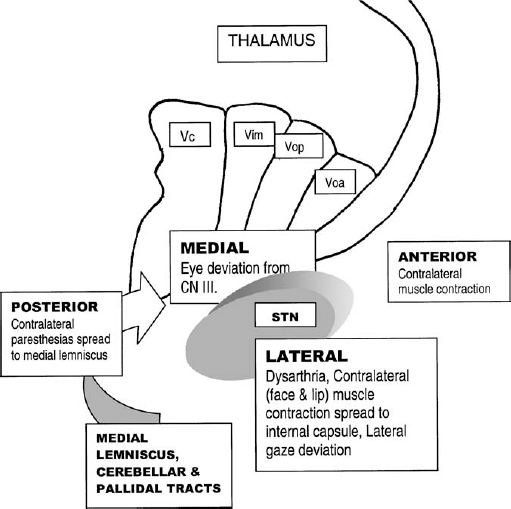
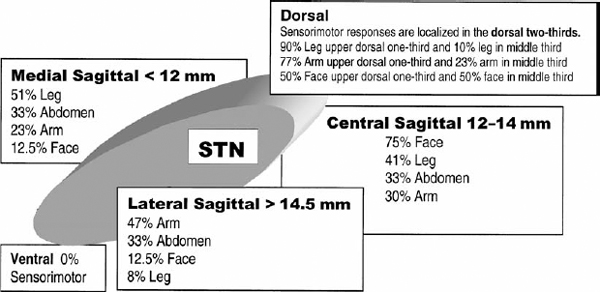
20 The optimal choice of imaging and localization for stereotactic surgery for movement disorders remains controversial. The debate as to whether microelectrode recording (MER) is necessary for optimal results is still active between groups involved in the surgical treatment of movement disorders. Some teams consider anatomical localization by radiographic imaging and macrostimulation to be sufficient, whereas others think that further electrophysiological localization is necessary to yield the best possible results. Both sides are strong proponents of electrophysiological confirmation of the target and are aware of the disasters that can result when these techniques are not used.1,2 The point in question is the degree of electrophysiology needed to optimize results. The answer to this salient question would require conducting a randomized prospective study of the clinical outcome following surgery with or without MER. To date, this study has not been reported. Such a study would have to be performed within the same institution using both techniques in a significantly large number of patients to be able to draw statistically significant conclusions. Failing this, analyses of anatomical, electrophysiological, and outcome data are being debated. In 1961, Albe-Fessard et al3 provided the first report of MERs taken in humans during stereotactic surgery. During that decade, the technique was incorporated by many others as part of stereotactic procedures to define the borders of anatomical structures and targets.4–6 These original studies were performed with larger tipped bipolar electrodes (50 μm) that recorded mainly multiunit activity and local field potentials.7 Currently, the science of MER has advanced to the point where many consider it a routine technique in stereotactic and functional neurosurgery. Bertrand et al8 and Jasper and coworkers9 reported using two to three μm tipped electrodes and were probably the first to record predominantly single units in the human thalamus. Other groups have described MER in the activity of the human thalamus and consider it a routine technique for thalamotomy.10–14 In 1966, Laitinen et al recorded the impedance and phase angle of brain tissue of 65 patients undergoing stereotactic surgery.15 Impedance provides additional intraoperative anatomical information but does not allow three-dimensional mapping or electrophysiological mapping. Pallidotomy is a long-standing surgical procedure that has been rediscovered. Combined with the development of modern imaging and electrophysiological techniques, pallidotomy provides an effective treatment for Parkinson’s disease (PD). Multiple functional centers that perform pallidotomy use MER routinely to generate intraoperative target refinement by electrophysiological mapping to correct for anatomical variability and errors in targeting.16–34 Methods for the physiological localization of the subthalamic nucleus were based on similar work in the parkinsonian monkey.35 The first subthalamic deep brain stimulation procedures performed in human subjects were reported in 1993.36 Recordings of neuronal activities contributed greatly to identifying the subthalamic nucleus and determining the target for chronic stimulation within it. The majority of investigators experienced in performing subthalamic surgery use MER as the reference targeting method.37–42 A dramatic improvement in parkinsonian symptoms can be obtained using bilateral high-frequency subthalamic stimulation.41 It has been shown to be a safe and effective method for treating medically refractory idiopathic PD.43 Subcortical target localization is not only a science but also an art that improves with experience.44 The surgeon must know how much importance to place on neuroanatomical versus neurophysiological data during every operation. Each patient’s individual brain map must be constructed in the surgeon’s mind, to determine the optimal location for lesion or DBS lead placement. The sources of error can be varied and multifactorial, with the consequences of error being serious. In attempting to compensate for the lack of direct anatomical visualization of the brain, the stereotactic and functional neurosurgeon is faced with problems of considerable magnitude.45 We believe all of these introduce sources of error, which can