerbation such as spinal cord sensitization and to evaluate drug treatments. However, direct insertion of a needle into the spinal cord can be difficult in humans, so studies using fMRI and PET have been progressing as an alternative.
32.2.1 Spinal fMRI Studies in Healthy Subjects
Although research to evaluate spinal cord function has only just scratched the surface, our group has been proceeding with the analysis of spinal cord activity induced by a mechanical pain stimulation device, using the blood oxygen level-dependent (BOLD) method. Functional tasks to elicit spinal cord activity are conducted using our original plastic mechanical pain stimulation device. Functional MRI using echo planar imaging (EPI) was conducted on healthy subjects administered painful stimuli, BOLD signal changes were recorded with a 3-T MRI scanner, and analysis was undertaken using statistical parametric mapping (SPM) (www.fil.ion.ucl.ac.uk/spm/). Since large mechanical movements occur in the cervical vertebrae due to breathing and swallowing, we created and utilized a cervical collar to suppress these movements during imaging. As a result, we were able to visualize the inner spinal cord area that was activated in response to the nociceptive stimuli applied in the study (Fig. 32.1). However, variations in activation were seen between subjects. Before clinical application can be achieved, the function of a spinal cord that has flattened and shrunk in size due to spinal cord compression will need to be assessed in the future. We therefore believe that further technical improvements in the method are essential [1].
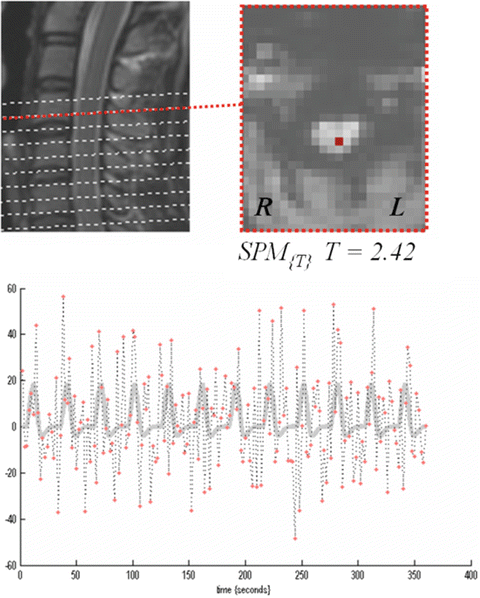

Fig. 32.1
Time-dependent changes in BOLD signal and site of spinal cord activity
32.2.2 Spinal PET Imaging in Patients with Neuropathic Pain in the Spinal Cord
PET and SPECT are imaging modalities that detect gamma-ray distributions emitted from drugs administered to the body, allowing the creation of tomographic images. PET is a method in which radioisotopes that emit positrons are injected into the body to produce gamma-rays that arise during pair annihilation between positron and electrons in the body and is performed with a sensor set up to enclose the body. Uchida et al. investigated glucose metabolism in the cervical spinal cord using fusion images of 3-T MRI and 18 fludeoxyglucose (FDG)-PET in patients with compressive cervical myelopathy [2]. We applied this PET technology to assess whether functional changes in the spinal cord occur in patients with neuropathic pain in the spinal cord. We observed sites of FDG uptake in the spinal cord and were able to obtain an image that suggests enhanced neural activity due to pain. However, some aspects of the results, such as resolution, remain to be optimized in the future (Fig. 32.2).
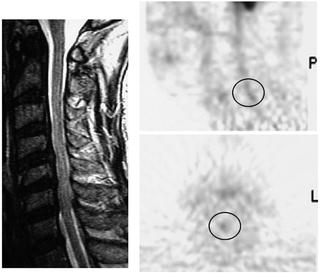

