Fig. 19.1
Graph showing proportions of patients without local recurrence stratified by tumor volume. The study group was divided into four equal quartiles of volume. Plot obtained from the multivariate Cox proportional hazards model analysis (From Patel et al. (2010). Used with permission)
Whole-Brain Radiotherapy
Shortly after the discovery of x-rays in 1895, radiation was used to treat cancers. Over the past 100 years, radiation therapy has evolved into a multifaceted form of treatment. For many years, WBRT was used as the primary treatment modality for brain metastasis. Patients treated with WBRT alone exhibited high rates of recurrence. In 1990, Patchell et al. (1990) demonstrated that surgical excision followed by WBRT decreased the local recurrence rate to 20% from 52% in patients treated with WBRT alone. Since then, surgery and SRS have been established as the primary treatment modalities for most brain metastases, but WBRT has played an indispensible role as an adjuvant, as it is thought to help prevent both local and distant recurrence. The landmark study by Patchell et al. (1990) prospectively demonstrated a reduction in local recurrence from 46 to 10% with postoperative WBRT (Patchell et al. 1998). Similarly, Aoyama et al. (2006) found that WBRT after SRS decreased the rate of local recurrence from 76.4 to 46.8%. The European Organization for Research and Treatment of Cancer (EORTC) conducted a randomized trial with 359 patients to evaluate whether WBRT increases intracranial tumor control after surgery or SRS in patients with one to three brain metastases (Kocher et al. 2011). After 2 years, adjuvant WBRT had reduced local recurrence from 59 to 27% postoperatively and from 42 to 23% after SRS (Kocher et al. 2011). Despite this evidence, some clinicians withhold WBRT after resection of single brain metastases. However, McPherson et al. (2010) conducted a retrospective review of 358 patients with newly diagnosed single brain metastases who were treated by resection with or without adjuvant WBRT. They found that adjuvant WBRT significantly reduced both local and distant recurrence, with this difference being particularly significant in lesions >3 cm in maximal diameter (Fig. 19.2).
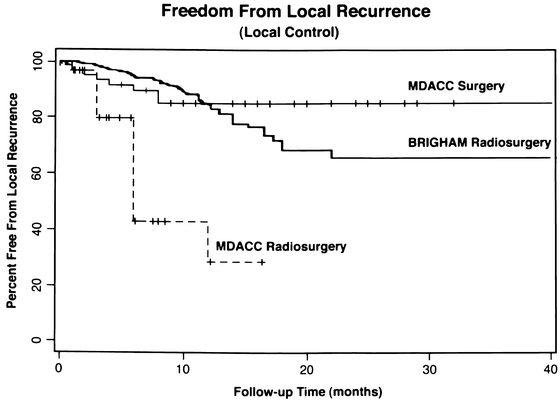

Fig. 19.2
Freedom from local recurrence (local control) of brain metastases with radiosurgery and with surgery. Curve showing time from radiosurgical treatment to local failure (BRIGHAM Radiosurgery) in 42 patients treated at Brigham and Women’s hospital (Mehta et al. 1992) superimposed on curves showing time from surgical resection (MDACC Surgery) to local recurrence in 62 patients and time to local failure in 31 patients radiosurgically treated (MDACC Radiosurgery) at The University of Texas M.D. Anderson Cancer Center (Bindal et al. 1996) (From Sawaya 1999. Used with permission)
Primary Treatment Modality
Both surgery and SRS are used to treat brain metastases, usually with adjuvant WBRT. The ideal treatment regimen varies based on the tumor histology, size of the lesion, patient’s clinical status and presentation. For example, small cell lung cancer is so radiosensitive that it is commonly treated with WBRT. Germ cell tumors are treated with chemotherapy because of their exquisite chemosensitivity. Surgery is favored when treating radioresistant tumors, such as melanoma and renal cell carcinoma.