affect optimal target localization. As long as these errors exist, MER can serve as a quality assurance procedure that can instantaneously determine spatial accuracy at each step.46 There are two main sources for errors of localization—imaging errors and individual anatomic variations—and they can be additive.34 Imaging errors introduce distortion during the image-acquisition phase and exist with any radiographic method used to identify the target. Much controversy surrounds the use of MER and the role of distortion in magnetic resonance images in reducing the accuracy of lesion placement. MR imaging has been widely reported to introduce distortion resulting from inhomogeneity in the magnetic field and nonlinear magnetic field gradients.47,48 Computed tomography does not create distortions, but the accuracy of the CT- or MR imaging–guided stereotactic system is dependent on the scan slice thickness, the interslice spacing, and the size of the target itself.49 Reports of submillimeter accuracy while using 1 to 3 mm slice thickness reflect a failure to understand the spatial relationships involved. Ventriculography is associated with imaging distortions because of distortion and shifting of the third ventricle. It can provide intraoperative anatomical correction to a limited degree. Of clinical consequence, ventriculography prolongs hospital stays and is frequently accompanied by unpleasant side effects.50 Complications related to ventriculography include generalized seizures, disorientation, and confusion.51 The surgeon’s choice of landmarks introduces yet another source of error. It is well documented that there is substantial individual variation in the anterior and posterior commissure–based landmarks of subcortical nuclei.45,52,53 As a result of this variation, coordinates are typically reported as a range rather than as exact numbers. Even the choice of the location of the AC will vary, as some neurosurgeons use the posterior edge, others use the center, and still others use the anterior edge of the AC as the start of the AC–PC line. Location and identification of the PC carries even greater variability. Excessive reliance on a standardized formula for target selection is therefore inappropriate. There is increasing use of direct anatomical targeting as image quality improves.38,41,54–59 However, this will still require correction of target choice based on physiological characteristics. Individual anatomic variations based on the patient’s age, size, position at the time of surgery, and brain shift during surgery all play a role for possible sources of error. The thalamocapsular border shifts in accordance with the width of the third ventricle. The thalamic border is more lateral when the ventricle is wider.60 Variability is greater in the globus pallidus than in the thalamus because the GP is farther away from the midline and has a more complex shape.34 The oblique anteromedial to posterolateral course of the internal capsule also has been reported to cause variability in target location.29 Lehman61 conducted an anatomic variability study on the related subthalamic structures using 40 brain specimens. Results of the study revealed a relationship between structures in the subthalamic nucleus varying in a mediolateral direction based on the inter-commissural distance. A further source of error may derive from use of the human brain reference atlas. It has been reported by Lozano et al31 that there are remarkable differences between the original edition of the Schaltenbrand and Bailey atlas62 and the more recent Schaltenbrand and Wahren atlas.63 The 18.5 mm lateral map in the original edition is identical to the 20 mm lateral map in the later edition. Consequently, the location of the AC in the Schaltenbrand and Bailey atlas is 2 to 3 mm dorsal to that shown in the original atlas. It is also well known that each of these stereotactic atlases produced in the 1950s is routinely used today and is based on a single anatomical subject that creates an inherent source of error. A computer overlay of these atlases on MR images or CT scans is a waste of time, for no matter how the atlas images are stretched to try and fit the individual patient’s anatomy, there will be inaccuracies. Human or mechanical imprecision can introduce sporadic and random sources of error, which can be very difficult to pinpoint. The stereotactic frame itself, with