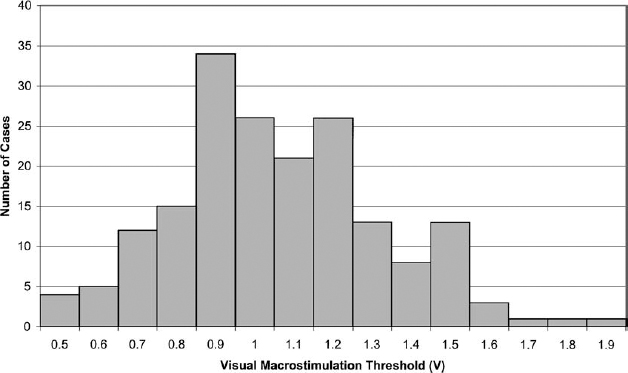
5 Macrostimulation and microelectrode recording techniques have been used to identify and verify intraoperatively the location of surgical targets, although the necessity and methodology of both remains controversial. Despite the availability of sophisticated image-based targeting software, the use of some form of electrophysiological target refinement and verification is important in preventing damage to critical structures. This chapter details our electrophysiological technique of macroelectrode stimulation and impedance monitoring in the posteroventral pallidotomy (PVP). It includes a retrospective review of our methods in 203 cases and the complications in 1156 consecutive cases. The techniques of macrostimulation generally can be divided into two objectives: localization of the effective target site for alleviation of the symptoms of Parkinson’s disease and determining the proximity to critical structures such as the optic tract and internal capsule. High-frequency stimulation inactivates neuronal activity in immediately adjacent tissue. With proper targeting, this suppresses clinical symptoms, thus verifying that the electrode is in the optimal position. For instance, dramatic and immediate tremor suppression is achieved with high-frequency stimulation of the ventrointermedius thalamus. This degree of specificity for target optimization in the posteroventral pallidotomy, however, is not achieved by the use of high-frequency stimulation.1 Nevertheless, the inactivation of adjacent tissue with the formation of a functional lesion forms the basis for high-frequency deep brain stimulation implants, either in the thalamus, the globus pallidus, or the subthalamic nucleus. Macrostimulation in the posteroventral pallidotomy can also activate fibers of the optic tract and internal capsule from a relatively remote site. Measuring the minimum stimulus intensity to elicit these effects can be used to gauge the distance of the electrode tip to these critical structures. As the electrode is advanced through the globus pallidus, the threshold decreases as the structure is approached. Thus, macrostimulation is an effective real-time tool for localization of the electrode tip relative to these critical structures.2,3 Knowledge of whether the electrode tip is within white or gray matter, based on relative changes in impedance, can provide additional verification of electrode position.2–8 Data from two groups of patients are presented in this chapter: those collected for methods and those for complications. The methods data were collected from every PVP case retrospectively, beginning with current patients and excluding patients with incompletely recorded information. A total of 203 cases of PVP were included in the methods analysis. Some of these patients also underwent other simultaneous stereotactic procedures, including thalamotomy (n = 17) and bilateral pallidotomy (n = 10). These patients had advanced Parkinson’s disease with significant dopaminergic side effects, including dyskinesia and fluctuating “on/off” periods. The typical patient was advanced in age, had a prolonged disease course, and had marked clinical benefit from the PVP The clinical outcomes for these patients are not presented here, but several randomly selected subsets of these patients have been studied in detail with significant clinical improvement from these pallidotomies (unpublished data).9 The complications data presented in this study are collected from research in progress and will be reported in more detail elsewhere. The majority of patients in the methods group are also in the complications group. In the complications group, 796 consecutive patients who underwent PVP were studied. Of these, 272 patients had simultaneous bilateral pallidotomy, and 88 patients had staged bilateral pallidotomy. Thus, a total of 1156 posteroventral pallidotomies were performed. Additionally, 108 of these patients underwent combined pallidotomy and thalamotomy. Every patient underwent macrostimulation guidance just prior to lesioning. In addition, 73 patients underwent microelectrode recording in the posteroventral pallidotomy. A total of 110 microelectrode recordings were performed in both 36 unilateral and 37 simultaneous bilateral PVP. There is an obvious independence of complications between sides in bilateral PVP. Therefore, complications were calculated based on cases, not patients (bilateral pallidotomy had two cases regardless of whether it was simultaneous or staged). There are several elements of the surgical method that are relevant. More detailed methods have been described previously2,10 A Cosman-Roberts-Wells (CRW; Burlington, MA) arc system was used in all cases. Preoperative MRI-based targeting was conducted on a 1.0 or 1.5 