Fig. 9.1
Immune system and the environmental impact on the brain
Post-Traumatic Stress Disorder
Several studies have evaluated levels of cortisol in children exposed to trauma (Johnson et al. 2013; Shonkoff et al. 2012; McGowan et al. 2009; Carrion et al. 2002; McCrory et al. 2010). In a study of maltreated children with PTSD, 24 h levels of cortisol were increased (De Bellis et al. 2010). Goenjian et al. (1996) reported lower levels of basal cortisol in children 5 years after an earthquake. Gunnar et al. (2001) reported higher diurnal levels of cortisol after 6.5 years post trauma. In a study of girls who had attempted suicide. Girls with early sexual abuse had lower salivary levels of morning cortisol were reported by King et al. (2001). Carrion et al. reported higher levels of cortisol in 33 youth ages 10–16 and, and combined with changes in brain MRI. Youth with post-traumatic stress symptoms (PTSS) (n = 45) had decreased total brain tissue in comparison with healthy controls (n = 15). There was a significant negative association between pre-bedtime cortisol levels and left ventral prefrontal cortex gray volumes (Carrion et al. 2010a). Studies in adults report similar findings in patients with PTSD (Vythilingam et al. 2002). Some developmental differences have been reported in levels of IL-6 in children with PTSD. In both adults and children, IL-6 is elevated in individuals with PTSD. In adults the levels of IL-6 continue to be elevated chronically, while in children the IL-6 levels tend to normalize over time (Pervanidou et al. 2007).
The pathophysiological mechanisms underlying the development of neurobiological consequences of childhood trauma have not been fully elucidated. Disruption of the HPA axis with subsequent cortisol induced toxicity has been proposed as one mechanism for neuroimaging differences (Bremner 1999, 2006; McCrory et al. 2010; Marin et al. 2007). Another proposed mechanism is the induction of an inflammatory response leading to neuronal injury (Miller et al. 2009c; Herberth et al. 2008). A more recent and novel hypothesis is that brain injury could be caused by disruption of the BBB leading to influx of inflammatory cells, as well as toxic compounds which could lead to damage to several brain areas (Neuwelt et al. 2011; Falcone et al. 2015; Andrews and Neises 2012; Angelov et al. 2009; Cucullo et al. 2011; Esposito et al. 2001, 2002, 2003; Janigro 2012; Marchi et al. 2004; Rapoport et al. 1972; Reiber 2001). The last hypothesis has not been investigated as thoroughly due to the of lack of easily available tools to measure the integrity of the BBB until recently (Cucullo et al. 2011; Marchi et al. 2003, 2004; Kapural et al. 2002). Studies in soldiers before and after a parachute jump describe how chronic stress can activate peripheral blood mononuclear cells (PBMC) and increase several inflammatory markers such as nerve growth factor, cortisol, and adrenocorticotropic hormone (Aloe et al. 1994).
Animal model studies suggest that activation of PBMCs can potentially exacerbate CNS damage via inflammation (Ajmo et al. 2008). Lewitus et al. (2008) described the role of T lymphocyte infiltration at the level of the BBB and an increase of intercellular adhesion molecules (ICAM-1) on the BBB. ICAM-1 has also been associated with increased migration of inflammatory cells to the BBB. de Pablos et al. (2006) described increased levels of inflammatory markers in animals secondary to stress that were associated with a decreased number of neurons. A leaky BBB can permit further accumulation of PBMCs, that potentially will produce more inflammation and production of matrix metalloproteinases, further increasing the BBB permeability (Rosenberg 2009).
Studies in animal models have demonstrated that stressful situations, such as immobilization, forced swimming, and training in a water maze produced BBB breakdown (Sharma 2011; Sharma et al. 1990, 1991, 1992, 1995, 2007a, b, 2009, 2010, 2012; Sharma and Dey 1981, 1984, 1986, 1988; Sharma and Johanson 2007). Sharma et al. concluded after multiple investigations on the role of the BBB under stress that specific opening of the BBB occurs under different kinds of stressful situations: “emotional stress,” “heat stress,” “cold stress,” “swimming stress,” “immobilization stress,” and “surgical stress.” Adaptation or resilience is what makes a difference between how these stressors continue to impact the brain overtime. After the initial stress the glucocorticoid system is activated, increasing levels of ACTH that help the organism cope with the increased demands of stress (Sharma 2011; Sharma et al. 1990, 1991, 1992, 1995, 2007b, 2009, 2010, 2012, 2013; Sharma and Dey 1981, 1984, 1986, 1988; Sharma and Johanson 2007; Sharma and Cervos-Navarro 1991). Catecholamines (adrenaline, noradrenaline) are released and alter the BBB function. The initial mild to moderate stress can be an adaptive response that helps the organism to cope with the increasing demands while prolonged, chronic, severe stress can lead to brain changes in selective areas of the brain and potentially later on contributing to the development of PTSD (Shalev et al. 2009) (Fig. 9.2).
