Intracranial Aneurysms, Diagnosis and Treatment
Key Points
Intracranial aneurysms are delicate structures prone to procedural rupture or perforation. Treat them very gently.
Rotational 3D angiography has improved enormously the angiographic interrogation of complex intracranial anatomy. Small or blister aneurysms are difficult to see on conventional planar views and a low threshold for performing a 3D run during diagnostic studies is a good policy.
The term aneurysm usually refers to a persistent pathologic dilatation of an arterial wall. In certain diseases, particularly arteriovenous malformations, the possibility of aneurysms affecting venous structures must be considered as well. Arterial aneurysms may be described according to configuration as fusiform, when the whole vessel circumference is involved (Figs. 18-1 and 18-2), or saccular, when the lesion is eccentric. Mild dilatation of a segment of vessel is called ectasia. The point at which diffuse ectasia becomes an extended fusiform aneurysm is often difficult to define objectively. The term pseudoaneurysm is used when the circumferential containment of the lumen by the arterial wall is lost to a substantial degree and is dependent on an improvised barrier of clot, adventitia, or surrounding tissues.
Extradural Aneurysms and Pseudoaneurysms
Extradural aneurysms are unlikely to cause subarachnoid hemorrhage, unless they rupture with particular force against the dura. They are therefore, usually considered to be less immediately life-threatening than subarachnoid aneurysms. The commonest location for true aneurysms of the extradural carotid artery is in the cavernous segment.
Aneurysms of the Petrous and Cervical Segments of the Internal Carotid Artery
Aneurysms of the petrous and cervical segments are uncommon. They are sometimes seen in patients with connective tissue diseases. However, pseudoaneurysms in these areas are encountered more frequently. They can be related to complications of posttraumatic or idiopathic dissection, parapharyngeal infection, infection of the petrous air cells, skull fracture, surgical or traumatic laceration, or tumor invasion. When complications of bleeding or embolic phenomena in the internal carotid artery territory occur, these lesions can constitute a serious risk to life. Bleeding from a petrous aneurysm may occur when erosion of the adjacent supporting bone permits rupture of the aneurysm into the middle ear or sphenoid sinus. These aneurysms or pseudoaneurysms may also present with mass effect as they bulge into adjacent structures, particularly the floor of the middle cranial fossa where they may compress branches of the trigeminal nerve.
Aneurysms of the Cavernous Segment of the Internal Carotid Artery
Aneurysms in this location are relatively common and are seen particularly in older patients. They may be fusiform or have a relatively defined neck. Frequently, there are variable degrees of intraluminal thrombus and atherosclerotic mural changes. They are often giant in size (>25 mm), in which case they are likely to present with mass effect and neuropathy of the paracavernous cranial nerves. They may rupture into the cavernous sinus and establish a carotid-cavernous fistula. It is uncommon for them to rupture into the subarachnoid or subdural spaces.
Cavernous aneurysms projecting medially into the sella turcica are thought to carry a particular risk of subarachnoid hemorrhage if they rupture, but even this phenomenon for aneurysms in this particular location is rare. Additional care is advised for large cavernous aneurysms when the support of the sphenoid sinus wall has become eroded. Rupture of an aneurysm in this direction is life-threatening due to exsanguinating epistaxis, a feature to bear in mind when reading the CT bone windows.
Most small cavernous aneurysms are incidental observations on angiographic or axial examinations. Asymptomatic cavernous aneurysms are usually not treated unless there is compelling evidence of imminent complications.
False Aneurysms of the Intracranial Circulation
Cerebral aneurysms can be classified according to location, size, etiology, or configuration. When classified by
the integrity or otherwise of their mural components, they can be separated into true aneurysms (intima and adventitia intact) or pseudoaneurysms. A pseudoaneurysm is one in which the wall of the artery has been perforated. The apparent lumen of the opacified aneurysm is contained by an organized extraluminal hematoma.
the integrity or otherwise of their mural components, they can be separated into true aneurysms (intima and adventitia intact) or pseudoaneurysms. A pseudoaneurysm is one in which the wall of the artery has been perforated. The apparent lumen of the opacified aneurysm is contained by an organized extraluminal hematoma.
