Fig. 13.1
Three-dimensional navUS of a recurrent low-grade glioma is displayed side-by-side with preoperative MRI in axial, coronal and sagittal reconstructions. Ultrasound and MR images correspond exactly to each other. The ventricles are outlined in red, the resection cavity of the previous operation is outlined in yellow and the falx is marked with an orange arrow
Contrast-Enhanced Ultrasound
In contrast-enhanced US, a contrast-enhancing agent is applied intravenously during the examination. Contrast-enhancing agents consist of microbubbles filled with gas. Microbubbles have a high echogenicity, thus they have a high ability to reflect echo waves. Moreover, the microbubbles begin to oscillate during insonation in response to the ultrasonic energy field and therefore emit waves with a characteristic (harmonic) spectrum of frequencies. This can easily be distinguished from tissue signal. Contrast-enhanced transcranial ultrasonography has been successfully used for perfusion imaging in cerebral ischemia and in the examination of intracerebral neoplasms. However, there is only limited data on the intraoperative use of contrast-enhanced US. So far, it has not yet reached intraoperative routine.
Applications
Tumor Visualization and Delineation
Conventional Two-Dimensional Ultrasound
Early studies on iUS focused on tumor visualization, characterization and delineation (LeRoux et al. 1992; van Velthoven and Auer 1990). We therefore know today, that most tumors show a higher echogenicity than normal brain tissue, irrespective of whether they show contrast-enhancement on MRI or CT. Surrounding edema can be distinguished from tumor tissue as it is less echogenic, but boundaries might be ill demarcated in some cases. Low-grade gliomas are usually homogeneous and have a moderate echogenicity. High-grade gliomas often appear inhomogeneous depending on tumor cysts, necrosis or intratumoral hemorrhage and are usually more echogenic than low-grade gliomas. Hyperechogenic areas within the tumor with an acoustic shadow deep to the lesion are due to calcifications and are more common in oligodendrogliomas. Metastases are highly echogenic and mostly well demarcated. They may appear either homogeneous or inhomogeneous depending on their histopathology. Tumor cysts are usually an- or hypoechoic like the ventricles with a small echoic rim surrounding the cyst (LeRoux et al. 1992). Intratumoral hemorrhage appears highly echogenic in case of a new hemorrhage. Two weeks after hemorrhage, the central parts of the hematoma become iso- to hypoechoic and 6–8 weeks later, the hematoma is isoechoic (Enzmann et al. 1981). As an example how different tumor entities may appear on iUS, please refer to Fig. 13.2.
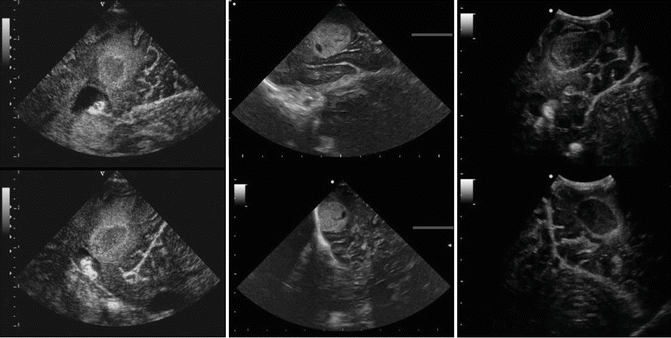

Fig. 13.2
Conventional 2D iUS showing different tumor entities in two perpendicular image planes. Left: Left occipital glioblastomas. The tumor is displayed as a hyperechoic rim surrounding a only slightly hyperechoic area that represents the central necrosis. The tumor is surrounded by a hyperechoic edema. Nearby, the dorsal horn of the lateral ventricle with its plexus and the falx are displayed. Middle: Right temporal metastasis of a renal cell carcinoma. Please note the rather homogeneous appearance of the rumor with a small hypoechoic cyst. Right: Left occipital metastasis of a renal cell carcinoma after intratumoral hemorrhage 3 weeks prior to surgery. The old hematoma is nearly anechogenic by now, surrounded by a hyperechoic rim
Contrast-Enhanced Ultrasound
Otsuki et al. (2001) presented a coded harmonic angio mode to visualize tumor vessels intraoperatively in a few number of patients. Harmonic imaging with US contrast agents improved the signal-to-noise ratio between brain parenchyma and vasculature in these patients. Kanno et al. (2005) studied 40 patients with an US contrast-enhancing agent during operations. Power Doppler US with contrast enhancement provided better data on the precise real-time position of the tumors and their relationship to adjacent vessels than US obtained before the injection of the contrast agent. Contrast-enhanced power Doppler correlated well with digital subtraction angiography. Engelhardt et al. (2007) performed contrast-enhanced imaging with a low mechanical index during resection of different brain tumors in a small number of patients. Scans were inspected in real-time during tumor resection, and radiofrequency ultrasound data was analyzed off-line. The perfusion pattern of the tumor was semi-quantitatively evaluated and compared with that in normal brain tissue. The distinction of tumor from non-infiltrated parenchyma was facilitated by the influx of contrast agent.