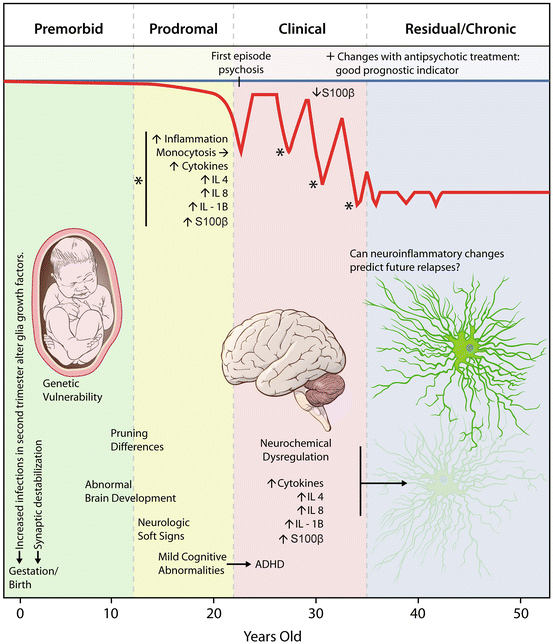

Fig. 9.2
Inflammatory changes in patients with psychosis- Please move the figure next to the psychosis section
Autism
There is very strong evidence for the role of inflammation in patients with autism. In children with autism spectrum disorder (ASD), 40 studies reported data on PIM. Al-Ayadhi and Mostafa (2012) in a study of 45 patients with ASD reported increases in levels of IL-17A in a group of ASD patients compared to controls. Manzardo et al. (2012) described 99 ASD patients vs. 40 controls and found decreased levels of IL-1alpha and IL-6 in ASD compared to controls. In a very small sample of ten patients, Bent et al. reported no changes in several cytokines but no control group was available. Also, there was no data on the gender of the participants (Bent et al. 2012). In a study in Saudi Arabia comparing 20 male patients with ASD with 19 controls, levels of IFN gamma were significantly elevated in patients with ASD (El-Ansary et al. 2011). Jyonuchi et al. identified elevated levels of IL-1, IL-6, IL12p40, IL-17, IL-23, IFN gamma, and TNF alpha after stimulation of PBMC. In study of 47 patients with ASD vs. 37 controls, there was decreased production of IL-1, IL-6, IL12p40, IL17, IL23, IFN gamma, and TNF alpha in ASD patients vs. controls (Jyonouchi et al. 2012). In a Brazilian study of 24 patients with ASD vs. 24 controls, levels of IFN gamma were increased in patients with ASD (Tostes et al. 2012). Ashwood et al. reported increased levels of IFN gamma and TNF alpha in a group of 66 patients with ASD vs. 77 controls after stimulation of PBMC. No changes were observed in IL-1B, IL-2, IL-4, IL-5, IL-6, IL8, IL-10, and IL-13 (Ashwood et al. 2011a). One study included 97 patients with ASD, 87 controls, and 39 developmentally delayed patients after plasma multiplex luminex, analysis of levels of IL-1B, IL6, IL-8, IL-12p40 were increased in patients with ASD (Ashwood et al. 2011b). In Saudi Arabia 25 males ages 4–12 vs. 16 controls, there were decreased levels of IL-6 and TNF alpha in ASD. In a study of 6 patients with ASD vs. 6 controls, levels of TNF alpha and IL-6 were increased in the ASD group (El-Ansary et al. 2011). Jyonouchi et al. (2011) reported decreased production of IL-6, IL beta, IL 12, IL 23, IL 10, sTNFRII, and TGF B in 30 ASD patients compared to 28 controls after stimulation of PBMC. Suzuki et al. (2011) in Japan reported levels of 48 plasma inflammatory markers increased in a group of 28 ASD patients vs. 28 controls. While Tobiasova in a study of 21 ASD patients vs. 15 controls, reported serum of IL-13 decreased in ASD patients. ASD patients (n = 77) were treated with risperidone and no effect was observed in the inflammatory markers vs. placebo in ASD patients (Tobiasova et al. 2011). In a postmortem study evaluating expression of IL-6, in the frozen cerebellum of 6 patients of ASD vs 6 controls, increased expression of IL-6 in the cerebellum in ASD was identified in patients compared to controls (Wei et al. 2011). Patients with ASD and fragile X in ASD with fragile X (n = 40) vs. patients with fragile X and no ASD (n = 64) vs. 19 controls, levels of IL1 alpha and IL12p40 were reported increased in patients with ASD/fragile X patients vs. controls (Ashwood et al. 2010). In a group of 17 patients with ASD vs. 16 controls, levels of IL-1 B, IL-6, TNF alpha, were increased after stimulation of the Peripheral Blood Monocytes (PBMC) (Enstrom et al. 2010). In a study of 19 patients with ASD, levels of IL-6 were decreased if their PBMC were treated with BDE-47. Levels of IL-8 were increased in the ASD patients (Ashwood et al. 2009). IL-6, TNF alpha, and IFN gamma were increased in the frontal cortex of ASD patients postmortem compared to controls (Li et al. 2009). Onore et al. (2009) reported decreased IL-23 in 34 patients with ASD. Multiple infections and food allergies are frequently reported in children with ASD. Jyonouchi et al. (2008) studied levels of inflammatory markers in this group and found decreased IL-1 B and IL10 in ASD and increased IL-23 in ASD. Chez et al. evaluated levels of CSF TNF alpha in ASD and found levels increased in 10 patients. No controls were recruited in this study (Chez et al. 2007).
Hyperbaric oxygen treatment is an alternative treatment (non-FDA approved) for ASD. In a group of 18 patients with ASD without controls, levels of high sensitive CRP were elevated in three patients with ASD; after treatment with hyperbaric oxygen, the levels of CRP normalized (Rossignol et al. 2007). Molloy et al. (2006) reported elevated levels of IL-4, IL-5, and IL-13 in 20 patients with ASD vs. 20 controls. Jyonouchi et al. (2005a) analyzed the impact of gastro intestinal (GI) symptoms in children with ASD and the impact on inflammatory markers compared to controls and found increases in TNF alpha and IL-12 in ASD, but without impact on the GI symptoms. The same group in a dietary intervention study evaluated the levels of TNF alpha after stimulation of PBMC, patients with ASD had increased TNF alpha. The restricted diet had no impact on other inflammatory markers (Jyonouchi et al. 2005b). Two studies evaluated intestinal biopsy samples in children with ASD, De Felice found no difference in inflammatory markers in patients vs. controls, and Ashwood found increased CD3 TNF, CD3 IL2, CD3 IFN gamma, and CD3 IL 4 in patients with ASD (Ashwood et al. 2004). Zimmerman et al. (2005) evaluated levels of CSF and serum of several inflammatory markers in patients with ASD and found decreased levels of IL1Ra and IL 6 in patients vs. controls. In a study evaluating brain tissue from area-Brodman 19 and cerebellar cortex, in 9 males with ASD vs. 9 male controls, the inflammatory pathway gene dysregulation was significant in patients with ASD (Ginsberg et al. 2012).