T Siemens scanner (Erlagen, Germany) on axial images parallel to the anterior commissure-posterior commissure (AC–PC) line, and on multiplanar reconstructed images in the sagittal and coronal planes. Patients received no sedation prior to surgery. Some patients received light propofol sedation during the drilling of the burr hole. These patients seemed to awaken from this fast-acting anesthetic refreshed and clear minded. In addition, mean arterial blood pressure was maintained below 90 mm Hg with a nitroglycerin drip to preclude intracranial hemorrhage and cardiac events. A 13 mm burr hole was drilled from 4.5 to 5.0 cm lateral from the midline, providing an oblique lateral to medial approach. This was important for several reasons. First, the electrode trajectory largely avoided the internal capsule, possibly preventing speech, cognitive, and motor deficits. Second, an angled approach is necessary for convergence of the electrode on the internal capsule and for the usefulness of the sequential motor macrostimulation thresholds approaching and intercepting our window range of values associated with ideal proximity to the internal capsule and optic tract. If a vertical approach was used, this convergence rarely occurred. Third, because of the dimensions of the globus pallidus internus and the medial sweeping internal capsule as it enters the cerebral peduncle, an oblique lateral to medial approach allowed the lesion to be tucked more tightly within the globus pallidus internus next to the internal capsule. The measurement of impedance and macrostimulation thresholds was dependent on the type of equipment used. In this study all measurements and lesions were conducted with a Radionics (Burlington, MA) RFG-3C generator, using a Radionics 1.8 mm diameter electrode with a 2.0 mm exposed tip. This equipment used constant voltage stimulation rather than a constant current mode. A disposable Radionics ground pad was placed on the right posterior thigh. Prior to stimulation, patients were educated about expected visual patterns. Patients typically described a “sunset” effect, or white “spots” of light, and some described colored effects. The location of these visual effects indicated the position of the electrode with respect to the optic tract. The expected position of visual effects was in the contralateral inferior visual field. If the effects were widespread, the stimulation intensity was reduced, typically producing a more localized visual field perception. If the effect was in the superior field, the electrode typically was placed too laterally and deep to the optic tract. Some patients had difficulty specifying the visual quadrant of the effects. However, this did not affect the threshold measurement. Stimulation of the optic tract was best conducted in a pitch black room. All lights were turned out, and the windows were covered. Patients were instructed to close their eyes. High-frequency stimulation was superior to low-frequency because of the sustained and more easily identified visual effects. The stimulation in this study was at 100 Hz with 1 msec pulses. Increasing the stimulation frequency or pulse duration increased the current density and thus lowered the measured threshold of detectable responses. The stimulation trials were brief, typically less than 2 sec, because of the rapid accommodation and disappearance of the visual effect if the stimulus was-sustained. The stimulus voltage was rapidly brought to the desired level and maintained throughout the trial. The first several trials were at suprathreshold levels, so that the patient could easily and reliably identify the stimulus effect. However, exceedingly high voltages caused an uncomfortable feeling or in some cases elevated blood pressure. Unlike thalamotomy, stimulation for these brief periods had little to no effect on the signs and symptoms of Parkinson’s disease or on speech. Macrostimulation within the globus pallidus elicited motor effects because of the adjacency of the electrode to the corticospinal and corticobulbar fibers in the posterior limb of the internal capsule. High-frequency stimulation (100 Hz) typically produced tetany of the hand and face. Stimulation at 2 Hz with 1 msec pulses elicited synchronous twitches of the face and tongue, and occasionally the hand. The tongue was the most reliable site where these twitches could be visually observed, typically on the more lateral aspects. The stimulus was sustained as needed to obtain a threshold and started at suprathreshold voltages to reliably identify the synchronous twitch. Then the stimulus was reduced until the stimulus was no longer visible. This was repeated until a reliable and consistent minimum voltage was measured, termed the motor threshold voltage. On occasion the twitch was difficult to identify. The tongue should be relaxed as much as consciously possible. The jaw was pushed open by the observer, and on occasion the patient was asked to swallow or was given small sips of water and instructed not to stick out his or her tongue. The Radionics RFG-3C measures impedance when the stimulation is off but in stimulus mode. It generates a 50 kHz constant current stimulus from which it measures the voltage and calculates the impedance, which takes into account the resistive and capacitive aspects of the electrode–tissue junction. Although there is a fair degree of variability between patients in absolute impedance measurements, the relative change within an individual is highly indicative of changes between gray matter, white matter, cerebrospinal fluid (CSF), and hemorrhagic blood. For these reasons, the impedance can provide important feedback information about the position and environment of the electrode tip. In this study, the impedance was recorded in increments as the electrode was advanced toward the final lesion site. In addition, the audible tone produced by the RFG-3C provides continuous feedback about changes in impedance. The impedance measurements were collected when the stimulus was set at 100 Hz. Although setting the stimulation frequency at 2 Hz theoretically should not change the impedance measurement equipment, there was a small increase in impedance measured when the stimulus was changed from 2 to 100 Hz. The impedance would stabilize typically within 20 sec of stopping electrode advancement, at which point the measurement was recorded. However, if the electrode position was unchanged over several minutes, the impedance would increase slightly. If the impedance suddenly dropped more than 30%, an intracranial bleed was suspected. If the electrode was retracted and advanced along a supposedly new tract, but the impedance was markedly lower than the first track, then the electrode was retracted and suspected of following the old trajectory. In this case, new CRW arc angles were used, and a different cortical entry point was chosen. The initial target described in this study is the most ventral site of the electrode, where the most ventral extent of the lesion was generated. To guide the electrode to this anatomical site, the visual and motor macrostimulation thresholds as well as the impedance were collected as the electrode was advanced incrementally. Impedance monitoring was continuous throughout the electrode advancements. Motor macrostimulation typically began 6 to 7 mm above the initial target. The electrode was advanced in 0.5 to 1.0 mm increments, and the motor macrostimulation thresholds were obtained. If the thresholds were appropriately converging within the normal range, the electrode continued to be advanced. However, if values were too low, the electrode was retracted and moved more anterior and/or lateral. If the thresholds were too high, the electrode was still advanced. When the initial target was too dorsal, as the electrode reached appropriate depths based on visual macrostimulation, the motor threshold would enter the normal range. Visual macrostimulation thresholds were obtained beginning ~2 to 3 mm dorsal to the initial target. If the thresholds were inappropriate or unobtainable, an anteroposterior (AP) and lateral x-ray was obtained, coregistered with the previously acquired AP and lateral third ventriculogram, and the position of the electrode was identified with respect to the floor of the third ventricle, which corresponds to the level of the optic tract and the mamillary body. If the electrode position was too dorsal but the trajectory looked appropriate, then the electrode was still advanced. If there was a gross error in the trajectory or position, a new trajectory was selected. Near the target the electrode was advanced slowly in 0.5 mm increments, with measurement of visual and motor macrostimulation thresholds as well as impedance. If the motor macrostimulation threshold was within the acceptable range, then the electrode was advanced until one or the other of the following occurred: either the visual macrostimulation threshold was 0.9 V or the impedance suddenly dropped 10 to 20% over 0.1 to 0.2 mm. At this location, AP and lateral x-rays were acquired and coregistered with the third ventriculogram to identify the position of the electrode. If the combination of motor and visual macrostimulation thresholds as well as the impedance and the radiologic site of the electrode were acceptable, then radio frequency lesioning commenced with sequential testing of visual fields, acuity, speech, and appendicular motor functions, including repetitive finger tapping and alternating dorsoplantar flexion of the feet. At the most ventral advancement of the macroelectrode, a test lesion was