its metal components, may create errors or distortions during image acquisition.64 Metal frames can bend to variable degrees depending on the magnitude and direction of the force. The Leksell arc can have up to 2 mm variability in the mediolateral direction depending on the snug fit to the head frame and the degree of stress placed on the arc when locking. An ever so slightly bent outer guide tube can quietly hide another compounding mechanical imperfection. We have noted anywhere from 2 to 4 mm deviations from the target in a random direction depending on which way the guide tube is secured into place, in relation to the bend in the tube. Fortunately, many of these errors will cancel each other out, but it is when they do not that problems arise. Despite the improvements in initial targeting by MR imaging–based direct visualization and mechanical quality assurance checks, we still believe functional stereotactic ablative and augmentative procedures are not only anatomical exercises but also true physiological targets. Microelectrode recording Methods for the physiological identification of the borders of the posterior ventral segment of the globus pallidus internalis have been described by Vitek et al.65 In pallidotomy one must avoid the optic tract at the ventral border of the globus pallidus and the internal capsule at the medial and posterior border of the globus pallidus. By physiologically confirming these boundaries, complications associated with coagulating these structures are avoided. The patient reports phosphenes in the contralateral visual field with stimulation, and MERs demonstrate bursts of action potentials when a strobe light is flashed into the patient’s visual field. The characteristic absence of cellular activity identifies the internal capsule along with motor or sensory responses with stimulation.66 The subthalamic nucleus is relatively small and deeply situated, surrounded by brain structures such as the internal capsule, medial lemniscus, zona incerta, ventral thalamic nuclei, and substantia nigra pars reticulata. Precise boundaries should be defined electrophysiologically to minimize potential stimulation-induced effects of chronic DBS.40 Lozano et al66 strongly believed that MER studies are important to define the target to optimize the lesion epicenter so that when the predicted lesion volume is taken into account, injury to adjacent eloquent structures can be avoided. Tasker stated that “even if one could visually inspect the brain structure in which a probe lay, there are still considerable variations in the somatotopographic and modality content of an identified anatomical structure.”67 Tasker emphasized yet another reason to favor MER. Cell clusters have been identified by several groups that alter discharge rates as a result of related movements.66,68 These findings suggest a somatotopic organization of the human globus pallidus. Similar results have also been reported in primates.69 In using the somatotopical information that MER provides, lesions could be made in areas containing neurons subserving the most disabled limb. Somatotopical organization of the subthalamic nucleus also has been well documented.70 A recent study demonstrated a distinct topographical representation of the leg, arm, and face such that the leg is located in the upper dorsal one third and centromedial portion. The face is located in the dorsal two thirds and central portion, and the arm is located in the dorsal two thirds but lateral region of the subthalamic nucleus (Fig. 20–1). Cells whose discharge rate is modulated by passive or active movements identify the so-called sensorimotor territory of a nucleus. Multiple groups have documented localization of a target based on sensorimotor findings.17,29,38,40,41,65,70,71 Primate studies have clearly identified the dorsolateral subthalamic nucleus as the motor territory, and electrodes should be placed in this region for maximal efficacy.72,73 Recent human clinical studies using MERs have also mapped the sensorimotor region of the subthalamic nucleus to the dorsolateral two thirds of the nucleus.70 Lesions and DBS placed outside the sensorimotor areas are going to produce suboptimal results. Microelectrode recording allows for mapping structures in three dimensions. The subthalamic nucleus is a biconcave lens-shaped structure. Its dimensions are ~10 mm rostrocaudal, 10.5 mm mediolateral, and 7 mm dorsoventral.41 This small obliquely oriented structure with an ovoid shape makes it difficult to target. Proponents of MER typically define the three-dimensional anatomy by placing two tracks in the same parasagittal plane to define the anteroposterior aspect of the target.65 The mediolateral plane is defined by a third trajectory laterally. The minimum number of tracks to outline a three-dimensional target is three.74 The recording of cellular activity by microelectrodes currently is the most exquisite localization method identifying a precise physiological target. FIGURE 20–1 Somatotopic organization of the subthalamic nucleus as documented by Rodriguez-Oroz et al. The so-matotopographical representation of the leg, face, and arm is demonstrated in percentages in a medial to lateral direction, respectively. (Source: Rodriguez-Oroz MC, Rodriguez M, Guridi J, et al. The subthalamic nucleus in Parkinson’s disease: Somatotopic organization and physiological characteristics. Brain. 2001;124(pt 9):1777–1790.) Macrostimulation in the globus pallidus is highly variable in its effect on symptoms and often has delayed effects.75,76 This variability extends to microstimulation. Lozano et al31 noted instances when patients reported visual sensations with microstimulation but no visual evoked potentials were recorded. Occasionally no electrically evoked visual sensations were found despite good recordings of flash potentials with MER. The lack of easily identifiable structures with micro- or macrostimulation strengthens the case in favor of MER being necessary to identify structures by patterns of neuronal spikes and firing rates.77 Targeting the ventral intermediate (Vim) nucleus in the human thalamus is described by Ohye and Narabayashi.78 Ablation or stimulation of the physiologically defined Vim abolished tremor. Extracellular recordings and electrical stimulation were performed in 56 patients for thalamotomy. Ninety percent of neurons in the Vim responded to a passive or active movement of a joint. Evidence for somatotopic representation in the Vim nucleus exists.79 Tasker et al80 described macrostimulation and MER with microstimulation. Stimulation for the thalamus identifies a wide range of structures and because of current spread permits the identification of a spectrum of structures at variable distances from the probe. Stimulation in the thalamus is most useful in plotting a map of the data and defining the rostral margin of the ventral caudal nucleus. The lesion is planned to encompass the identified kinesthetic and voluntary tremor cells but avoid the tactile cells and internal capsule that have been predetermined with microstimulation. Volume conduction extending over a radius of 3 mm with Tasker et al’s technique81 blurs the results because macrostimulation cannot distinguish the paresthetic responses induced by stimulation of the medial lemniscus, tactile thalamus, or patient misinterpretation of internal capsule stimulation. Bejjani et al41 described possible effects seen with acute stimulation at the borders of the subthalamic nucleus (Fig. 20–2). Microstimulation provides very limited information for subthalamic localization. Stimulation of the subthalamic nucleus itself in the parkinsonian patient may show improvement in contralateral rigidity, akinesia, or tremor. An encouraging sign of subthalamic localization is when stimulation induces contralateral dyskinesias. There is, then, a wealth of information regarding stimulation and target localization. More information is available from microstimulation when combined with macrostimulation. The smaller the target, the greater the need for MER and microstimulation to define the target before placing a macrostimulation lead over 10 times the diameter of the microelectrode. Localization with macrostimulation can provide information to move the probe, but after creating a lesion tract, it may be very difficult to move more than 2 to 3 mm to a new tract, as there is a tendency for the brain to shift and the probe end up in the same place (observed on fluoroscopy). Therefore, fine movements after macrostimulation may be difficult.
Is MER Necessary in Movement Disorder Surgery? The Case in Favor
DIANE K. SIERENS AND ROY A. E. BAKAY
Sources of Error
Why Not Pick the Physiological Target?
What Does Stimulation Have to Offer?
Neupsy Key
Fastest Neupsy Insight Engine