Three-Dimensional Ultrasound
Unsgaard et al. (2005) examined the ability of 3D US to delineate gliomas and metastases in 28 operations by taking 85 biopsies from tumor borders. US findings were in agreement with histopathology in 74% of low-grade astrocytomas, 83% of anaplastic astrocytomas, 77% of glioblastomas and 100% of metastases.
Lesion Localization/Planning the Optimal Approach
Numerous reports on the utility of iUS as a tool for intraoperative navigation exist. Moiydi and Shetty (2011) evaluated the use of conventional 2D iUS in 77 cases of both cranial and spinal lesions. In their hands, iUS was useful in identifying and delineating lesions in 100% of cases, in planning the durotomy in 78% of cases, in planning the corticectomy in 85% of cases and in visualizing adjacent structures in 96% of cases, respectively. They concluded that iUS is a useful tool in planning various stages of tumor resection. In a study comparing conventional iUS and navUS, we could show, that orientation can even be improved by navigating the US probe, thus facilitation anatomical understanding (Miller et al. 2007). Moreover navUS was useful in planning the correct transsulcal approach to subcortical lesions in eloquent areas (Zhou et al. 2009) and in visualizing structures adjacent to tumors. This was most obvious in cystic gliomas (Enchev et al. 2006), where an opening of a cyst during surgery might lead to a local tissue shift. With the aid of navUS, vessels that might be encased or displaced by tumor tissue could be visualized and landmarked as reported by our group (Sure et al. 2005). Rygh et al. (2006) showed that 3D US was helpful in visualizing hidden vessels adjacent to and inside the tumor thus facilitating tailored approaches and increasing the safety of biopsies. Similar to navigated 2D US, navigated 3D US solved the orientation problem experienced with conventional 2D US in neurosurgery as it allowed axial, coronal and sagittal reconstructions and a direct comparison of iUS and preoperative MRI in the same plane of view (Miller et al. 2012; Unsgaard et al. 2002). Three-dimensional US could be used as a navigational tool even without preoperative MRI in cases where no preoperative MRI was available, an intraoperative brain shift occurred or in case of a failure of the navigation system (Miller et al. 2011; Peredo-Harvey et al. 2012).
Resection Control
Two-Dimensional Ultrasound
The use of iUS for resection control during surgery of intracerebral tumors is documented in numerous reports. However, larger randomized controlled trials are lacking.
In an early study by LeRoux et al. (1994), US tumor volume estimates tended to be non-significantly larger than T1 gadolinium-enhanced volumes on preoperative MRI. The authors concluded that iUS might therefore aid in detecting areas of tumor extension beyond margins of blood-brain barrier breakdown. Tumor volumes on T2 weighted MRI were larger than US volumes except for low grade gliomas, again this was not statistically significant. US images helped to differentiate edema from solid tumor and normal brain. Overall, the authors concluded that, US information might help to enhance tumor resection. However, there were difficulties in estimating tumor volumes in previously treated lesions where gliosis was present.