Postsurgical Pseudoaneurysms
False aneurysms of the proximal or distal intracranial vessels after surgery are rare. They have been seen following vessel injury in the course of open or endoscopic sinus surgery (1,2,3,4), transsphenoidal pituitary procedures (5,6), stereotactic biopsy (7), or craniotomy for a variety of reasons (8).
Traumatic Intracranial Aneurysms
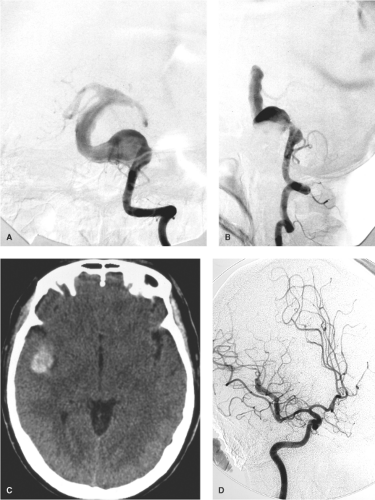
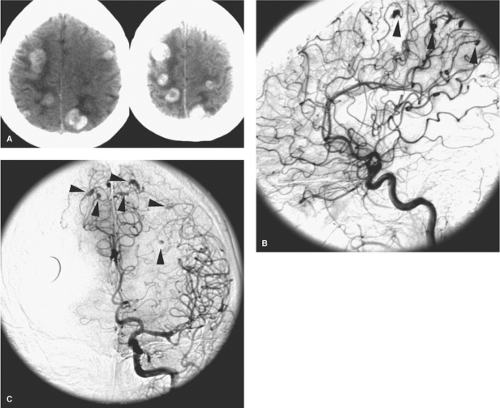
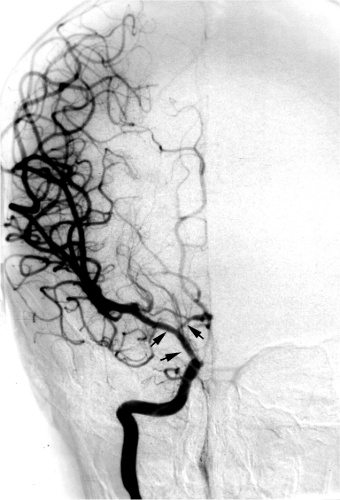
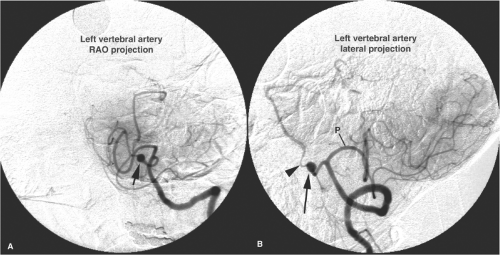
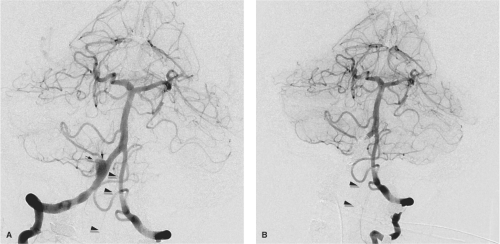
Pseudoaneurysms of the intracranial circulation may be seen as a result of penetrating or nonpenetrating head injuries (9), impaction on the vessel by bone fragments (10), or following surgical injury (11,12). They are typically seen in young males after severe injury to the head (Figs. 18-3 and 18-4), but more than 30% are identified in children (13,14). Posttraumatic lacerations of intracranial major vessels are often fatal, and in the past it would have been unusual for a patient so injured to reach medical attention.
A consistent theme in the literature dealing with posttraumatic pseudoaneurysms is that their discovery depends entirely on the index of suspicion of the managing physician. Without angiography, they frequently elude early detection. They can be present with subacute or delayed rebleeding or other complications, typically after a period of 1 or 2 weeks but possibly as long as months (9,15).
These injuries can be seen in the anterior or posterior circulation (16) and most frequently have a contiguous or related bone injury. Their discovery is usually associated with penetrating injuries in which the missile impact has scattered numerous fragments of bone or metal in diverging trajectories, particularly when fragments are seen close to the skull base (17).