In addition to providing histologic diagnosis and immediate relief of symptoms from mass effect, surgical excision has been shown to provide excellent local control, especially when compared with WBRT alone, as in the study by Patchell et al. (1990). Since then, surgery has played a central role in the treatment of brain metastasis. The rate of local recurrence after surgery alone has been reported to range from 15 to 46% without adjuvant therapy (Nieder et al. 2007; Patchell et al. 1998; Patel et al. 2010; Vecht et al. 1993). When reviewing our experience in patients who underwent surgery alone for a single, previously untreated brain metastasis, we identified two factors that independently affected local recurrence (Patel et al. 2010). In addition to tumor volume, the method of resection significantly influenced local recurrence. Patients undergoing piecemeal resection had a risk of developing local recurrence that was 1.7 times greater than those with tumors excised en bloc. Based on the multivariate analysis, this risk was negated in tumors larger than 9.71 cm3; however, for patients with smaller tumors, those with tumors resected by piecemeal methodology had a 2.7 times greater risk for recurrence than those with tumors resected en bloc.
SRS was first developed by Lars Leksell at the Karolisnka Institute (Leksell 1951). Subsequently, it has emerged as an alternative to surgery in the treatment of brain metastasis. A multi-institutional, randomized trial compared patients with one to three brain metastases smaller than 4 cm in maximal diameter that were treated either by WBRT alone or by WBRT followed by an SRS boost and demonstrated a reduction in local recurrence from 29 to 18% in patients receiving the SRS boost (Andrews et al. 2004).
Although there is no question that surgery or SRS in addition to WBRT reduces local recurrence compared with WBRT alone, whether surgery or SRS is the optimal treatment modality remains controversial. It is accepted that SRS is the treatment of choice for patients whose medical condition precludes them from being surgical candidates or who decline surgery. Lesions that are less than 1 cm in maximal diameter and surgically inaccessible (e.g., in deep cortex that controls eloquent functions) are also amenable to SRS. Similarly, surgery is usually the clear choice for patients diagnosed with symptoms of mass effect. Historically, the remainder of the lesions have been stratified as those either larger or smaller than 3 cm in maximal diameter. As outlined above, for large lesions, SRS is associated with a higher rate of recurrence than surgery. Chang et al. (2003) retrospectively reviewed the records of 135 patients who underwent SRS and found that lesions that were greater than 1 cm in maximal diameter had only a 56% local control rate. Thus, the 3 cm cutoff that is commonly used requires further study in a prospective manner.
Shinoura et al. (2002) compared patients who underwent surgery plus postoperative WBRT with those who underwent SRS alone and found that the mean times to recurrence were 25 and 7.2 months, respectively. Muacevic et al. (2008) published the results of a randomized trial (prematurely terminated owing to poor patient accrual) comparing surgery plus WBRT with SRS alone. They reported no difference in local control between these two groups. But both of these studies had approximately 30 patients in each arm and did not directly compare surgery and SRS because the SRS group did not receive WBRT. A prospective study at M.D. Anderson with both randomized and nonrandomized arms sought to compare patients receiving surgery or SRS alone for single brain metatastasis (Lang et al. 2008). The randomized arm had 30 patients in the surgical group and 29 patients in the SRS group; the nonrandomized arm had 89 and 66 patients, respectively. A multivariate analysis to eliminate confounding covariables demonstrated that patients in the SRS-alone group had significantly more local recurrences than those undergoing surgical excision. As expected, distant recurrence rates were the same in both groups.
It is difficult to compare the two treatments because of differences in the way recurrence is defined. Published values of local tumor control after SRS permit an increase in tumor size of up to 25%. It is important to keep this in mind when reviewing the available data and comparing these two treatment modalities, as this can overstate the level of local tumor control in patients receiving SRS.
Conclusion
There have been very few studies specifically addressing what factors affect the risk of local recurrence of brain metastasis; thus, most of the aforementioned information has been assembled from the secondary endpoints of various prospective and retrospective studies. Nevertheless, these studies represent an incredible body of work compiled over the past few decades. Table 19.1 summarizes the published class I studies to date. The factors that have been shown to affect local control can be divided into two categories: those that are intrinsic to the lesion and those that are treatment related.
Table 19.1
Summary of all class I studies evaluating treatment of brain metastasis
Study | Groups | Local control | Distant control | Median survival (months) |
|---|---|---|---|---|
Patchell et al. (1990) | WBRT (n = 23) | 48%a | 87%a | 3.5a |
Surgery + WBRT (n = 25) | 80%a | 80%a | 9.2a | |
Vecht et al. (1993) | WBRT (n = 31) | N/A | N/A | 6a |
Surgery + WBRT (n = 32) | N/A | N/A | 10a
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|