Looking at these studies, there is consistent evidence for increased TNF alpha in patients with ASD. Contradictory findings in patients with ASD on the levels of IL-6. One of the important anti-inflammatory cytokines was decreased in nine studies of children with ASD (Mitchell and Goldstein 2014).
Different animal models of maternal inflammation during the first trimester have been proposed in the pathophysiology of autism (Smith et al. 2007; Patterson 2002). The role of infections during pregnancy has been extensively discussed, especially from the perspective on how early inflammatory changes can potentially alter neural circuitry and pruning. Maternal immune activation can later cause cerebellar abnormalities in the offspring of mice. Similar characteristics are shared in the immunological animal models of schizophrenia and autism (Smith et al. 2007; Zuckerman and Weiner 2005; Meyer et al. 2005, 2006a, b, 2008a, b; Romero et al. 2007; Dammann and Leviton 1997; Zuckerman et al. 2003; Depino 2006; Gilmore et al. 2004; Nawa and Takei 2006).
Obsessive Compulsive Disorder, Tourette’s Disorder, and PANDAS
Several studies report increased inflammatory markers in patients with anxiety disorders. They reported an important relationship between the development of de-novo obsessive symptoms or tic disorder after the exposure to upper respiratory infection (Mitchell and Goldstein 2014).
Tourette Disorder
A study in China of 40 patients ages 4–19 with Tourette disorder (TD) compared to 40 controls reported elevated levels of IL-1, IL-6, and IL-17, in TD (Cheng et al. 2012). In a German study of 15 patients with TD vs. 10 controls, there were increased levels of TNF alpha and IL-1Ra in controls when compared to patients with TD (Matz et al. 2012). There was no differences in polymorphisms of children with TD compared to controls (Liu et al. 2011a, b; Li et al. 2013). Bos-Veneman et al. in 66 patients with TD measured serum levels of IL-2, IL-4, IL-5, IL-10, IL-12, IFN-gamma, and TNF-alpha to compare to 71 controls. There were no significant differences in serum concentrations in patients with TD (Bos-Veneman et al. 2010). In Taiwan a study of 159 patients of TD and 175 controls, polymorphisms in the IL1RN were associated with TD (OR of 7.65) (Chou et al. 2010).
Obsessive Compulsive Disorder
One study evaluated plasma levels of IL-1B, IL-2, IL-6, IL-12, and TNF alpha, in three groups: patients with TD (n = 32), patients with TD and OCD (n = 17) and controls (n = 14). Levels of IL-12 were increased in TD-OCD group compared to controls, and to those with TD only. In a similar study Leckman et al. reported increased levels of IL-12 and TNF alpha in patients with TD + OCD (n = 34) vs controls (n = 31). Levels of IL-12 and TNF were increased during worsening symptoms of TD + OCD (Leckman et al. 2005). Levels of TNF alpha correlated inversely with obsessive compulsive disorder (OCD) symptom severity (Gabbay et al. 2009c). To the contrary levels of TNF alpha correlated inversely with OCD. Luo et al. evaluated serum levels of high sensitive CRP (hsCRP) in a group of 47 TD + OCD patients and 19 controls. There were no differences in the levels of hsCRP between the groups or after worsening of OCD symptoms (Luo et al. 2004).
Pediatric Autoimmune Neuropsychiatric Disorder
A 1990 study by the NIMH in youth with OCD described a group of children with severe symptoms and sudden onset of psychopathology (Swedo et al. 2004). Researchers noted an association between the sudden onset of symptoms of obsessive thoughts and other neuropsychiatric symptoms: irritability, anxiety, mood lability, and regression. Symptoms occurred chronologically near infections from influenza, varicella, streptococcal infections, Lyme disease, and Mycoplasma infections. Streptococcal infections were the ones most frequently recognized in association with the neuropsychiatric symptoms. In 2010 the diagnosis changed from PANDAS to PANS Pediatric Acute Onset neuropsychiatric syndrome (Singer et al. 2012). Different treatments have been recommended to treat the acute symptoms of PANDAS such as antibiotics in the absence of confirmed strept infection. There are many case reports, but there are no randomized controlled trials for PANS (Murphy et al. 2012). There are studies evaluating the use of intravenous immunoglobulin (IVIG) vs placebo, with improvement of symptoms by 40 % for IVIG and 60 % for plasmapheresis (Perlmutter et al. 1999). Two trials evaluating prophylaxis for strept infections were beneficial in reducing the rate of recurrence in PANDAS (Snider and Swedo 2004). But further research is needed.
In some prepubertal patients with anorexia, there is an antecedent of streptococcal infection. These patients have increased neurological abnormalities such as motor hyperactivity, fidgeting, or pacing (Vincenzi et al. 2010). In a recent review of the literature from 1990 to 2008 authors suggest measuring a CBC, an ESR, CRP, neopterine, a throat swab for strep and ASO titers, AntiDNAse B, AnAb, and D8/17. PANDAS-AN continues to be a controversial diagnosis in the literature (Vincenzi et al. 2010; Puxley et al. 2008).
Externalizing Disorders
ADHD
Conflicting evidence has been reported in PIM in children with ADHD. There are three German studies by the same group on this topic. One study with a group of 35 patients with ADHD and controls, levels of IFN gamma in association with lower birth weight and prematurity were reported (Oades 2011; Oades et al. 2010a, b). The second study reported levels of IL-1 were elevated in patients with a history of paternal smoking (Oades 2011). Patients with increased IL-16 and symptoms of hyperactivity were associated with errors of commission. Levels of IL-13 were associated with symptoms of omission in patients with ADHD (Oades et al. 2010a). In the last study by the same group, no differences were observed between non-medicated patients with ADHD vs. medicated patients vs. controls. There was a trend towards elevated levels of IFN gamma in patients with ADHD (Oades 2011).
Trios (n=958) of children with ADHD and their parents were compared genetically to 816 patients and three SNPs vs. 816 patients reported 3 SNPs rs7172689, rs7172689, rs4128767 in the IL-16 gene in chromosome 15 were associated with inattention in patients with ADHD (Lasky-Su et al. 2008).
In a genetic study evaluating 220 patients with ADHD, there was no association with the IL1Ra gene polymorphism (Misener et al. 2004). Lasky-Su et al. (2008) analyzed 930 parents and patients and found IL-3 was associated with earlier onset ADHD. Segman et al. (2002) studied 86 patients with ADHD and parents, the IL-1Ra 4 repeat allele transmission was increased in patients with ADHD.
Disruptive Behavior
In an adult study examining 153 healthy adults, hostility was examined using the Cook-Medley Hostility Scale (CMHS), an association between hostility and cytokine production was described. Levels of IFN gamma, TNF alpha, and IL-5 were higher in patients with higher hostility (Janicki-Deverts et al. 2010). One study evaluated a sample of 113 children with a profile of behavioral dysregulation as measured by higher scores on the Child Behavior Check List (CBCL) scale for anxiety/depression, aggressive behavior, and attention problems. Fifty-one children scored 2.5 standard deviations above the norm on the CBCL. These children had increased CRP as compared to children with normal scores (n = 82) on the CBCL DP profile (Holtmann et al. 2013).
Suicide
Since not all suicidal behavior is associated with mood disorder, this section highlights suicide as a separate topic. Suicide rates are high among young people; it is the third leading cause of death in 15- to 24-year-olds and accounts for 13 % of all adolescent deaths annually (Falcone 2008; Nock et al. 2013). Suicidal ideation and attempts are very frequent in adolescents. A survey of adolescents showed that 19 % had seriously considered suicide in the last year, 15 % had made a plan and 8 % reported that they had made an attempt. Since 1950, the suicide rate of adolescents has quadrupled and represents 12 % of the mortality rate in this age group (Brent 2011). Multiple studies evaluating peripheral biological makers have been reported in suicidal patients and postmortem brains of those who have committed suicide (Birmaher et al. 1996). Different systems have been reported to show abnormalities in suicidal patients: the serotoninergic system, the HPA axis, neurotrophin factors, and the neuroimmune system (Serafini et al. 2013a). In the quest for biological markers for different mental illnesses, goals morphed based on genetic studies. If similar genes are involved in different psychiatric disorders, similar specific symptoms may have in common some specific biological pathways. Also independent environmental factors such as stress and adverse childhood experiences can alter the neuroimmune network in youth (Shonkoff et al. 2012). These alterations can predispose to suicide later in life (Dube et al. 2001, 2009; Danese et al. 2007). Special traits such as impulsivity and aggression might be more frequent in adolescent suicides (Nock et al. 2013; Nock 2009).
Although the exact pathophysiology of suicidal behavior has not been unraveled, several studies point to increasing evidence that there is an alteration in the immune system. There are at least 21 published studies of peripheral inflammatory makers in patients after a suicide attempt to date (Serafini et al. 2013a). Although the role of cytokines in suicidal patients has been investigated since the early 1980s and remains ambiguous, the results look more compelling (Johnson et al. 2002; Falcone et al. 2010a, b; Gabbay et al. 2009b; Bayard-Burfield et al. 1996; Carlborg et al. 2013; Dwivedi 2010; Dwivedi et al. 2003; Erhardt et al. 2013; Isung et al. 2012a; Janelidze et al. 2011; Kim et al. 2007; Lee and Kim 2010; Lindqvist et al. 2008, 2011; Serafini et al. 2013b; Steiner et al. 2008; Sublette et al. 2011). Melancholic patients have been consistently reported as having low monocyte counts and cytokine abnormalities (Rothermundt et al. 2001a). Due to the crucial role in the acute inflammatory reaction the monocytes–macrophage system has received the most attention (Serafini et al. 2013a). Cytokines produced by the monocytes like, IL-1beta, activate the complement system.
In adolescents, levels of several inflammatory markers have been identified in patients with suicidal ideation and behavior. Studies have reported alterations of IL-1B, IL-6, TNF-alpha, IFN-gamma, VEGF, S100B, orexin, BDNF, and Kyneurine (Falcone et al. 2010a; Gabbay et al. 2009b; Serafini et al. 2013a; Janelidze et al. 2011).
Individual Inflammatory Markers
Cytokines
The role of stress is important to help us understand the psychosocial and neurobiological underpinnings of suicidal patients (Slopen et al. 2012a; Segerstrom and Miller 2004). Stress activates the inflammatory cascade, and as a result of this reaction, microglia become activated (Slopen et al. 2012a; Bremner 1999; Serafini et al. 2013a; Singareddy et al. 2013; Lucas et al. 2013; Young 2013). These cells produce proinflammatory cytokines such as IL-1B, IL-2, IL-6, TNF; IL-1B and IL-6, impacting the permeability of the BBB (Oby and Janigro 2006; Janigro 1999, 2012). These increases have been identified in patients who have attempted suicide. Dysregulation of the HPA axis has been reported in some studies evaluating suicidal behavior. Alteration in the HPA axis is directly related to the production of cytokines. When monocytes are activated, they can cross the BBB and activate microglia, consistent with evidence from postmortem studies of patients who have committed suicide (Rothermundt et al. 2001c; Motivala et al. 2005; Steiner et al. 2008, 2013).
IL1 B
Levels of IL1 have been shown to potentiate the brain concentrations of serotonin and norepinephrine. This cytokine has also been associated with the activation of the HPA axis and is reported to increase the secretion of ACTH and cortisol (Steiner et al. 2008, 2013). Pandey et al. in a postmortem study of teenage suicide victims reported increased expression of IL1B in the area of Broadman 10 (Pandey and Dwivedi 2012; Pandey et al. 2012).