Pseudoaneurysms in the setting of closed head injury have also been seen where no violation of the dura or skull is present (18). Pseudoaneurysms may be seen with closed head injury, particularly in children, when frontolateral shear injuries cause significant impaction of the pericallosal and callosomarginal branches against the falx cerebri, or of the middle cerebral artery against the sphenoid ridge (19). Traumatic aneurysms may account for 14% to 39% of intracranial aneurysms in the pediatric population (20). Traumatic intracranial pseudoaneurysms may be a more significant problem in military experience than in civilian life. While they were described as rare in the Korean and Vietnamese wars (21,22), experience in Lebanon and in the Iran–Iraq wars in the 1970s and 1980s indicates an incidence higher than once supposed (9,17,23,24), a trend that has become even more prominent with the high incidence of blast and penetrating head injuries casualties returning from the Iraq and Afghanistan wars (25,26,27,28). As many as 35% of military casualties from these conflicts who undergo cerebral angiography for vascular problems demonstrate intracranial or extracranial pseudoaneurysms, as well as other cerebrovascular complications such as vasospasm (29), vessel occlusions, or late development of carotid-cavernous fistulas (30).
Intracranial pseudoaneurysms also feature prominently in low-velocity civilian injuries, such as in South Africa where stabbing injuries to the head are more commonplace than in the Occidental urban experience. In stabbing victims, vascular injuries to the head may reach an incidence of
30%, including pseudoaneurysms, dissections, fistulae, and occlusions (31).
30%, including pseudoaneurysms, dissections, fistulae, and occlusions (31).
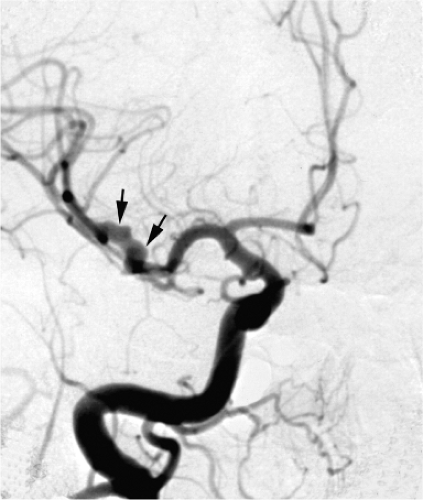
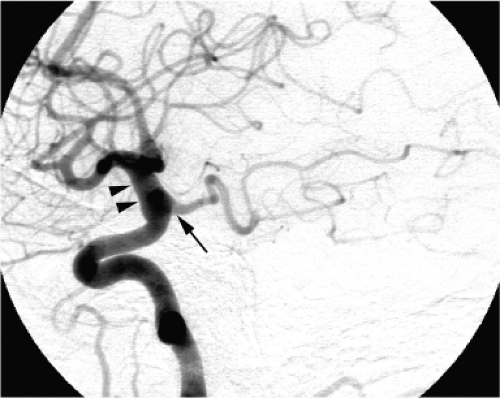
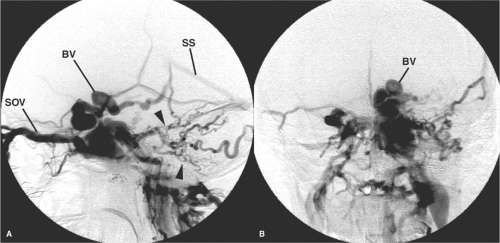
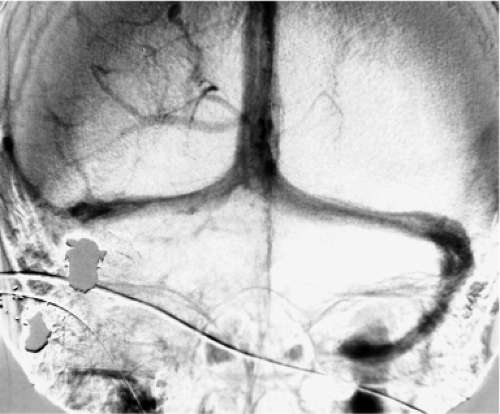
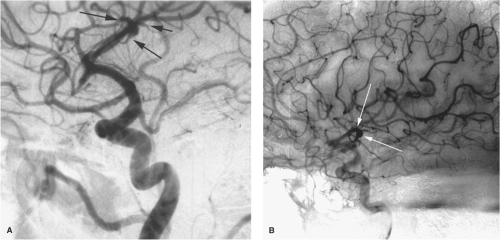
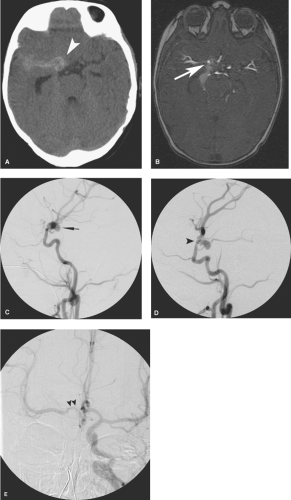
Complications specifically related to false aneurysms include delayed rupture; mass effect on adjacent brain or cranial nerves; or an associated intraparenchymal, subarachnoid, or subdural hemorrhage. Delayed presentation may be seen months or even years following the initial injury (32). Acute or delayed complications of carotid injuries in the sphenoidal region include carotid cavernous fistulae and massive epistaxis from the sphenoidal sinus (Figs. 18-5 and 18-6).