first generated at 67°C. The patient was instructed to read several sentences for the left PVP, or describe faces for the right PVP Lesioning was aborted if the patient had difficulty reading, especially during the left PVP. If the patient described a blurring or waviness in the peripheral vision or disappearance of the last letters of a word at the end of a sentence on the right, or developed slurred speech, the lesion was aborted, and the electrode was withdrawn at least 1 mm. A lesion at this temperature extends laterally but not lengthwise from the electrode.11 Therefore, with the 1.8 mm diameter electrode, lesions can be placed precisely within 0.5 mm of the optic tract. The electrode was then retracted 0.5 mm. A second lesion was generated at 69°C. This yields a lesion that extends 0.5 mm below the tip of the electrode. The electrode was then retracted another 0.5 mm, and lesions were made between 70° and 72°C. The electrode was retracted up to 6 mm, yielding lesions ~8 mm in length. The temperatures at more dorsal sites were based on the motor macrostimulation threshold. If this was low even at these more dorsolateral sites, then a lower lesion temperature was selected to prevent capsular insult. Macrostimulation of the optic tract generated visual perceptions of lights, sparkles, or sunsets, as described in the Methods section. The average visual macrostimulation threshold where the initial lesion was generated was 1.2 ± 0.5 V (n = 203) (see Table 5–1). See Figure 5–1 for a histogram of accepted thresholds based on the described equipment and methods. Our previous study has demonstrated a statistically significant decrease in the visual macrostimulation threshold as the electrode is advanced to the final target.2 The final target where the lesion was generated was changed in 139 of 203 (68%) cases from the initial image-based target an average of 0.9 ± 1.1 mm deeper along the electrode trajectory (range retracted 2.0 mm to advanced 4.0 mm). The majority of the time the electrode was advanced deeper (91%).
Macroelectrode Stimulation and Impedance Monitoring in the Posteroventral Pallidotomy
JONATHAN DENNIS CARLSON, ROBERT PAUL IACONO, EREK HELSETH, YOSHINORI HIGUCHI, AND GEORGE MAEDA
Methods
Patients
Surgical Methods
Equipment
Stimulation of the Optic Tract
Stimulation of the Internal Capsule
Impedance Measurement
Macroelectrode-Guided Posteroventral Pallidotomy Protocol
Lesion Generation
Results
Macrostimulation of the Optic Tract
| Average ± SD | Range | |
|---|---|---|
| 100 Hz visual macrostimulation threshold | 1.2 ± 0.5 V | 0.4 to 5.0 V |
| 2 Hz motor macrostimulation threshold | 2.7 ± 1.0 V | 0.8 to 6.0 V |
| Impedance at final target | 906 ± 142Ω | 470 to 1310Ω |
| Temperature of first lesion | 68 ± 2.1°C | 60 to 72°C |
| Maximum lesion temperature | 76 ± 2.9°C | 67 to 82°C |
A separate retrospective review of the intraoperative logbook was conducted of every consecutive PVP, beginning with the most current. This found that the visual macrostimulation threshold was detected and relied upon in 236 of 289 (82%) cases. The large majority of these patients without recorded data actually did have visual macrostimulation thresholds, which were simply not noted in the surgical log. Our subjective opinion is that in ~95% of the cases of alert awake and cognitively intact patients a visual macrostimulation threshold was detected and relied upon for adjusting the depth of the electrode. In those cases where no visual threshold was detected, a drop in impedance was relied upon to gauge the depth of the electrode as described in the Methods section. If neither of these were detected, the lesions were made more conservatively, with final verification of the electrode position based on AP and lateral x-rays coregistered precisely with a third ventriculogram.
Macrostimulation of the Internal Capsule
Macrostimulation of the corticobulbar fibers in the internal capsule elicited twitches of the tongue, as described in the Methods section. At the final target site, the motor threshold was 2.7 ± 1.0 V (n = 203) (see Fig. 5–2 and Table 5–1). In 10% of the cases (n = 21 of 203), the 2 Hz motor threshold was too low or too high at the final target of the first trajectory, indicating that the electrode was too close or too far from the corticobulbar fibers. In these cases, a new electrode trajectory was chosen between 1 to 3 mm away. In 16 patients there was a complete record of the stereotactic coordinates as well as motor threshold in both the first and second electrode tract. By plotting the changes in the motor threshold versus the changes in the AP coordinate, there was a relationship, indicating that as the electrode was moved posteriorly toward the internal capsule, the motor threshold decreased (r = 0.71, p < 0.05) (see Fig. 5–3).