IL 6
Levels of IL6 are reported to be elevated in patients with depression, exposure to childhood trauma, and suicide. Evidence from several meta-analysis from patients with mood disorder and schizophrenia has reported increased levels of IL-6. One third of the circulating levels of IL-6 come from adipose tissue. Smoking has also been linked to increased levels of IL-6. Three studies reported an increase in the levels of IL 6 in patients with suicide (Zorrilla et al. 2001; Dowlati et al. 2010; Steiner et al. 2008).
TNF Alpha
Increased levels of norepinephrine in patients who are receiving TNF alpha have been described (Dowlati et al. 2010). Animal studies found changes on levels of TNF alpha after treatment with antidepressants. Some theories involve the effect of antidepressants effect on the glucocorticoid receptor. Increased levels of TNF alpha have been reported in five studies with suicidal patients (Zorrilla et al. 2001; Dowlati et al. 2010; Serafini et al. 2013a; Pandey and Dwivedi 2012; Pandey et al. 2012), one of them in adolescents (Pandey et al. 2012).
IFN Gamma
This cytokine has been decreased after treatment in vivo or in vitro with antidepressants (Kubera et al. 1996). Two studies reported altered levels of IFN gamma in suicidal patients (Gabbay et al. 2009b; Janelidze et al. 2011), one of them in adolescents reporting a different biological pattern compared to the adults. Decreased levels of this cytokine were found in suicidal adolescents, although sample size was small (Gabbay et al. 2009b). IFN Gamma is one of the first steps on the Kyneurinic pathway (Hughes et al. 2012).
Kyneurinic/Quinolinic Acid
Inflammation increased the production of quinolinic acid (QUIN) and kyneurinic acid (KYNA). The Kyneurinic pathway is altered in patients with mood disorder and schizophrenia (Gabbay et al. 2009b; Hughes et al. 2012). Some preliminary studies on youth with ADHD and those exposed to trauma may also be implicated by this pathway. Longitudinal studies of patients undergoing treatment for mood disorders (MoodInflame Group) have demonstrated decreased levels of kyneurinic acid in patients who are improving. Evidence from postmortem studies of mood disorder patients has reported alteration in certain areas of the brain on the Kyneurinic pathway in a group of depressed patients. Two studies have reported alterations on the Kyneurinic pathway in those attempting suicide (Steiner et al. 2013; Hughes et al. 2012; Claes et al. 2011; Miura et al. 2008). Sublette compared levels between healthy controls and depressed patients who had attempted suicide and those who had not (Sublette et al. 2011). Erhardt et al. (2013) reported levels of increased QUIN acid in the CSF of 64 medication free suicide attempters. The authors hypothesize that alteration of the KYN pathway in suicide might be related to the glutamatergic transmission, since QUIN is an NMDA receptor agonist. The fact that ketamine has shown promising results in improving suicidality in four small studies might suggest that the NMDA receptor could be an important pathway to further study in patients with suicidal behavior (DiazGranados et al. 2010; Larkin and Beautrais 2011; Price et al. 2009; Zarate et al. 2012).
S100B
S100B is a glial and trophic marker, located primarily on the cytoplasm of oligodendrocytes and astrocytes (Arolt et al. 2003; Andreazza et al. 2007). S100B stays in the brain, unless there are alterations of the BBB. At which time this protein is identified in serum (Marchi et al. 2004). In a study on adolescents presenting with suicidal behavior, levels of S100B were increased in patients compared to controls despite a diagnosis of MDD, bipolar disorder, or schizophrenia (Falcone et al. 2010a). This study was later replicated by our group using the Columbia Suicidality Screen Rating Scale (CSSRS) as the measure for suicidal ideation in adolescents. Levels of S100B were increased in those adolescents who were at greater risk for suicide and may be possibly a tool to consider for assesment of risk (Falcone et al. 2010a).
Brain Derived Neurotrophic Factor
Brain derived neurotrophic factor (BDNF) is a neurotrophic classified as a nerve growth factor, affecting growth, plasticity, and neurogenesis. Some suggest that suicidal behavior may be related with alterations in neural plasticity. There is evidence from postmortem studies reporting a decreased number of glial cells, and glial density in the frontal cortex and hippocampus (Oby and Janigro 2006; Janigro 1999). Five studies have reported alteration in the BDNF levels of suicidal patients (Gabbay et al. 2009b; Li et al. 2013; Erhardt et al. 2013; Janelidze et al. 2011; Sublette et al. 2011; Pandey et al. 2012; Boehm et al. 2010). Additionally the mRNA expression of BDNF was decreased in the pre-frontal cortex and hippocampus of suicide victims (Dwivedi et al. 2003).
Vascular Endothelial Growth Factor
Vascular endothelial growth factor (VEGF) is an angiogenic protein with an important role in neuroprotection and neurogenesis. Viikki et al. (2010) reported that patients with a VEGF polymorphism were resistant to depression treatment. Two studies reported a negative correlation of VEGF plasma levels proportionate to depression severity (Isung et al. 2012a, b). VEGF is reported to be low in the plasma of suicide attempters. Additionally, VEGF is a key factor in the integrity of the BBB and studies have reported alteration in the permeability of the BBB in patients with suicidal behavior (Serafini et al. 2013b).
Orexin
One of the hypothalamic neuropeptides that modulates the HPA axis has been linked to the regulation of sleep. Two studies have reported decreased CSF levels of Orexin in patients who attempted suicide. After patients received treatment (6 and 12 months) levels of orexin increased (Kim et al. 2007; Karege et al. 2005).
Conclusions
There are important advances in the field of psychoneuroimmunology in the last 25 years; however, most of the studies on inflammatory markers and psychiatric disorders come from adult populations (Zorrilla et al. 2001; Drexhage et al. 2010; Dowlati et al. 2010; Padmos et al. 2008; Shalev et al. 2009; Serafini et al. 2013a). In this chapter we focus on studies in children and adolescents (Carlton et al. 2009; Falcone et al. 2008a, 2010a, c, 2013; Gabbay et al. 2009b; Ginsberg et al. 2012; Janelidze et al. 2011; Falcone and Janigro 2008). Pediatric populations are key in our process to understand the development and progression of mental illness and in the case of finding novel treatments for those patients who have comorbid increase of inflammatory markers and different psychiatric disorders. The evidence in adult studies points to a developmental advantage when the treatment is started early (Akhondzadeh et al. 2007; Bresee et al. 2006; Muller et al. 2002, 2005; Rapaport et al. 2005; Riedel et al. 2005). The study of inflammatory markers in children is a complex field, important confounding factors have to be taken into account, such as age, Tanner stage, weight, use of medications, sleep, variability of the marker during the day, life stressors, trauma history, and family history of psychiatric disorder (Mitchell and Goldstein 2014; Lucas et al. 2013; Bahn and Schwarz 2011; Gottschalk et al. 2013; Lopresti et al. 2014). Most studies in pediatric populations are cross-sectional and retrospective. This limits our ability to identify pathogenic cause for mental illness, and only permits discussion of an association between inflammation and diverse psychiatric disorders reviewed. Despite this limitation, there is important preliminary evidence for the role of inflammation in psychiatric disorders in pediatric populations (Pardo et al. 2005; Falcone et al. 2010a, 2013; Mitchell and Goldstein 2014; Gabbay et al. 2009b, c; Padmos et al. 2009; Carrion et al. 2002, 2007, 2008, 2010a, b; Chez et al. 2007; Oades 2011; Oades et al. 2010a, b; Pandey and Dwivedi 2012; Carrion and Wong 2012; De Bellis and Kuchibhatla 2006). Taking into account the high incidence of comorbidities in child psychiatry, it is very unlikely that inflammatory markers can be used solely for diagnostic purposes. However they could be very helpful to evaluate treatment response, disease progression and help us monitor high-risk patients that may potentially benefit from preventive treatment (Mitchell and Goldstein 2014; Goldstein and Young 2013). There is progress in identifying the onset, course, and recurrence of behavioral disorders in children and adolescents; however, further research is needed to understand the role of inflammation and the relationship with psychosocial and psychobiological factors.
References
Ajmo Jr CT, Vernon DO, Collier L, Hall AA, Garbuzova-Davis S, Willing A, et al. The spleen contributes to stroke-induced neurodegeneration. J Neurosci Res. 2008;86(10):2227–34. doi:10.1002/jnr.21661.PubMedCentralPubMed
Akhondzadeh S, Tabatabaee M, Amini H, Ahmadi Abhari SA, Abbasi SH, Behnam B. Celecoxib as adjunctive therapy in schizophrenia: a double-blind, randomized and placebo-controlled trial. Schizophr Res. 2007;90(1–3):179–85.PubMed
Al-Ayadhi LY, Mostafa GA. Elevated serum levels of interleukin-17A in children with autism. J Neuroinflammation. 2012;9:158. doi:10.1186/1742-2094-9-158.PubMedCentralPubMed
Aloe L, Bracci-Laudiero L, Alleva E, Lambiase A, Micera A, Tirassa P. Emotional stress induced by parachute jumping enhances blood nerve growth factor levels and the distribution of nerve growth factor receptors in lymphocytes. Proc Natl Acad Sci U S A. 1994;91(22):10440–4.PubMedCentralPubMed
American Academy of Pediatrics, Stirling J, Jr, Committee on Child Abuse and Neglect and Section on Adoption and Foster Care, American Academy of Child and Adolescent Psychiatry, Amaya-Jackson L, National Center for Child Traumatic Stress, et al. Understanding the behavioral and emotional consequences of child abuse. Pediatrics. 2008;122(3):667–73. doi:10.1542/peds.2008-1885.
Andreazza AC, Cassini C, Rosa AR, Leite MC, de Almeida LM, Nardin P, et al. Serum S100B and antioxidant enzymes in bipolar patients. J Psychiatr Res. 2007;41(6):523–9. doi:10.1016/j.jpsychires.2006.07.013.PubMed
Andreoli AV, Keller SE, Rabaeus M, Marin P, Bartlett JA, Taban C. Depression and immunity: age, severity, and clinical course. Brain Behav Immun. 1993;7(4):279–92. doi:10.1006/brbi.1993.1028.PubMed
Andrews JA, Neises KD. Cells, biomarkers, and post-traumatic stress disorder: evidence for peripheral involvement in a central disease. J Neurochem. 2012;120(1):26–36. doi:10.1111/j.1471-4159.2011.07545.x.PubMed
Angelov L, Doolittle ND, Kraemer DF, Siegal T, Barnett GH, Peereboom DM, et al. Blood–brain barrier disruption and intra-arterial methotrexate-based therapy for newly diagnosed primary CNS lymphoma: a multi-institutional experience. J Clin Oncol. 2009;27(21):3503–9. doi:10.1200/JCO.2008.19.3789.PubMedCentralPubMed
Anisman H, Ravindran AV, Griffiths J, Merali Z. Endocrine and cytokine correlates of major depression and dysthymia with typical or atypical features. Mol Psychiatry. 1999;4(2):182–8.PubMed
Arolt V, Rothermundt M. The immunology of psychiatric disorders. Psychother Psychosom Med Psychol. 2005;55(1):36–48.PubMed
Arolt V, Peters M, Erfurth A, Wiesmann M, Missler U, Rudolf S, et al. S100B and response to treatment in major depression: a pilot study. Eur Neuropsychopharmacol. 2003;13(4):235–9.PubMed
Ashdown H, Dumont Y, Ng M, Poole S, Boksa P, Luheshi GN. The role of cytokines in mediating effects of prenatal infection on the fetus: implications for schizophrenia. Mol Psychiatry. 2006;11(1):47–55.PubMed
Ashwood P, Anthony A, Torrente F, Wakefield AJ. Spontaneous mucosal lymphocyte cytokine profiles in children with autism and gastrointestinal symptoms: mucosal immune activation and reduced counter regulatory interleukin-10. J Clin Immunol. 2004;24(6):664–73. doi:10.1007/s10875-004-6241-6.PubMed
Ashwood P, Schauer J, Pessah IN, Van de Water J. Preliminary evidence of the in vitro effects of BDE-47 on innate immune responses in children with autism spectrum disorders. J Neuroimmunol. 2009;208(1–2):130–5. doi:10.1016/j.jneuroim.2008.12.012.PubMedCentralPubMed
Ashwood P, Nguyen DV, Hessl D, Hagerman RJ, Tassone F. Plasma cytokine profiles in Fragile X subjects: is there a role for cytokines in the pathogenesis? Brain Behav Immun. 2010;24(6):898–902. doi:10.1016/j.bbi.2010.01.008.PubMedCentralPubMed
Ashwood P, Krakowiak P, Hertz-Picciotto I, Hansen R, Pessah I, Van de Water J. Elevated plasma cytokines in autism spectrum disorders provide evidence of immune dysfunction and are associated with impaired behavioral outcome. Brain Behav Immun. 2011a;25(1):40–5. doi:10.1016/j.bbi.2010.08.003.PubMedCentralPubMed
Ashwood P, Krakowiak P, Hertz-Picciotto I, Hansen R, Pessah IN, Van de Water J. Altered T cell responses in children with autism. Brain Behav Immun. 2011b;25(5):840–9. doi:10.1016/j.bbi.2010.09.002.PubMedCentralPubMed
Bahn S, Schwarz E. Serum-based biomarkers for psychiatric disorders. Nervenarzt. 2011;82(11):1395–6, 1398, 1400 passim. doi:10.1007/s00115-011-3346-z.
Bayard-Burfield L, Alling C, Blennow K, Jonsson S, Traskman-Bendz L. Impairment of the blood-CSF barrier in suicide attempters. Eur Neuropsychopharmacol. 1996;6(3):195–9.PubMed
Bent S, Bertoglio K, Ashwood P, Nemeth E, Hendren RL. Brief report: hyperbaric oxygen therapy (HBOT) in children with autism spectrum disorder: a clinical trial. J Autism Dev Disord. 2012;42(6):1127–32. doi:10.1007/s10803-011-1337-3.PubMed
Bergink V, Gibney SM, Drexhage HA. Autoimmunity, inflammation, and psychosis: a search for peripheral markers. Biol Psychiatry. 2014;75(4):324–31. doi:10.1016/j.biopsych.2013.09.037.PubMed
Birmaher B, Rabin BS, Garcia MR, Jain U, Whiteside TL, Williamson DE, et al. Cellular immunity in depressed, conduct disorder, and normal adolescents: role of adverse life events. J Am Acad Child Adolesc Psychiatry. 1994;33(5):671–8. doi:10.1097/00004583-199406000-00008.PubMed
Birmaher B, Ryan ND, Williamson DE, Brent DA, Kaufman J. Childhood and adolescent depression: a review of the past 10 years. Part II. J Am Acad Child Adolesc Psychiatry. 1996;35(12):1575–83. doi:10.1097/00004583-199612000-00008.PubMed
Boehm J, Fischer K, Bohnert M. Putative role of TNF-alpha, interleukin-8 and ICAM-1 as indicators of an early inflammatory reaction after burn: a morphological and immunohistochemical study of lung tissue of fire victims. J Clin Pathol. 2010;63(11):967–71. doi:10.1136/jcp.2010.079863.PubMed
Boksa P. Maternal infection during pregnancy and schizophrenia. J Psychiatry Neurosci. 2008;33(3):183–5.PubMedCentralPubMed
Bonaccorso S, Puzella A, Marino V, Pasquini M, Biondi M, Artini M, et al. Immunotherapy with interferon-alpha in patients affected by chronic hepatitis C induces an intercorrelated stimulation of the cytokine network and an increase in depressive and anxiety symptoms. Psychiatry Res. 2001;105(1–2):45–55.PubMed
Bos-Veneman NG, Bijzet J, Limburg PC, Minderaa RB, Kallenberg CG, Hoekstra PJ. Cytokines and soluble adhesion molecules in children and adolescents with a tic disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(8):1390–5. doi:10.1016/j.pnpbp.2010.06.028.PubMed
Brambilla F, Monteleone P, Maj M. Interleukin-1beta and tumor necrosis factor-alpha in children with major depressive disorder or dysthymia. J Affect Disord. 2004;78(3):273–7. doi:10.1016/S0165-0327(02)00315-4.PubMed
Bremner JD. Does stress damage the brain? Biol Psychiatry. 1999;45(7):797–805.PubMed
Bremner JD. Traumatic stress: effects on the brain. Dialogues Clin Neurosci. 2006;8(4):445–61.PubMedCentralPubMed
Brent DA. Preventing youth suicide: time to ask how. J Am Acad Child Adolesc Psychiatry. 2011;50(8):738–40. doi:10.1016/j.jaac.2010.09.017.PubMed
Brent DA, Kolko DJ, Birmaher B, Baugher M, Bridge J, Roth C, et al. Predictors of treatment efficacy in a clinical trial of three psychosocial treatments for adolescent depression. J Am Acad Child Adolesc Psychiatry. 1998;37(9):906–14. doi:10.1097/00004583-199809000-00010.PubMed
Brent DA, Oquendo M, Birmaher B, Greenhill L, Kolko D, Stanley B, et al. Peripubertal suicide attempts in offspring of suicide attempters with siblings concordant for suicidal behavior. Am J Psychiatry. 2003;160(8):1486–93.PubMed
Brent DA, McMakin DL, Kennard BD, Goldstein TR, Mayes TL, Douaihy AB. Protecting adolescents from self-harm: a critical review of intervention studies. J Am Acad Child Adolesc Psychiatry. 2013;52(12):1260–71. doi:10.1016/j.jaac.2013.09.009.PubMed
Bresee CJ, Delrahim K, Maddux RE, Dolnak D, Ahmadpour O, Rapaport MH. The effects of celecoxib augmentation on cytokine levels in schizophrenia. Int J Neuropsychopharmacol. 2006;9(3):343–8.PubMed
Brown GR, Anderson B. Psychiatric morbidity in adult inpatients with childhood histories of sexual and physical abuse. Am J Psychiatry. 1991;148(1):55–61.