Posttraumatic aneurysms represent a difficult management problem, as there is a significant risk of profuse bleeding at surgery when the dura is opened. Trapping of the pseudoaneurysm or occlusion of the parent vessel proximal to the laceration by surgical or endovascular means may represent the best therapeutic option in certain circumstances (25,27,33).
Immediate angiography for patients with penetrating head injuries is likely prudent as approximately 12% to 35% of such patients will have an intracranial pseudoaneurysm (29,34). Follow-up angiography may be necessary for patients in whom spasm or other vascular abnormalities preclude adequate evaluation.
Fusiform Intradural Aneurysms
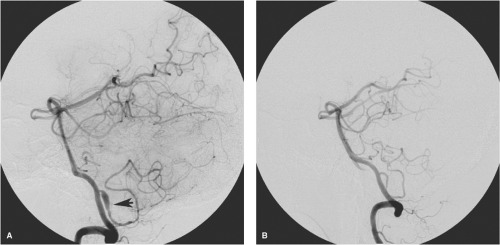
Fusiform aneurysms are frequently associated with vessel tortuosity, hypertension, atherosclerosis, and advancing age. They typically affect the cavernous or supraclinoidal internal carotid artery and the basilar trunk. They often present with symptoms related to mass effect (Fig. 18-7). With large aneurysms, stagnation of blood can lead to thrombus formation, and patients may present with embolic stroke (35,36). Rupture is not common when the condition is mild (37). Surgical management of a ruptured fusiform
segment of vessel can be difficult (38). Techniques, including wrapping, proximal occlusion, or bypassing, must make allowance for preserving important perforator vessels, which might take origin from the dysplastic segment (39). Endovascular treatment with flow-diverting stents, such as Pipeline or Silk, will undoubtedly play more of a role in the treatment of these difficult lesions in the future (40,41,42,43).
segment of vessel can be difficult (38). Techniques, including wrapping, proximal occlusion, or bypassing, must make allowance for preserving important perforator vessels, which might take origin from the dysplastic segment (39). Endovascular treatment with flow-diverting stents, such as Pipeline or Silk, will undoubtedly play more of a role in the treatment of these difficult lesions in the future (40,41,42,43).
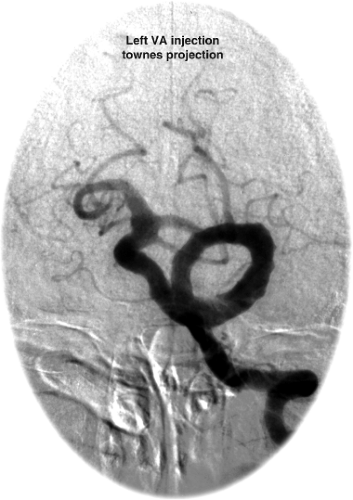
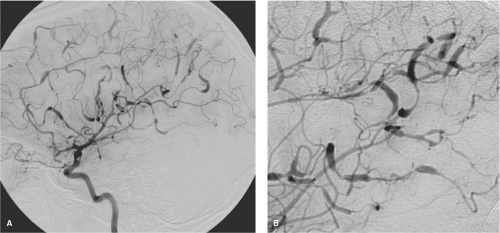
When ectasia of the basilar artery reaches bizarre proportions, the terms megadolichobasilar anomaly or giant fusiform aneurysm are used (Fig. 18-8) (36,44). They represent less than 1% of intracranial aneurysms. Although they are classically described as affecting the vertebrobasilar circulation, giant fusiform or serpentine aneurysms are also seen in the anterior circulation (39) (Fig. 18-8). This group of usually elderly patients typically presents with mass effect, brainstem compression, cranial nerve deficits, obstructive hydrocephalus, subarachnoid hemorrhage, and embolic strokes due to stagnation of blood. Underlying, partially healed dissection likely explains the genesis of many of these aneurysms (45). Furthermore, long-term clinical follow-up of this group of patients indicates a poor prognosis due to recurrent subarachnoid hemorrhage or progressive mass effect.
Mycotic and Inflammatory Aneurysms
Mycotic aneurysms are classically described as occurring distally in the cerebral circulation and as being not necessarily related to vessel bifurcation points. They may be subtle in appearance and are frequently best seen in the parenchymal phase of the injection when the arteries are already washing out.
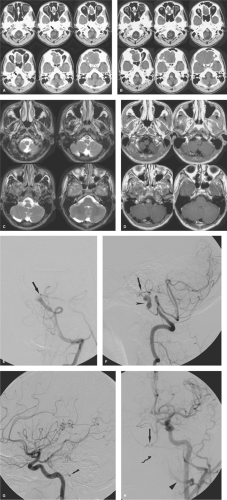
Intracranial aneurysms of inflammatory origin may be of several types: Bacterial; syphilitic (Heubner arteritis); related to angiocentric organisms such as mucormycosis, aspergillus (46), or other fungi; or associated with systemic arteritides such as giant cell arteritis, human immunodeficiency virus (HIV), and polyarteritis nodosa (Fig. 18-9).
In the modern era, an increased incidence of aneurysms related to HIV or its associated infections has been described. This may be primarily due to a necrotizing vasculitis related to the virus itself, particularly in congenital infections, or due to acquired diseases such as tuberculosis or syphilis (47,48,49).
Presentation and Incidence of Mycotic Aneurysms
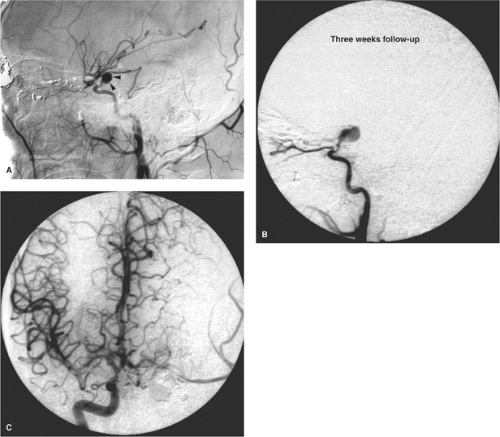
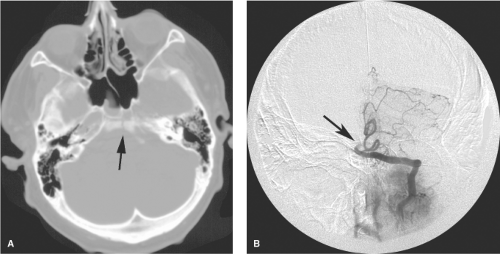
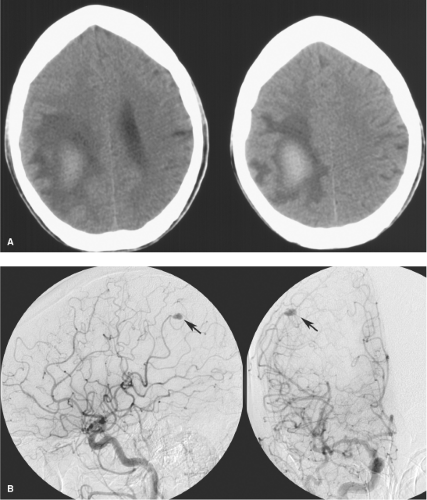
Mycotic intracranial aneurysms present most commonly with acute subarachnoid hemorrhage or intraparenchymal hematoma developing as a complication of already established septic disease. Occasionally, young patients may present with an embolic neurologic deficit, such as hemiplegia, as the presenting event of bacterial endocarditis (Figs. 18-10 and 18-11) (50). Other common presentations include seizure or focal neurologic signs due to vasculitis or vegetative emboli in an area of the brain distal to a silent aneurysm (51). Direct vessel invasion from adjacent infected paranasal sinuses may also be seen (Figs. 18-12 and 18-13).