Two studies compared sonographic imaging results after resection to histopathology by taking probes from various points at the resection margin. Woydt et al. (1996) acquired a series of 78 biopsies from the resection cavity under continuous sonographic control at the end of surgery in 45 patients with gliomas. 47 out of 53 biopsies (89%) taken from tissue sonographically suspected as tumor revealed solid tumor tissue of which 72% were microscopically assessed as unsuspicious by the surgeon. Six samples contained tumor infiltration zone. Six of twenty-five biopsies taken from sonographically tumor-free borders were diagnosed as tumor tissue. Of 34 cases with gross total removal according to the surgeon’s assessment 25 showed sonographic signs of residual tumor tissue, which was confirmed histologically as solid tumor tissue in 22 of these cases. The authors concluded, that iUS following resection of supratentorial gliomas could detect residual tumor tissue with high specificity and thus improve gross total resection. In a second study by Chacko et al. (2003), iUS was useful in defining lesion margins in 71.4% of 35 cases of intracerebral lesions, however in previously radiated cases the margins were ill-defined. 97% of samples from the resection margins were correctly identified as tumor. There were two false negative cases (3%). However, there was a high rate of false positive results (46% of histologically negative probes).
Hammoud et al. (1996) performed a prospective study of 70 patients with a variety of different intraparenchymal brain lesions in 1996 to evaluate the efficacy of iUS in localizing and defining the borders of tumors and in assessing the extent of their resection (as compared to postoperative MRI). In gliomas without previous therapy, tumors were well localized in all patients, margins were well defined in 15/18 cases and the extent of resection was well defined in all patients, respectively. In patients with gliomas that had undergone previous therapy and showed post-radiation changes, the extent of resection was poorly defined. All metastatic lesions were well localized, had well-defined margins and the extent of resection was well defined on iUS.
In a comparative study by Gerganov et al. (2009), iMRI and 2D iUS were compared for resection control in 26 patients with various tumor entities. The authors showed that small tumor remnants (<1 cm) were not detected by the examiner in 2 of 21 patients with tumor remnants identified on 1.5 T iMRI. Suspicious signals on iUS were misinterpreted as tumor remnants in another two cases. A statistical analysis was not performed in this study.
In a recent study by the same group on resection control for low-grade gliomas, tumor borders prior to resection were clear on iUS in 9 of 11 patients as compared to 10 of 11 cases on iMRI (Gerganov et al. 2011). Image quality diminished during surgery, leading to difficulties in interpretation. One tumor remnant was not identified on iUS, and one artifact was falsely interpreted as a tumor remnant. Intraoperative MRI appeared superior for resection control in these cases.
Contrast-Enhanced Ultrasound
There is very little data on the use of contrast-enhanced US for resection control. Preliminary data in a very low number of cases suggested that remaining tumor tissue could be distinguished from surrounding brain tissue more easily as compared to conventional US (He et al. 2008).
Three-Dimensional Ultrasound
Rygh et al. (2008) compared 3D US to histopathological findings at different stages of resection. They could show a very high sensitivity (95%) and specificity (95%) to delineate tumor before resection. During resection, sensitivity dropped slightly to 88%, but specificity was only 42%. After resection sensitivity dropped to 26%, but specificity increased to 88%, while both positive and negative predictive value were 62%.
To date, only one study has compared low–field 0.2-T iMRI and 3D US in seven patients only (Tronnier et al. 2001). The ability to detect tumor was comparable in both imaging modalities in high-grade gliomas and metastases, whereas in the case of a low-grade glioma US was more helpful. The reliability of the ultrasound diminished during tumor resection, so that tumor remnants were not at all or were poorly visible in four patients.
The value of 3D US was evaluated by correlating 3D US and postoperative MRI in a small randomized blinded prospective study by Rohde and Coenen (2011). Three-dimensional US correctly identified tumor remnant in five of seven patients (71%) and correctly confirmed complete tumor removal in three of five patients (60%). Overall the intraoperative situation was correctly visualized in 8 of 12 patients (67%).
Figure 13.3 shows an example of a 3D US of a glioblastoma prior to and after resection.
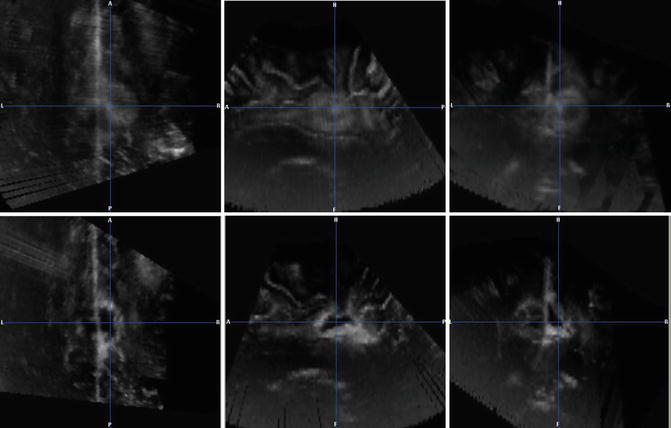

Fig. 13.3
Three-dimensional US of a left parasagittal glioblastomas (left to right: axial, sagittal and coronal views). Upper row: Glioblastoma before resection. Lower row: Resection cavity after resection. Please note, that the resection cavity is partly filled with blood which appears very hyperechoic and should not be mistaken for tumor. The tumor tissue prior to surgery appeared less echogenic than blood
Visualizing and Compensating Brain-Shift
Deformations of the brain during surgery may be due to loss of cerebro-spinal fluid, swelling or retraction. These deformations may range from 2 to 25 mm depending on the size, the location and the kind of tumor (Lindner et al. 2006).
Several groups worked on compensating brain shift via a co-registration of iUS and preoperative MRI (Coenen et al. 2005; Reinertsen et al. 2007). Coenen et al. (2005) defined ultrasound landmarks in the vicinity of the tumor and the nearby fiber tracts and used a sequential landmark registration to assess fiber tract deformation. They could show on postoperative diffusion-weighted imaging that the actual fiber tract position at the end of surgery corresponded to the sonographically predicted fiber tract position. Reinertsen et al. (2007) validated a vessel-based registration technique for the correction of brain shift. There are multiple reports on other possible co-registration- and deformation algorithms, however large patient trials are still lacking. Further studies are needed to create robust deformation models to allow fusion of MRI and iUS.
Guidance of Biopsies
Tsutsumi et al. (1989) first achieved real-time imaging in single burr hole procedures in 1989. Histologic diagnosis could be established in 87–95% of cases of US-guided tumor biopsies via a single burr hole trepanation (Benediktsson et al. 1992; Di Lorenzo et al. 1991; Lunardi et al. 1993; Strowitzki et al. 2000).
Ultrasound for Intra- and Extramedullary Spinal Tumors
As shown by our group and others, iUS can be used for a differential diagnosis of different intradural spinal tumors. While intramedullary tumors showed an inhomogeneous pattern, extramedullary tumors were displayed with a homogeneous signal on iUS. Extramedullary tumors were sharply demarcated from the myelon in all cases. Moreover, iUS proved to be useful in displaying the size and exact location of the lesion, thus allowing adjusting the extension of the bony opening (Regelsberger et al. 2005; Zhou et al. 2011).
Discussion
Technical Considerations
iUS allows real-time imaging, thus giving a direct update of the intraoperative anatomy. Only minimal preparation time is needed for scanning. As compared to iMRI, it is less time-consuming, easily repeatable and far less expensive. No special operating room or operating equipment is needed. Ultrasound equipment and disposables are relatively low in cost and basically everywhere available. Moreover, there is no irradiation with the technique. The operating surgeon can perform the examination and can gain an immediate feedback on the intraoperative situation as well as real-time blood flow information. Intraoperative US can even be combined with neuronavigation or endoscopy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