Brown AS, Hooton J, Schaefer CA, Zhang H, Petkova E, Babulas V, et al. Elevated maternal interleukin-8 levels and risk of schizophrenia in adult offspring. Am J Psychiatry. 2004;161(5):889–95.PubMed
Brown DW, Anda RF, Tiemeier H, Felitti VJ, Edwards VJ, Croft JB, et al. Adverse childhood experiences and the risk of premature mortality. Am J Prev Med. 2009;37(5):389–96. doi:10.1016/j.amepre.2009.06.021.
Buter J, de Vries EG, Sleijfer DT, Willemse PH, Mulder NH. Neuropsychiatric symptoms during treatment with interleukin-2. Lancet. 1993;341(8845):628.PubMed
Caplan R, Siddarth P, Gurbani S, Hanson R, Sankar R, Shields WD. Depression and anxiety disorders in pediatric epilepsy. Epilepsia. 2005;46(5):720–30.PubMed
Capuron L, Miller AH. Cytokines and psychopathology: lessons from interferon-alpha. Biol Psychiatry. 2004;56(11):819–24. doi:10.1016/j.biopsych.2004.02.009.PubMed
Capuron L, Ravaud A, Miller AH, Dantzer R. Baseline mood and psychosocial characteristics of patients developing depressive symptoms during interleukin-2 and/or interferon-alpha cancer therapy. Brain Behav Immun. 2004;18(3):205–13. doi:10.1016/j.bbi.2003.11.004.PubMed
Capuron L, Miller A, Irwin MR. Psychoneuroimmunology of depressive disorder: mechanisms and clinical implications. In: Ader R, editor. Psychoneuroimmunology, vol. I. Paris: Elsevier; 2007. p. 509–30. ISBN: 978-0-12-088576-3.
Carlborg A, Jokinen J, Jonsson EG, Erhardt S, Nordstrom P. CSF kynurenic acid and suicide risk in schizophrenia spectrum psychosis. Psychiatry Res. 2013;205(1–2):165–7. doi:10.1016/j.psychres.2012.08.021.PubMed
Carlton E, Falcone T, Tuxhorn I, Janigro D. Psychosis and epilepsy in childhood – the inflammatory hypothesis. Schizophr Res. 2008;98(Suppl 1):80.
Carrion VG, Wong SS. Can traumatic stress alter the brain? Understanding the implications of early trauma on brain development and learning. J Adolesc Health. 2012;51(2 Suppl):S23–8. doi:10.1016/j.jadohealth.2012.04.010.PubMed
Carrion VG, Wong SS. Can traumatic stress alter the brain? Understanding the implications of early trauma on brain development and learning. J Adolesc Health. 2012;51(2 Suppl):S23–8. doi:10.1016/j.jadohealth.2012.04.010.
Carrion VG, Weems CF, Ray RD, Glaser B, Hessl D, Reiss AL. Diurnal salivary cortisol in pediatric posttraumatic stress disorder. Biol Psychiatry. 2002;51(7):575–82.PubMed
Carrion VG, Weems CF, Reiss AL. Stress predicts brain changes in children: a pilot longitudinal study on youth stress, posttraumatic stress disorder, and the hippocampus. Pediatrics. 2007;119(3):509. doi:10.1542/peds. –516, 2006-2028.PubMed
Carrion VG, Garrett A, Menon V, Weems CF, Reiss AL. Posttraumatic stress symptoms and brain function during a response-inhibition task: an fMRI study in youth. Depress Anxiety. 2008;25(6):514–26. doi:10.1002/da.20346.PubMed
Carrion VG, Weems CF, Richert K, Hoffman BC, Reiss AL. Decreased prefrontal cortical volume associated with increased bedtime cortisol in traumatized youth. Biol Psychiatry. 2010a;68(5):491–3. doi:10.1016/j.biopsych.2010.05.010.PubMedCentralPubMed
Carrion VG, Haas BW, Garrett A, Song S, Reiss AL. Reduced hippocampal activity in youth with posttraumatic stress symptoms: an FMRI study. J Pediatr Psychol. 2010b;35(5):559–69. doi:10.1093/jpepsy/jsp112.PubMedCentralPubMed
Caserta MT, O’Connor TG, Wyman PA, Wang H, Moynihan J, Cross W, et al. The associations between psychosocial stress and the frequency of illness, and innate and adaptive immune function in children. Brain Behav Immun. 2008;22(6):933–40. doi:10.1016/j.bbi.2008.01.007.PubMedCentralPubMed
Cheng YH, Zheng Y, He F, Yang JH, Li WB, Wang ML, et al. Detection of autoantibodies and increased concentrations of interleukins in plasma from patients with Tourette’s syndrome. J Mol Neurosci. 2012;48(1):219–24. doi:10.1007/s12031-012-9811-8.PubMed
Chez MG, Dowling T, Patel PB, Khanna P, Kominsky M. Elevation of tumor necrosis factor-alpha in cerebrospinal fluid of autistic children. Pediatr Neurol. 2007;36(6):361–5. doi:10.1016/j.pediatrneurol.2007.01.012.PubMed
Chou IC, Lin HC, Wang CH, Lin WD, Lee CC, Tsai CH, et al. Polymorphisms of interleukin 1 gene IL1RN are associated with Tourette syndrome. Pediatr Neurol. 2010;42(5):320–4. doi:10.1016/j.pediatrneurol.2010.01.006.PubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