Although mycotic aneurysms are typically described as being found in unusual peripheral locations, multiple in number, and not necessarily related to vessel bifurcations, this archetype is not always valid (52). They may be single in presentation and centrally located. Mycotic aneurysms can be seen in the cavernous carotid artery when a systemic illness is complicated by cavernous thrombophlebitis (53). When an aneurysm is seen in an unusual peripheral location, the possibility of an infectious embolic etiology should be considered, but not all mycotic aneurysms are atypical in appearance or location.
Bacterial aneurysms were typically associated with bacterial endocarditis when this disease was more common but can be seen with any septicemic state, particularly with respiratory infections. Older studies suggest that the incidence of intracranial aneurysms in patients with endocarditis varies between 4% and 15% or perhaps higher, and many of these aneurysms remain asymptomatic (53,54).
Mycotic aneurysms are related to impaction of a septic embolus in the intima of a peripheral cerebral artery with development of a resulting arteritis, focal mural necrosis, and aneurysm formation. This explains the friable nature of the wall of these lesions and their propensity to bleed spontaneously or during surgery.
Role of Endovascular Intervention in Management of Mycotic Aneurysms
Some inflammatory aneurysms, a third or more in some series, tend to regress with treatment of the primary infection (45,46,47) (55,56,57). Possibly as many as a third of them will resolve spontaneously with medical treatment alone (52,55). Better results are seen in patients with single, unruptured aneurysms. Sequential angiography in patients with infective endocarditis may be reasonable where a high index of suspicion for development of lesions, even in the setting of active treatment, much be maintained. Surgical evacuation of ruptured aneurysms with hematoma and endovascular treatment of unruptured aneurysms are typically necessary when medical management fails (51,58,59,60).
Oncotic Intracranial Aneurysms
Invasion of the intracranial vessel wall with destruction of the internal elastic lamina and cavitation with aneurysm or pseudoaneurysm formation is a rare but well-recognized event from metastatic deposits of atrial myxoma,
choriocarcinoma (61,62), renal cell carcinoma, or bronchogenic carcinoma (63,64,65). Invasion of arteriolar walls by gliomatous tumors may explain the very rare association of false aneurysms with primary brain tumors (66). Features shared by oncotic aneurysms are that they may be delayed at the onset (i.e., they may not be seen on an initial angiogram), may be multiple, may grow rapidly, or may thrombose spontaneously. They may resolve with chemotherapy for the primary lesion (67).
choriocarcinoma (61,62), renal cell carcinoma, or bronchogenic carcinoma (63,64,65). Invasion of arteriolar walls by gliomatous tumors may explain the very rare association of false aneurysms with primary brain tumors (66). Features shared by oncotic aneurysms are that they may be delayed at the onset (i.e., they may not be seen on an initial angiogram), may be multiple, may grow rapidly, or may thrombose spontaneously. They may resolve with chemotherapy for the primary lesion (67).
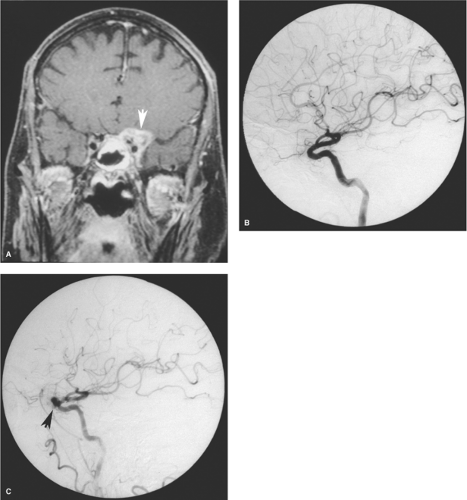
Left atrial myxoma may be complicated by or present with neurologic symptoms related to arterial obstruction by myxomatous emboli or intracranial aneurysmal rupture with subarachnoid or intraparenchymal hemorrhage (Fig. 18-13) (68,69,70). The destructive and invasive nature of the histologic findings with embolization of cardiac myxoma is ascribed to the high degree of elaboration of matrixmetalloproteinases and interleukin-6 by this tumor (71,72). Although this is a rare clinical presentation, it is a treatable disease that can be seen in otherwise healthy young patients. Systemic emboli from myxoma have been reported in up to 45% of patients, one-half of these going to the cerebral circulation (73). The diagnosis may be delayed due to the initially small size of the cardiac tumor (74). Angiographic features described in these patients include intraluminal filling defects, fusiform or saccular peripheral aneurysms, vessel occlusions, and delayed passage of contrast. Some peripheral aneurysms may be extremely subtle and could be missed if not looked for carefully on the late arterial or parenchymal angiographic phase.