Fig. 32.2
Cervical spinal FDG PET (left cervical radiculopathy, 60’s years, male). In collaboration with the Department of Orthopedic Surgery at the University of Fukui
Previous findings from functional neuroimaging of the spinal cord suggest that both fMRI and PET will be able to be applied to humans in the future as methods to evaluate pain. However, further investigations are necessary before these approaches can be used in clinical cases such as cervical myelopathy.
32.3 Functional Brain Imaging and Musculoskeletal Pain
Previous studies of functional brain imaging in healthy subjects have revealed that the pain signals that ascend through the spinal cord are primarily projected to the region of cerebral cortex called the “pain matrix,” an area related to pain. This region includes the first and second somatosensory areas (S1 and S2), insular cortex, anterior cingulate gyrus, and medial prefrontal cortex via the thalamus. However, in clinical settings where patients with chronic pain require pain evaluation, plastic changes and sensitization of the nervous system may be induced, and a transmission pathway may not necessarily be experienced in a similar manner to that in healthy subjects with acute pain. Here, we show functional neuroimaging findings from previous studies of chronic musculoskeletal pain, focusing on the latest research outcomes.
32.3.1 Brain Activity in the Thalamus and Acute and Chronic Pain
The thalamus is said to be the center of senses and is where pain transmission from the aforementioned spinothalamic tract terminates. The thalamus has therefore been a major focus in pain research for many years, and many detailed analyses have been conducted.
In studies that have investigated neural activity in the thalamus using PET or SPECT, neural activity in the thalamus is known to be enhanced in acute pain pathology. However, several reports on chronic pain have indicated a decrease in thalamic activity on the side contralateral to where the transmission of stimulation primarily occurs in pain conditions [3, 4]. Such decreases in contralateral thalamic activity are also known to be exacerbated with longer disease staging, and this is thought to imply plastic changes in the central nervous system [5]. Several causes have been postulated for such changes: (1) persistent and chronic pain activates the inhibitory system to suppress thalamic function; (2) the cortical sensory area has fallen into a persistently and easily excitable state due to chronic input from the thalamus, and even small pain signal inputs from the thalamus equate to the recognition of pain; or (3) enhancement of blood flow is not required because synaptic transmission at the thalamus has become extremely efficient. However, several reports on chronic pain have indicated enhancement of thalamic activity on the contralateral side, so further mechanistic clarification is necessary.
32.3.2 Evaluation of Induced Pain in the Brain Using fMRI
Functional MRI offers not only good spatial resolution but also moderate temporal resolution and is appropriate for the evaluation of brain activity when and where pain is inflicted.
32.3.2.1 Brain Activity when Pain Stimulation Is Inflicted (Comparison Between Healthy Subjects and Patients with Neuropathic Pain)
With the objective of investigating brain activity in patients with allodynia who experience pain from stimuli that would not normally provoke pain, Ikemoto et al. captured the brain activity of such patients using fMRI following mechanical stimulation (non-nociceptive stimulation) that would not normally provoke pain at the site in which they experience pain and compared these results to when healthy subjects were provoked with mechanical nociceptive and non-nociceptive stimulation [6]. Results indicated that when healthy subjects were provoked with mechanical nociceptive stimulation, enhancement of activity in the thalamus, somatosensory areas (S1 and S2), cingulate gyrus, and cerebellum was detected. On the other hand, even though stronger pain was observed in patients on the VAS compared to healthy subjects, activity in the thalamus, which is the center for pain from the periphery, was not detected. Activities are now known to arise in S1, S2, cingulate gyrus (primarily anterior cingulate gyrus), as well as the motor cortex and supplementary motor cortex (Fig. 32.3) [7]. Peyron et al. [8] conducted a study of brain activity upon inflicting mechanical stimulation that would not normally provoke pain on the affected side as well as the unaffected side in patients with allodynia and reported results similar to those reported by Ikemoto et al.
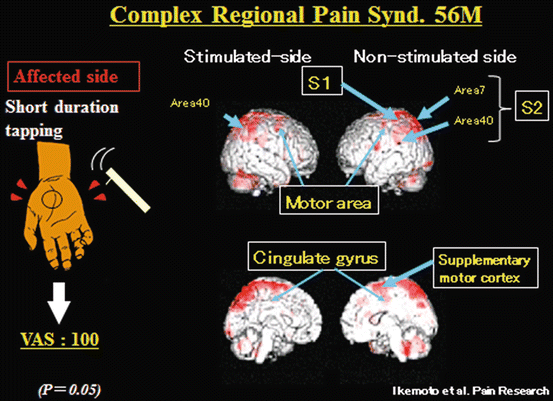

Fig. 32.3
In CRPS patient, even though stronger pain was observed on the VAS , activity in the thalamus, which is the center for pain from the periphery, was not detected. Activities are now known to arise in S1, S2, cingulate gyrus (primarily anterior cingulate gyrus), as well as the motor cortex and supplementary motor cortex
32.3.2.2 Brain Activity Sites During Reproduction of Joint Pain and Low Back Pain Induced by External Stimulation
Even in chronic pain that is generally considered to represent a repetition of acute pain in conditions such as osteoarthritis of the knee, pain while walking and localized tenderness are observed. Since pain is confined in these patients, attempts have been made to stimulate the tender sites with a special device. As a result, fMRI revealed that patients with osteoarthritis of the knee exhibit significant neural activity in areas of the pain matrix such as bilateral thalami, S2, insula, supplementary motor cortex, and anterior cingulate gyrus, as well as pain when pressure stimulation is applied to tender sites [9].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








