Scatterplot between HOMA-IR and right hippocampal volume corrected for total brain volume (Rasgon et al., 2011).
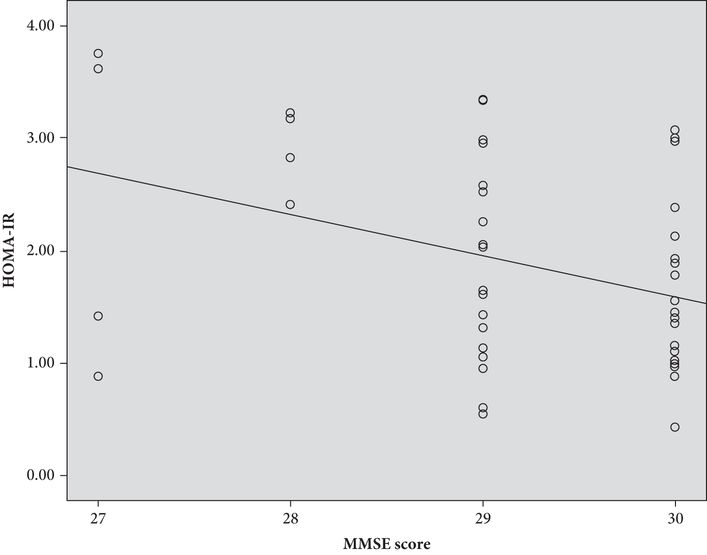
Results also indicated significant associations between higher HOMA-IR and reduced scores on a global measure of cognition (MMSE), estimated IQ, and verbal and visual attention measures regardless of APOE ε-4 carrier status. Figure 15.2 displays a scatterplot between HOMA-IR and MMSE.
Given that the study sample included postmenopausal women currently using hormone therapy, subsequent analyses assessed the possible confounding effects of the reproductive variables of endogenous and exogenous estrogen exposure. The relationship between HOMA-IR on right and total hippocampal volume, as well as MMSE scores, remained significant whereas reproductive variables on either hippocampal volumes or cognitive variables did not.
Insulin resistance and the default mode network
Pioneering work by a number of investigators has established the importance of the default mode network (DMN), or “resting state” network, as a biomarker of cognitive function and age-related decline (Greicius, Srivastava, Reiss, & Menon, 2004; Sorg et al., 2007; Zhou et al., 2010). Indeed, the DMN has proven to be a robust correlate of pathological brain aging (Zhou et al., 2010) and risk of dementia (Luchsinger, 2010). It encompasses widely separate brain regions, including the medial prefrontal cortex (MPFC), the posterior cingulate cortex (PCC), and lateral parietal cortices, all of which display a high degree of functional activity during rest (Fox et al., 2005). The DMN essentially “deactivates” during mental tasks with moderate or greater cognitive demand and “activates” during mental rest with eyes closed (Fox et al., 2005). Utilizing functional connectivity analysis allows the opportunity to examine DMN connections with critical brain regions, such as the hippocampus. Previous data on DMN functional connectivity in patients with DM2 are limited to one report (Musen et al., 2012), which identified that HOMA-IR was inversely correlated with reduced functional connectivity within the DMN, compared with control subjects.
Insulin resistance in risk for pathological brain aging was also assessed by utilizing a contrast analysis of functional connectivity between the DMN and the hippocampus during rest using fMRI in a subsample (n = 20) of women from the above study (Kenna et al., 2013). The sample was dichotomized into two groups based on the overall group mean fasting plasma insulin value (e.g. higher insulin levels (> 8 μIU/mL) and lower insulin levels (≥ 8 μIU/mL)).
Initial functional connectivity analysis showed the expected positive associations among the main DMN nodes (i.e. the MPFC, PCC, bilateral LPC), as well as reciprocal associations between the left and right hippocampal regions in the total sample (Figure 15.3). In the dichotomized groups, those with higher insulin exhibited significantly reduced positive associations between the MPFC seed and left and right parahippocampal regions, and conversely, between both the left and right hippocampal seed regions and the MPFC region, relative to subjects with lower insulin levels (Figure 15.4). Functional connectivity between the PCC and LPC with the hippocampal regions did not differ between the two contrast groups.