Intracranial Dissections and Dissecting Aneurysms
Dissection of intracranial vessels, once considered an extremely rare disease (75), is now a more recognized entity representing as many as 3% to 7% of patients presenting with nontraumatic subarachnoid hemorrhage and possibly as many as 50% of aneurysms in the younger pediatric age group (76,77,78,79,80,81). In general this phenomenon is a devastating disease when the presentation is hemorrhagic with a mortality rate between 17% and 46% or more (78,82). Intracranial dissections are usually clinically idiopathic but have been reported after head trauma, electrocution, with syphilitic and other arteritides of the intracranial vessels, and with conditions such as polyarteritis nodosa, fibromuscular
dysplasia, and migraine (Figs. 18-14–18-17). Before 1950, more than one-half of recognized cases were associated with the meningovascular phase of syphilis (Heubner endarteritis), a disease seen now as a result of the AIDS epidemic. Some cases of intracranial dissection in older patients might be related to rupture of intramural arteriosclerotic plaques, but this is unlikely to be an explanation for this disease in younger patients (83).
dysplasia, and migraine (Figs. 18-14–18-17). Before 1950, more than one-half of recognized cases were associated with the meningovascular phase of syphilis (Heubner endarteritis), a disease seen now as a result of the AIDS epidemic. Some cases of intracranial dissection in older patients might be related to rupture of intramural arteriosclerotic plaques, but this is unlikely to be an explanation for this disease in younger patients (83).
Intracranial dissections are more common in the posterior than in the anterior circulation (Fig. 18-18). Patients typically present in two different ways, either with an acute subarachnoid hemorrhage related to rupture of the arterial pseudoaneurysm or with nonhemorrhagic complication such as suboccipital headache, mass effect, focal neurologic deficits, or ischemic complications (84). Patients presenting with nonhemorrhagic complications have a much better outcome, whereas those with pseudoaneurysms or subarachnoid bleeding account for most of the mortality associated with this disease. The media and adventitia of the intradural vertebral and carotid arteries are thinner than in the extracranial vessels. Therefore, arterial rupture with subarachnoid hemorrhage as a complication of intracranial dissections is more likely to develop than is the case with extracranial dissections. Intracranial vessels lack an external elastic membrane and have fewer elastic fibers in the media (85). Furthermore, the intracranial arteries are relatively deficient in vasa vasorum (86). Poor healing of intracranial vessels due to the lack of vasa vasorum may be part of the explanation for the greater likelihood of complications in many diseases of these vessels compared with extracranial vessels. In autopsy specimens of intracranial dissection with subarachnoid hemorrhage, extension of clot between the media and adventitia is seen with destruction of all three mural layers (77,87). Intracranial dissections usually affect the major vessel trunks of the circle of Willis, less commonly in the distal vessels (88).
Dissections in the anterior domain of the circle of Willis are less common than in the posterior, and have a predilection for the carotid terminus in children and young adults (76,89,90). Patients may have a variable course after the initial ictus of hemispheric ischemia. Massive infarction and swelling with herniation is the mechanism of death when the deterioration is rapid (91). When vascular occlusion is only partial, patients can survive with variable deficits (Fig. 18-19).
Intracranial dissections can be recognized angiographically by an irregular tapering of the vessel lumen, a linear filling defect within the lumen, retention of contrast within the wall of the vessel, or by the presence of an irregular aneurysm or pseudoaneurysm associated with focal luminal narrowing or irregularity (92,93,94). The angiographic and MRA appearance of the dissected vessel is sometimes described as a “pearl and string” sign, referring to the appearance of fusiform dilatation interrupted by segments of stringlike narrowing (Figs. 18-16 and 18-17). In a small number of patients, a double set of parallel lumens may be seen angiographically (75).
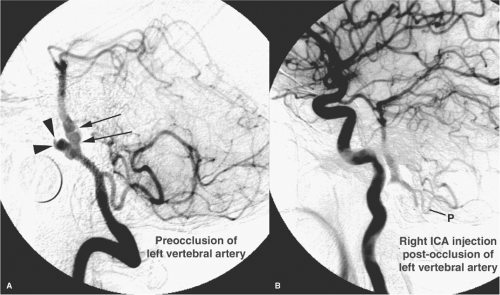
It is important to distinguish dissecting aneurysms from saccular bifurcation aneurysms, as the treatment may be different. Even with good-quality angiography, the signs of intracranial dissection may be subtle. The possibility of a dissection as a source for subarachnoid hemorrhage should be borne in mind during aneurysm search studies. The disease may be bilateral in the vertebrobasilar system in 5% to 10% of cases (95). Optimal treatment for dissecting extradural and intradural aneurysms sometimes involves altering or eliminating, if possible, the flow pattern in the vessel around the pseudoaneurysm. This hemodynamic alteration will suppress the inflow jet. This can be done by surgical clipping of the vessel or vessel reimplantation (96). Endovascular techniques include coiling of the pseudoaneurysm, stenting the vessel, trapping the pseudoaneurysm or occluding the parent vessel proximally, if the patient’s collateral flow will tolerate such a procedure (Fig. 18-20) (97,98,99,100,101,102,103,104,105). It is important to perform this occlusion as close as possible to the aneurysm to eliminate the possibility of continued anterograde flow through collateral vessels (106). Without treatment, dissecting aneurysms with subarachnoid hemorrhage have a rebleed risk reported to be between 30% and 70% (78,107,108,109). Therefore, immediate treatment of intracranial dissecting pseudoaneurysms is advised bearing in mind that ruptured dissecting aneurysms are extremely fragile with a significant risk of intraprocedural rebleeding. The possibility of spasm from subarachnoid hemorrhage related to this disease may affect the efficacy of collateral vessels where vessel occlusion is planned. Dissections without subarachnoid hemorrhage may do well without surgical intervention,
although some of these patients may progress to a chronic fusiform dilatation of the vertebrobasilar system (93) and may ultimately need surgical or endovascular treatment. An important discriminator for the clinical effects of the disease seems to be whether extension to the basilar artery from the vertebral artery occurs. When the disease is more confined and there is no subarachnoid hemorrhage, patients have a far more favorable outcome, and most will resolve their symptoms and angiographic findings within a few months (82,103,110,111).
although some of these patients may progress to a chronic fusiform dilatation of the vertebrobasilar system (93) and may ultimately need surgical or endovascular treatment. An important discriminator for the clinical effects of the disease seems to be whether extension to the basilar artery from the vertebral artery occurs. When the disease is more confined and there is no subarachnoid hemorrhage, patients have a far more favorable outcome, and most will resolve their symptoms and angiographic findings within a few months (82,103,110,111).
Saccular (Berry) Aneurysms
The cognomen “berry” for saccular intracranial aneurysms was introduced in 1931 by Collier (112) because of the fanciful resemblance of these aneurysms with their shining coats to berries hanging from the arterial tree. The perfect berry-shaped aneurysm with a spherical contour and confined neck is uncommon. Many aneurysms have a more complex structure, often with more than one distinct compartment or lobule and a neck that can be of variable size. A congenital defect in the arterial media may be one of the explanations for later development of this acquired disease.
Histopathologic examination demonstrates that unruptured small aneurysms have a thin wall measuring 30 to 150 μm in thickness, composed of endothelium and adventitia similar to that of the parent vessel (113). As the aneurysm enlarges, some portions of the wall become collagenized and thickened with endothelial cells, fibroblasts, and elastic fibers. More attenuated portions become points of potential rupture. When an aneurysm ruptures, it is usually assumed that it is the dome that has given way, but this is not always the case. The wall at the neck may sometimes be the most fragile segment. After the aneurysm ruptures and stabilizes, the rupture point is supported by weak fibrin nets for approximately the first 3 weeks. This correlates with the clinically observed period of high risk for rerupture. After 3 weeks, the wall becomes infiltrated with capillaries. Stronger collagen is incorporated into the healing wall with a diminishing risk of rerupture (114).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree