Fig. 22.1
Schematic illustration showing the major co-morbidities associated with adult attention deficit hyperactivity disorder, including alcohol and drug dependence (From Kessler et al. (2006))
22.2 SPECT/PET Studies in Addiction
22.2.1 Studies in Animals
In general, animal models of psychiatric disorders simulate specific components of broad behavioural syndromes where clear evidence of neuropathology and altered brain metabolism is often lacking. However, modelling drug addiction is particularly challenging due to (1) the wide variability of clinical presentations, co-morbidities, and symptoms which make it difficult to discern whether there is only a single illness; (2) difficulties in controlling environmental variables that putatively affect disease expression; (3) difficulties in establishing genetic linkages; and (4) controlling for age of onset, gender and ethnicity effects, and poly-drug abuse. However, over the last decade, several promising animal models have been developed that capture key transitional stages of the addiction pathway, in particular, neurobehavioural vulnerability markers which carry high face validity and are not confounded by neural plasticity mechanisms associated with repeated cycles of drug bingeing and withdrawal.
However, a stark consequence of the historical underpinnings of addiction research is that the majority of molecular imaging studies have focused on markers of DA transmission in the fronto-striatal networks. Most, if not all, drugs with abuse potential increase DA release in the striatum, including the NAcb (Imperato and Di Chiara 1988a; Carboni et al. 1989), and can as a result displace radioligands selective for DA receptors. The benzamide [11C]-raclopride and the SPECT tracer [123I]-IBZM are commonly used for this purpose given their vulnerability to competition from endogenous DA (Innis et al. 1992; Dewey et al. 1993). Stimulant drugs such as methamphetamine, cocaine, methylphenidate, and to a lesser extent even nicotine decrease the binding of [11C]-raclopride and [18F]-fluoroclebopride in the striatum (Mach et al. 1993; Cumming et al. 2003), an effect also evident in mice treated with amphetamine and quantified with [18F]-fallypride–PET (Rominger et al. 2010). The close relationship between radioligand displacement and stimulant-evoked DA release is corroborated by animal studies employing in vivo microdialysis in conjunction with [123I]-IBZM–SPECT (Laruelle et al. 1998) and [11C]-raclopride–PET (Endres et al. 1997). This is also supported by an influential study of non-human primates showing that behavioural sensitisation (a putative marker of addiction) is linked to the enhanced displacement of [123]-IBZM by amphetamine (Castner et al. 2000). However, direct microdialysis measurements have not supported a sensitised DA response in rhesus monkeys exposed to high cumulative doses of cocaine (Bradberry 2011). The reason for this discrepancy is unclear but may relate to procedural and/or species differences.
Preclinical PET studies further demonstrate that D2/3 receptor availability is profoundly diminished in the striatum by chronic exposure to psychostimulant drugs, for example, as shown in both cynomolgus monkeys and rats treated with amphetamine (Ginovart et al. 1999; Dalley et al. 2009) and adolescent rats treated with methylphenidate (Thanos et al. 2009). This effect is remarkably consistent across species including humans (as shown in Fig. 22.2), and based on research in animals, this may result, in part, from chronic drug exposure (Nader et al. 2006; Groman et al. 2012). However, dysfunction of D2/3 receptors is also present prior to drug exposure in both experimental animals (Dalley et al. 2007) and humans (Buckholtz et al. 2010) which express high levels of impulsive behaviour. Thus, impaired D2/3 receptor signalling may underlie impulsive behaviour and be a susceptibility marker that is further compromised by chronic drug abuse.
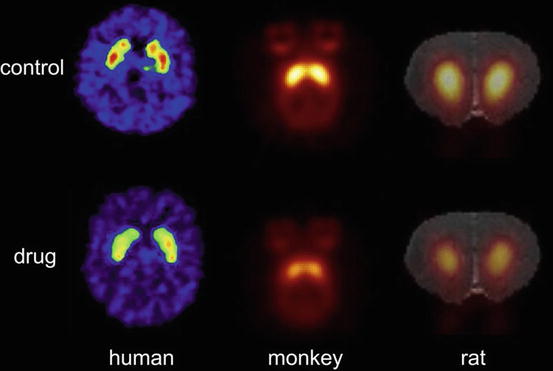

Fig. 22.2
Positron emission tomography scans showing reduced D2/3 receptor availability in the striatum of a recently abstinent cocaine addict (Volkow et al. 2002), a rhesus macaque monkey exposed to 3-month intravenous cocaine self-administration (Nader et al. 2006), and a Lister-hooded rat exposed to prior intravenous amphetamine self-administration (Dalley et al. 2009) (From Dalley et al. (2011). Reprinted with permission from Cell Press)
Consistent with this view, compelling evidence indicates that personality traits that encompass novelty/sensation seeking and impulsivity can predispose to drug use and have a detrimental impact, speeding the development of drug addiction (Chakroun et al. 2004; Koob and Le Moal 2005; Nigg et al. 2006; Belin et al. 2008; Verdejo-Garcia et al. 2008). However, there is a paucity of preclinical PET/SPECT studies that have explicitly investigated novelty-/sensation-seeking traits. In one study the behavioural response to novelty of Gottingen minipigs was investigated using [11C]-raclopride–PET (Lind et al. 2005). The core findings of this study show that the duration of contact with novel objects correlated with the amphetamine-induced decline in [11C]-raclopride binding potential. Thus, novelty-preferring animals evidently show enhanced DA release in the striatum just like the addiction-prone, novelty-reactive high-responder rat described by Piazza and colleagues (1989). Possibly consistent with these findings, it was recently shown in rats that biased decision–making under uncertainty was influenced by D 2/3 receptor expression in the striatum (Cocker et al. 2012).
A recent study in male cynomolgus monkeys showed that social dominance predicted low novelty reactivity, measured by the latency to touch a novel object, a trait associated with resilience to addiction (Czoty et al. 2010). In this study a significant positive correlation was observed between caudate D2/3 receptors, assessed using [18F]-fluoroclebopride–PET, and latencies to touch the novel object. This intriguing research resonates with findings in humans showing higher social status to predict higher baseline [11C]-raclopride binding in healthy volunteers (Martinez et al. 2010). It is also consistent with a now classic study showing the transition from single-housing to group-housing results in increased binding of [18F]-fluoroclebopride to striatal D2/3 receptors in the socially dominant cynomolgus monkey, the same subgroup which self-administered significantly less cocaine than their less dominant cage mates (Morgan et al. 2002).
Work over a number of years has shown that impulsivity in rats predicts intravenous cocaine and nicotine self-administration (Perry et al. 2005; Dalley et al. 2007; Diergaarde et al. 2008), as well as an increased propensity to develop compulsive cocaine self-administration (Belin et al. 2008), and to relapse after a period of abstinence (Economidou et al. 2009). In our laboratory, impulsive rats were selected from an outbred Lister-hooded strain, measured by their increased propensity to ‘jump the gun’ and respond before the presentation of a discriminative visual cue using a 5-choice serial reaction time task, a computerised operant test of sustained visual attention and impulsivity (Robbins 2002). The impulsive phenotype is present in a small but stable proportion of tested rats (8–14 %). In a previous microPET study, impulsive rats showed significantly reduced [18F]-fallypride uptake in the ventral striatum (including the NAcb), but not the caudate putamen (Dalley et al. 2007), in the absence of any prior exposure to stimulant drugs (see Fig. 22.3). Thus, low pre-existing D2/3 receptor availability in the ventral striatum may contribute to the risk for drug escalation and relapse to drug seeking. Based on a recent ex vivo autoradiography study, the locus of this deficiency in D2/3 receptors in high-impulsive rats appears to be the NAcb shell (Jupp et al. 2013), a region acknowledged to mediate primary drug reinforcement (Everitt and Robbins 2005).
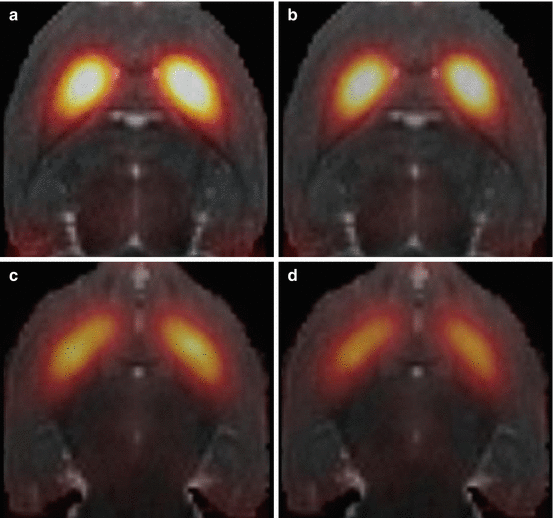

Fig. 22.3
Horizontal MR-co-registered PET scans showing reduced uptake of the high-affinity D2/3 receptor antagonist [18F]-fallypride in the ventral, but not dorsal, striatum of high-impulsive rats. The scans show, respectively, D2/3 receptor binding in the dorsal and ventral striatum of low (a, c)- and high (b, d)-impulsive rats (From Dalley et al. (2007). Reprinted with permission from AAAS)
22.2.2 Studies in Humans
In broad terms PET and SPECT studies in addiction have centred on (1) molecular correlates associated with the acute subjective experience of alcohol and other abused drugs and (2) residual and persistent markers of dysfunction in abstinent drug addicts. Subjects who self-report a ‘high’ following systemic administration of methylphenidate also show a significant displacement of [11C]-cocaine (Volkow et al. 1999) and [11C]-methylphenidate (Volkow et al. 2006) binding in the striatum, indicative of increased competition from endogenous DA. Analogous PET findings are reported for amphetamine-induced euphoria in normal healthy volunteers (Drevets et al. 2001) and for habitual smokers but only in those that reported a positive subjective experience (Montgomery et al. 2007). These PET studies thus substantiated the view that pharmacological treatments increasing DA release, especially in the NAcb, should evoke a positive hedonic experience.
However, this view belies the complexity of contributory mechanisms to the dopaminergic response of stimulant drugs measured by PET and SPECT. For example, there is convincing evidence to suggest that brain DA is not closely associated with the state of euphoria produced by abused substances (Leyton et al. 2007) but rather ‘drug wanting’ and the personality trait of novelty seeking–exploratory excitability (Leyton et al. 2002). Moreover, higher levels of trait impulsivity were predicted by diminished midbrain D 2/3 receptor binding and greater amphetamine–induced DA release in the striatum, which in turn was associated with stimulant craving (Buckholtz et al. 2010). In addition, high psychosocial stress levels predicted greater effects of amphetamine on striatal [11C]-raclopride binding (Wand et al. 2007), whereas trait impulsivity and a history of life stresses had complex effects on the effects of an amphetamine challenge in humans (Oswald et al. 2007). Furthermore, environmental cues in healthy subjects with previous exposure to amphetamine immediately prior to scanning were sufficient to reduce [11C]-raclopride binding (Boileau et al. 2007), similar to the effects of discrete visual cues conditioned with cocaine use (Wong et al. 2006). In addition, the subjective rating of pleasure produced by a bolus injection of methylphenidate in normal healthy volunteers depended inversely on baseline D2/3 receptor availability in the caudate putamen (Volkow et al. 2006). Elucidating the myriad of factors that predict individual differences in baseline and evoked changes in DA activity remains an important area of research in this field.
As the primary molecular target of cocaine and other stimulant drugs, the DA transporter (DAT) is a key biomarker for PET and SPECT research. Cocaine-abstinent addicts show increased DAT levels in the caudate putamen compared with healthy controls, measured by [99mTc]-TRODAT-1 SPECT (Crits-Christoph et al. 2008). This abnormality was found to be reversible in so far as [99mTc-TRODAT-1] uptake negatively correlated with the duration of time since the last exposure to cocaine. In an earlier study by Malison and colleagues, using [123I]-β-CIT SPECT, striatal DAT levels increased by approximately 20 % in cocaine addicts that were abstinent for less than 96 h (Malison et al. 1998). However, these findings conflict with studies of human methamphetamine (MA) abusers. For example, Volkow and colleagues investigated the density of DAT in abstinent MA abusers using PET and [11C]-d-threo-methylphenidate and observed lower levels in the caudate and putamen (Volkow et al. 2001a, b). Similar findings were reported in an earlier study using [11C]-WIN-35,428, following an abstinence period of more than 3 years on average (McCann et al. 1998) which upheld findings showing reduced DAT in the NAcb, anterior PFC, orbitofrontal cortex (OFC), dorsolateral PFC, and amygdala of abstinent MA abusers (Sekine et al. 2001; Sekine et al. 2003). In contrast, a longitudinal PET study using [11C]-d-threo-methylphenidate during both short (<6 months)- and long-term (12–17 months) periods of MA abstinence reported increases in DAT in the caudate and putamen (Volkow et al. 2001a). The extent of DAT recovery was associated with the severity of abuse and the duration of abstinence. Finally, the reinforcing effects of DAT blockade by cocaine have been assessed using [11C]-cocaine–PET (Volkow et al. 1997a, b). Here, it was shown that intravenous administration of cocaine at doses typically used by humans (i.e. 0.3–0.6 mg/kg) blocked 60–77 % of DAT binding sites. The magnitude of the self-reported ‘high’ positively correlated with the degree of DAT occupancy; at least 47 % DAT blockade was needed for subjects to perceive the euphoric effects of cocaine.
A striking consequence of withdrawal from many drugs of abuse in humans, including stimulant- and opiate-based substances, is a diminished supply of D2/3 receptors in the caudate and putamen (Wang et al. 1993; Volkow et al. 2001a, b). While questions are sometimes raised about the causal basis of this effect and its generalisability to generically unrelated drugs, it is a finding that is remarkably consistent in the preclinical literature (Nader and Czoty 2005; Dalley et al. 2009; Groman et al. 2012) and thus represents a putative biomarker of prior chronic drug exposure. Not only do drug addicts show a reduction in D2/3 receptors in the caudate and putamen; they also show a general blunting of DA release in these regions, measured by the attenuation of psychostimulant-induced changes in [11C]-raclopride binding (Volkow et al. 1997a, b; Martinez et al. 2009). Those addicted to cocaine also show a reduced ‘high’ relative to control subjects in response to intravenous methylphenidate (Volkow et al. 1997a, b). Collectively, these findings indicate that the brain reward system is rendered profoundly hypoactive in drug addicts, an outcome caused most parsimoniously by chronic exposure to addictive substances.
A fundamental research question is the extent to which abnormalities of D2/3 receptor binding in abstinent addicts are reversible and therefore recoverable after a protracted withdrawal period. Previous PET studies indicate that this abnormality can persist for many months following drug withdrawal (Sonsalla et al. 1989; Sharma et al. 2007; Yuan et al. 2010; Boileau et al. 2012). In a recent longitudinal study using [11C]-raclopride–PET, DA release was measured in the caudate putamen of MA abusers following a 60 mg oral dose of methylphenidate or placebo (Wang et al. 2012). The subjects were initially scanned within 6 months of the last MA exposure and again after 9 months. During early withdrawal MA abusers exhibited a lower availability of D2/3 receptors in the caudate compared with control subjects. As expected, following a methylphenidate challenge, there was a decrease in striatal (caudate, putamen, ventral striatum) D2/3 receptor availability in both MA abusers and controls. However, this decrease was attenuated in the left putamen of MA abusers, which may reflect a decrease in DA release in this region. Interestingly, the MA abusers with lower baseline D2/3 receptor levels than control subjects within the dorsal striatum (caudate and putamen) experienced no D2/3 receptor alterations following methylphenidate challenge and relapsed during the 9-month follow-up period. Correspondingly, MA abusers who experienced an increase in methylphenidate-evoked DA release, comparable with control subjects, did not relapse and successfully completed a 9-month abstinence period. The authors concluded that abstinent MA abusers with low striatal DA function have a greater likelihood for relapse (Wang et al. 2012). Similar conclusions have been drawn for cocaine dependence where blunted DA responsiveness appears to play a critical role in influencing choice preference for cocaine and thereby a putatively enhanced risk for relapse (Martinez et al. 2007).
As discussed above MA abuse is linked to reduced levels of D2/3 receptors and DA release (Volkow et al. 2001a, b), which supports the proposal by some that MA addiction could potentially be treated with drugs that increase DA transmission (Kish 2008). Not only do MA addicts show reduced D2/3 receptor densities, they also show higher levels of impulsivity than healthy control subjects, as determined using the Barratt Impulsiveness Scale (Lee et al. 2009). The underlying neural basis of increased impulsivity is only partly understood (see (Dalley et al. 2011)) but may be influenced by an upregulation of D3 receptors noted previously in abstinent stimulant addicts and rats (Mash 1997; Segal et al. 1997; Neisewander et al. 2004). Researchers have investigated the hypothesis that D3 receptor availability is elevated in MA abusers using [11C]-(+)-propyl-hexahydro-naphtho-oxazin–PET ([11C]-(+)-PHNO), a selective D3 ligand. The main findings of this research showed that ([ 11 C]-(+)-PHNO binding was increased in the substantia nigra compared with healthy control subjects but was decreased in the D 2 –rich caudate putamen and that ([ 11 C]-(+)-PHNO binding in the substantia nigra was related to self-reported ‘drug wanting’ (Boileau et al. 2012). The authors concluded that D3 receptors are upregulated in the brain of MA abusers. Thus, D3 receptor antagonism may be a viable strategy to reduce the risk of relapse by curbing impulsive behaviour. Although this was the first study to specifically examine D3 receptor density in MA addiction, all subjects were poly-drug users having reported the use of cocaine, MDMA, benzodiazepines, opiates, THC, and ketamine. Moreover, while [11C]-(+)-PHNO is a useful radioligand for the investigation of D3 receptors, it lacks absolute specificity; consequently the significance of this data must be interpreted with caution.
The quantification of regional glucose metabolism by PET has been widely utilised for the investigation of brain function in addiction. Volkow and colleagues undertook a [18F]-FDG–PET study of MA abusers and found a global increase in metabolism compared with control subjects (Volkow et al. 2001a). However, this outcome was unexpected, given past evidence of hypometabolism in abstinent cocaine addicts (Volkow et al. 1992). The authors hypothesised that the increased metabolism in MA abusers might reflect gliosis or inflammatory processes (Volkow et al. 2001a). In contrast, London and colleagues investigated metabolism in recently abstinent (4–7 days) MA abusers and reported a decrease in glucose metabolism in the anterior cingulate cortex and insula (London et al. 2004). However, they also reported that hypermetabolism in the lateral OFC, middle and posterior cingulate, amygdala, ventral striatum, and cerebellar vermis which they hypothesised might reflect a short abstinence period because these regions are also thought to play a role in drug craving (Everitt and Robbins 2005).
Using [18F]-FDG–PET, Berman and colleagues compared glucose metabolism in the brain of MA abusers after less than 1 week of abstinence and again after short-term periods (average of 3 months) in comparison to healthy control subjects (Berman et al. 2008). During the first month of abstinence, a critical period when relapse frequently occurs, there were no detectable metabolic changes in subcortical regions, but a widespread increase in cortical glucose metabolism combined with a marked increase was observed in the parietal cortex (Berman et al. 2008). Following their first year of abstinence, MA abusers continued to show metabolic deficits in the striatum while their thalamic metabolic response normalised (Wang et al. 2004). In summary, FDG–PET studies in MA addicts have yielded a complex array of findings with some discrepancies between different studies. These inconsistencies may be due to a number of factors such as differing routes of administration (e.g. nasal inhalation versus smoking or intravenous injection), the duration of abstinence, and also secondary exposure to other drugs of abuse.
The vesicular monoamine transporter (VMAT-2) redistributes monoamines such as DA from synaptic vesicles to the cytosol and is often targeted as a marker of DA neuron terminal integrity. Using [11C]-dihydrotetrabenazine (DTBZ)–PET Johanson and co-workers found that VMAT-2 density was decreased in abstinent MA abusers compared with control subjects (Johanson et al. 2006). Surprisingly, however, there was no correlation between VMAT-2 levels and the duration of abstinence. This might have been due to the continued use of other drugs during this period including alcohol, cocaine, opiates, and marijuana (Johanson et al. 2006; see Boileau et al. 2008).
The density of the 5-HT transporter (5-HTT) has also been investigated in abstinent stimulant abusers. Binding of the selective 5-HTT ligand [11C]-(+)-McN-5652 was reduced in the anterior cingulate cortex, OFC, dorsolateral PFC, temporal regions, thalamus, caudate, putamen, and the cerebellum of abstinent MA abusers (Sekine et al. 2006). In this study, decreased 5-HTT density in the OFC, anterior cingulate cortex, and temporal regions was associated with higher levels of aggression (Sekine et al. 2006). Diminished 5–HTT availability was also evident in abusers of 3,4–methylenedioxymethamphetamine (MDMA), the principal component of ecstasy, most notably in the cerebral cortex (Kish et al. 2010; Erritzoe et al. 2011; Urban et al. 2012), a perturbation that correlated inversely with the number of lifetime MDMA exposures (Erritzoe et al. 2011).
22.3 PET/SPECT Studies in ADHD
22.3.1 Studies in Animals
To date there have been very few neuroimaging studies in animal models of ADHD, and clues to the aetiology of this disorder have mainly come from PET and SPECT studies in humans. The efficacy of psychostimulants used to treat ADHD is thought to be mediated by blockade of DAT and NET (Fone and Nutt 2005), thus restoring a presumed underlying deficiency in catecholamine transmission. Our own work has focused on an innate form of impulsivity in rats associated with an increased propensity to anticipate the onset of a visual target cue using a 5-choice serial reaction time task (Dalley et al. 2007). Using [18F]-fallypride–microPET, we found that D2/3 receptor availability was significantly reduced in the ventral striatum (including the NAcb) but not the dorsal striatum. Interestingly, impulsive rats exposed to, and subsequently withdrawn from, intravenous cocaine self-administration exhibited a selective restoration of deficit D2/3 receptors in the ventral striatum, measured by [18F]-fallypride–microPET that was accompanied by a profound decrease in impulsivity (Caprioli et al. 2013). These findings tentatively suggest that stimulant drugs may exert their clinical effects in ADHD by modulating D2/3 receptor signalling in the striatum.
Other PET investigations in animals include studies of the neurofibromatosis-1 mutant (Nf+/−) mouse, which exhibits a reduced expression of the Nf1 gene (Brown et al. 2010). Children with the neurofibromatosis-1 (NF1)-inherited cancer syndrome develop benign and malignant tumours (Gutmann et al. 1997) and exhibit ADHD-like symptoms (Hyman et al. 2005). A recent [11C]-raclopride–microPET study of Nf1 mutant mice demonstrated that the non-selective attention deficit arises from presynaptic defects in striatal DA homeostasis (Brown et al. 2011). Pharmacological correction of the non-selective attention abnormality was achieved by administering methylphenidate or l-deprenyl, both of which increase striatal DA levels. This murine model may thus represent a useful preclinical platform for microPET imaging studies and the development of therapeutic strategies for NF1-related attention disorder.
22.3.2 Studies in Humans
As the primary target of methylphenidate and other stimulant drugs, DAT has been widely investigated in the context of ADHD (Fone and Nutt 2005). The first DAT neuroimaging study was conducted in a small group of adults with ADHD using [123I]-altropane–SPECT and showed that DAT levels in non-medicated patients were approximately 70 % higher than those in control subjects (Dougherty et al. 1999). However, subsequent research found far smaller increases with some even failing to reach significance (Dresel et al. 2000; van Dyck et al. 2002; Larisch et al. 2006). Dresel and colleagues investigated DAT binding in 17 treatment drug-naıve adults with ADHD and compared this with 10 age- and gender-matched control subjects, measured using 99mTc-TRODAT-1 SPECT (Dresel et al. 2000). Those with a diagnosis of ADHD exhibited a 17 % increase in DAT binding in the striatum compared with control subjects. DAT density was also compared in 9 treatment-naïve children with ADHD and 6 without ADHD using [123I]-IPT SPECT; the main findings showed a mean increase in DAT binding in the basal ganglia of ~45 % compared to control subjects (Cheon et al. 2003). Using [11C]-altropane–PET, Spencer and co-workers found that overall DAT binding was increased by 28 % in adults with ADHD relative to control subjects (Spencer et al. 2005). However, an earlier [123I]-β-CIT SPECT study failed to show a difference in striatal DAT density between adult patients with ADHD and control subjects (van Dyck et al. 2002). Furthermore, using [123I]-FP-CIT SPECT, Hesse et al. found that the striatal DAT binding ratio (specific to non-displaceable binding) was reduced in treatment-naive adults with ADHD (Hesse et al. 2009). These conflicting results may be due to the wide spectrum of symptoms present in those with ADHD as opposed to differences in imaging techniques or the differing profile of receptor specificity of different radioligands.
At clinically effective doses, methylphenidate occupies 50 % of the [11C]-cocaine binding sites in the caudate and putamen of healthy volunteers (Volkow et al. 1998) and substantially increases competition with DA at [11C]-raclopride binding sites (Volkow et al. 2002). It has also been shown that methylphenidate lowers DAT availability in both normal subjects and patients with ADHD. Using [123I]-FP-CIT SPECT, Vles et al. investigated the effects of methylphenidate treatment on DAT binding in 6 patients with ADHD (aged 6–10 years). Following 3-month treatment, striatal DAT binding was reduced by 28–76 % (Vles et al. 2003). A poor response to methylphenidate occurs in approximately 30 % of patients with ADHD, which may be caused by lower baseline DAT availability than others with ADHD. Krause et al. investigated the relationship between DAT availability and treatment outcome using [99mTc]-TRODAT-1 SPECT and demonstrated that patients who exhibited a poor response to methylphenidate had reduced DAT availability. In contrast, those with high DAT availability responded well to treatment with methylphenidate (Krause et al. 2005; la Fougere et al. 2006).
22.4 Conclusions and Future Perspectives
The research findings reviewed in this chapter demonstrate the extraordinary progress in the discovery of addiction-related biomarkers made possible by translational molecular neuroimaging. After two decades of clinical and basic research, a consensus has emerged that the transition to compulsive stimulant drug seeking must entail pre-existing individual neurobiological risk factors, modified and exacerbated by both drug exposure and contextual variables, including stress. The cascade of molecular mechanisms driving the shift from initial drug use to habitual and ultimately compulsive drug seeking are largely unknown but are, without doubt, critically influenced by predisposing neural and behavioural endophenotypes (e.g. low D2/3 receptor availability, impulsivity, novelty/sensation seeking) and by neuroplasticity mechanisms induced by repeated bouts of drug bingeing and withdrawal (Nader et al. 2008). The great majority of PET studies in this field have been motivated by the indisputable contributions of the brain DA systems to (1) reward (Wise and Hoffman 1992; Everitt and Robbins 2005) (2) the mediation of initial pharmacological effects of virtually all abused drugs (Imperato and Di Chiara 1988a, b), and (3) behavioural traits such as impulsivity and novelty/sensation seeking (Piazza et al. 1989; Dalley et al. 2007; Buckholtz et al. 2010). Additionally, co-morbid brain disorders such as ADHD, which critically appear to influence disease progression (Verdejo-Garcia et al. 2008), show remarkable overlaps with addiction with respect to brain DA dysfunction, as reviewed herein. An important question, therefore, is whether the treatment of juveniles with ADHD would curb or even prevent the future development of addiction in adults. A recent PET study in juvenile non-human primates found that early exposure to methylphenidate did not have an impact on the vulnerability to cocaine abuse in older animals nor did this compound influence the regulation of DAT and D 2/3 receptors in the striatum (Gill et al. 2012). This important study suggests that methylphenidate treatment per se is not a risk factor for addiction. However, further studies will be needed to investigate the effects of this, and related compounds, in animals pre–selected for trait–like impulsivity, especially as ADHD is accompanied by reduced D 2/3 receptor availability in the striatum (Volkow et al. 2009), just as in addiction (Sect. 22.2.2).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