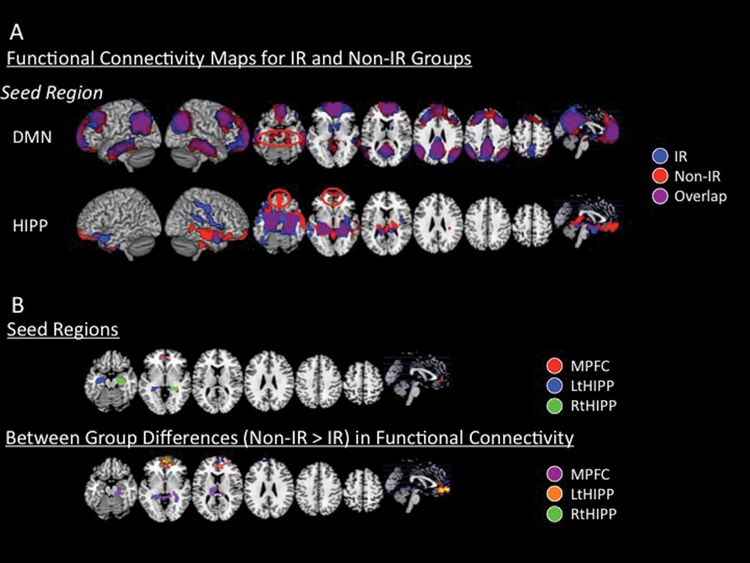
Functional connectivity maps for the higher insulin and lower insulin contrast groups. (A) Brain regions that showed positive association with seed regions’ time series. Seeds were placed in the DMN regions as well as in the left and right hippocampi. Weighted average of the four seed regions for the DMN and two seed regions for the hippocampi are shown in this figure (p = 0.01 whole-brain corrected). Maps of each individual seed region are shown. Red circles indicate brain regions that displayed activation in the lower insulin group (red) but not the higher insulin group (blue), and are in the MPFC and hippocampal regions. (B) Between-group differences in functional connectivity. Upper panel: Seed regions where the functional connectivity maps showed significant differences between groups. Lower panel: Compared with the lower insulin group, higher insulin subjects showed significantly reduced positive associations between the MPFC seed and bilateral hippocampal regions (pink-purple) as well as reduced positive associations between the left hippocampal seed and MPFC region (orange-yellow) and right hippocampal seed and MPFC (blue-green) (p = 0.01 whole-brain corrected) (Kenna et al., 2013).
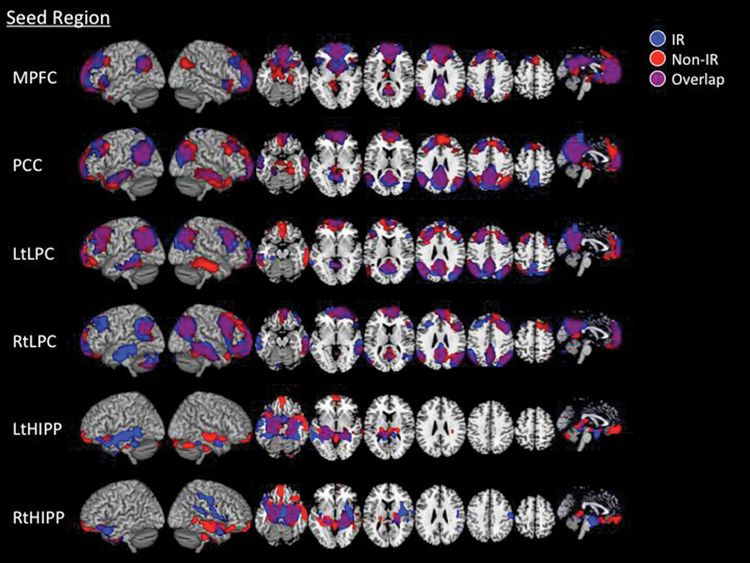
Functional connectivity maps for the higher insulin and lower insulin groups. Brain regions that displayed positive associations with seed regions’ time series. Seeds were placed in the DMN, MPFC, PCC, left and right LPC, and left and right hippocampi; p = –0.01 corrected (Kenna et al., 2013).
Exploration of potential confounding effects of group differences in IQ and executive function on the significant brain regions showed continued significant main effects of insulin group on MPFC–hippocampal connectivity even when controlling for IQ. Correlations of the MPFC–hippocampal connectivity and fasting plasma insulin values in the group as a whole were observed to be much stronger in the right and left hippocampus than connectivity with IQ.
These two described studies are the first, to our knowledge, to report on the effects of the modifiable risk factor, IR, on structural and functional integrity of the hippocampus. These findings may have a direct impact on patients with MDD who are at heightened risk for both IR and pathological brain aging. While we can only speculate, pending further data, we suggest that IR acts as a catalyst for deleterious brain morphology and network connectivity.
Summary
There is strong converging evidence to support the negative effects of glucose dysregulation on brain structure and function that lead to cognitive decline and dementia. Our cumulative findings suggest that in middle-aged, euthymic adults with a history of MDD, indirect measures of IR are associated with disrupted memory and executive functioning, and corresponding metabolic decline in the MPFC, reductions in hippocampal volumes, and aberrant intrinsic connectivity between the hippocampus and MPFC (Kenna et al., 2013; Rasgon et al., 2011, 2014). Insulin resistance is the underlying metabolic condition of DM2, and may be corrected with pharmacological and behavioral treatments. Together with the literature to date on the impact of IR on cognitive function and risk of MDD, the cumulative data strongly suggest the importance of clinically addressing IR in aging individuals, especially those with a history of MDD. Taken together, the evidence underscores the importance of identifying IR in patients with MDD and the development of interventions to prevent the onset of DM2.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree