, Götz Penkert2 and Thomas Schelle3
(1)
Neurological Department, Freiberg Hospital, Freiberg, Germany
(2)
Neurosurgical Department, Friederiken-Hospital, Hannover, Germany
(3)
Neurological Department, Municipal Hospital Dessau, Dessau-Rosslau, Germany
8.1 Spinal Accessory Nerve
8.1.1 Anatomy
At the level of the skull base, the accessory nerve exits through the jugular foramen together with the internal jugular vein to which it is situated laterally. It leaves the jugular vein behind the digastric muscle and then runs on fibers of the levator scapulae downwards, where the nerve course is covered by the sternocleidomastoid muscle belly. At the lateral border of this muscle, three nerves arise quite superficially: the occipital minor nerve, the great auricular nerve (both nerves wrap around the lateral muscle border and run upwards), and the spinal accessory nerve which runs downwards and laterally into the posterior triangle of the neck. The exit point of these three nerves serves as an important landmark for finding the spinal accessory nerve surgically. In the whole posterior triangle, the nerve is situated subcutaneously, and it is therefore frequently in danger of being damaged during diagnostic extirpations of lymph nodes. Before entering the trapezius muscle it divides into several motor branches as functional supply.
It is very important to remember that parts of the trapezius muscle are additionally supplied by motor branches of the cervical plexus, as, e.g. described by Allieu et al. (1982) [1]. Anatomical descriptions even exist to the effect that cervical plexus motor branches can substitute the accessory nerve completely. We have occasionally observed patients suffering from impairment of physical activities and with visible atrophy of the superior part of the trapezoid muscle following surgery on persistent lateral neck cysts. They are located more cranially than lymph nodes, and surgery may easily hit this cervical plexus supply. We remember two cases with preserved continuity of the spinal accessory nerve, although with severe motor deficits following a neck cyst surgery. Intraoperative nerve action potentials, during exploration of these cases, surprisingly revealed normal function of the accessory nerve indicating isolated damage to the plexus branches.
Nerve connections between the 11th nerve and the 1st and 2nd cervical roots do additionally exist [2]. They carry afferent fibers, but they are usually not involved in the typical focal spinal accessory nerve lesions we have to deal with.
8.1.2 Damaging Factors
For the most part, spinal accessory nerve lesions are of iatrogenic origin, and only quite rarely are they due to a perforating injury [3]. As mentioned above, superficial lymph node biopsies in the posterior triangle are the main damaging cause [4, 5]. Real nerve entrapments of spontaneous onset do not exist. We nevertheless include this iatrogenic nerve injury in our compilation because of its focal character as is to be seen in Fig. 8.1.
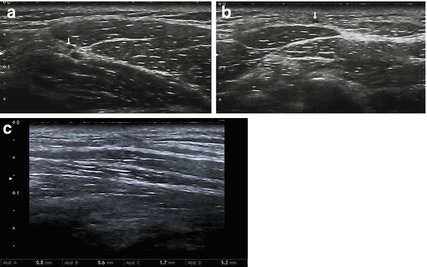

Fig. 8.1
Neuroma in continuity after lymph-node excision of the spinal accessory nerve. (a, b Cross-sections. c Longitudinal section). (a) Spinal accessory nerve (arrow) at the anterior border of the trapezius muscle. (b) Spinal accessory nerve (arrow) at the posterior triangle of the neck at a depth of about 3 mm beneath the skin surface. (c) Neuroma in continuity
8.1.3 Clinical Symptoms
The main complaints consist of pain in the shoulder region and of a lowered position of the affected shoulder [6]. Subsequently, the trapezoid deficit causes an asymmetrical aspect of both shoulders.
Why does pain occur despite the fact that the spinal accessory nerve exclusively carries motor fibers in only two-thirds of its lateral course? Overstretching forces to the brachial plexus have been discussed as etiology [6], but it seems more reasonable that pain is the result of overstretched fibrous ligaments or the result of overloaded muscles which have to compensate permanently.
The supraspinous and deltoid muscles abduct the humerus into a 90° position; then its further abduction requires rotation of the scapula [7]. This movement needs traction of fibers of the superior part of the trapezoid muscle and additionally of the levator scapulae and rhomboid muscles. The latter muscles have to compensate lost forces of the trapezoid fibers after a focal nerve lesion. The winging of the scapula is less distinct as in cases of serratus anterior muscle palsy following functional loss of the long thoracic nerve.
8.1.4 Electrodiagnostics
In order to differentiate major lesions, where a surgical repair is applicable, from minor lesions, electrodiagnostic workup of the spinal accessory nerve should include both nerve conduction studies and needle electromyography. Different techniques with updated normal values have recently been published in the literature [8, 9]. They allow examination of all segments of the trapezoid muscle (upper, middle and lower parts) separately. Using two different stimulation sites (posterior cervical angle at the midpoint of the posterior margin of the clavicular head of the sternocleidomastoid muscle for distal stimulation and the anterior-inferior angle of the mastoid process for proximal stimulation), a distal and a proximal motor latency. Additionally, a CMAP amplitude can be measured, moreover, the motor nerve conduction velocity can be calculated [9]. Thus, an early stage of lesion with only poor demyelization is already detectable. The motor nerve conduction velocity of the spinal accessory nerve in healthy individuals (upper segment of trapezoid muscle) is 65.7 ± 3.1 m/s and the CMAP amplitude 11.6 ± 3.5 mV [9]. Furthermore, as described in Chap. 5, needle electromyography helps to identify an axonal lesion by deriving evidence of denervation potentials in the trapezoid muscle. In the case of an additional absence of voluntary motor unit potentials or reinnervation potentials, a major lesion which probably needs surgical repair can be assumed [10].
8.1.5 Imaging
The superficial part of the course of the spinal accessory nerve is very easily accessible with high resolution ultrasound [11, 12]. The nerve runs across the posterior triangle of the neck with either a straight (56 %) or tortuous (44 %) course at a depth of about 3 mm beneath the skin surface and penetrates the anterior border of the trapezius muscle. This point can be used as landmark. A mean nerve caliber of 0.76 ± 0.12 mm is reported in the literature [11]. Especially in the case of iatrogenic nerve lesions in the cervical triangle, high resolution ultrasound can depict the neuroma formation in continuity (Fig. 8.1) or, alternatively, a nerve transection [13]. On the other hand, the cisternal and intraforaminal course of the spinal accessory nerve is, meanwhile, best visualized by means of high spatial resolution b-FFE gradient echo MRI [14].
8.1.6 Treatment
The extracranial course of the spinal accessory nerve is never irritated or entrapped by pre-existing anatomical structures but is focally damaged due to mechanical factors of a perforating character. Such injuries leave behind a small region of scar tissue. When exploring the nerve, when exploring the nerve the area should initially be avoided, exposure should be started at a more central level of the nerve course, which means at the lateral margin of the sternocleidomastoid muscle. The exposure of the wrapping occipital minor and auricular major nerves acts as a landmark and is extremely helpful. This principle is almost mandatory as, in the case of nerve discontinuity, the proximal nerve stump rather frequently disappears behind the muscle belly of the sternocleidomastoid [7]. In order to follow these principles, the original biopsy skin incision which corresponds to skin creases situated transversely has to be extended. Vertical limbs must be added to the primary small horizontal incision to allow nerve exploration within healthy tissue above and below the scar area. Unfortunately, such incisions are cosmetically undesirable [15]. Millesi therefore advocates several skin incisions situated parallel and with explicit regard to the skin creases,
Usually the nerve enters and exits an area of hard scar tissue. We then need a microscope and microsurgical instruments to answer the question of whether nerve continuity is evident or absent.
It may take about half an hour. The spinal accessory nerve is thin and of monofascicular structure at the level of the posterior triangle. In the case of persevered nerve continuity, the fibrosis involves the epineurium. This epineurium has to be longitudinally incised over the short distance of the lesion [7, 15]. Because of the small nerve caliber and its intraneural pattern, internal neurolysis steps are obsolete.
Cases with lost nerve continuity need nerve repair, very rarely achieved by end-to-end suture, mostly by one single auto-logous nerve graft. We have described the details of accessory nerve repair previously [15]. Although the focally damaged spinal accessory nerve lies superficially, just under the skin, exploration and repair remain a great challenge in peripheral nerve surgery for the following reasons: despite the little procedure that injured the nerve, we may even be confronted with almost complete nerve avulsions. The central avulsion then happens behind the sternocleidomastoid muscle. We are pleased when we find a small neuroma indicating the proximal nerve end. The same holds true in the periphery: the avulsion can hit the ramification of the nerve; distal nerve stumps do not develop neuromas. The identification of the distal nerve stump may therefore easily present more difficulties than the exposure of the proximal neuroma. Each surgeon now takes full responsibility for the primary surgical intervention. Secondary re-explorations lead to rather more difficulties and can fail completely due to extended scar tissue. As we have to expect a large field of varying pathologies – from short distance scar entrapment to long distance avulsion, spinal accessory nerve surgery should not be underestimated.
8.2 Brachial Plexus
8.2.1 Thoracic Outlet Syndrome (TOS)
8.2.1.1 Anatomy
The literature about anatomy, damaging factors, and treatment is so immense and differing in detail that we have to restrict our statements to personal experiences. Citations of literature will also be limited to examples which are representative of specific viewpoints.
We must define the neurogenic form of the syndrome as a nerve irritation syndrome. The five cervical nerve roots run together with the subclavian artery and vein between the scalene muscles laterally and downward. The two lower roots first rise a little through the upper thoracic outlet, and then unite to the inferior trunk which turns downwards. The interior margin of the first rib and sometimes, additionally, a cervical rib has thereby to be overcome. This anatomical situation is in itself completely normal because it is pre-existing in every human. An irritation syndrome of the inferior trunk can, however, start when anomalies or phylogenetically residual structures occur with fibrous ligaments within the scalene muscles, additional scalene muscles, fibrous ligaments between an elongated cervical transverse process and the internal margin of the first rib. Sometimes, if a cervical rib is present, such a fibrous band starts from its end, and runs obliquely through the thoracic outlet to insert somewhere at the first rib. Slim persons with a long and thin neck, predominantly women, are particularly prone to develop lower plexus irritation due to these structures. We should not forget that additional structures run transverse between the scalene muscles and fibrous bands: the subclavian artery and vein, and the transverse colli artery sometimes of large caliber. Pulsating forces within the arteries are quite able to press the comparably soft nerves against the fibrous ligaments. Due to arm abduction, daily activities, or unfavourable positioning overnight, the pre-existing narrowness in the thoracic outlet increases so that vascular and neurogenic symptoms will easily be induced or will deteriorate.
8.2.1.2 Damaging Factors
In the thoracic outlet we are not confronted with a particularly narrow tunnel as elsewhere in limbs, but exclusively with an unfavourable combination of life-long pre-existing structures which leave behind a narrowness. The onset of symptoms is, therefore, usually independent of any incident or injury. Typical damaging factors are often unknown. Instead, symptoms start spontaneously. Patients almost never have a reasonable explanation as to why and exactly when discomfort came into their life. Therefore patients and physicians take a long time to realise that a thoracic outlet syndrome might be the answer.
8.2.1.3 Clinical Symptoms
Patients’ complaints consist of electric-current-like sensations almost always irradiating into the ulnar aspect of the hand. Sometimes they observe these disturbing sensations during special arm positions or movements. In cases that have progressed further, patients sustain atrophy of the intrinsic hand muscles. Permanent numbness is not in the foreground, but it is discomfort by disturbing dysesthesias instead. As most of these patients with thoracic outlet syndrome don’t present any objectively noticeable deficits, physicians often suspect that patients are exaggerating their symptoms.
The onset of symptoms is usually unrelated to any trauma; they start spontaneously, and they increase chronically. Nevertheless, very rarely, the patient is able to remember a certain period of time when the symptoms actually emerged. Sometimes he even discusses a potential incident which he then suspects is responsible, like having carried heavy moving boxes on his shoulder in the past.
The typically nonspecific symptoms may be presented bilateral. On the other hand, the existence of a cervical rib on only one side does not really dispose which side is symptomatically affected. As fibrous ligaments are already mentioned as being responsible for symptom origin, the side and size of a cervical rib that has been examined in the patient’s X-ray do not determine which is the affected side of a plexus irritation. We even observed the opposite situations more frequently with a cervical rib on one side and symptoms due to fibrous bands on the other. Nevertheless, bilateral symptoms are possible with and without cervical ribs, with and without the enlargement of the seventh vertebra’s transverse processes, but always in cases of very slim persons.
From the neurological viewpoint it may be important that the Trömmner reflex runs through the inferior trunk, and that the Roos testing (paresthesias during finger motion with arms abducted, elbow joints 90° flexed, and hand palms to outside) turns out pathologically on the affected side. In our own experience, we particularly rely on the findings during supraclavicular palpation: this maneuver presses nerves against the disturbing ligamentous structure and immediately increases the amount of dysesthesias. We are convinced that this sign is a real aid in reaching the diagnosis. It indicates the location of a slightly degenerating and regenerating process like the Tinel sign. It even indicates the affected structure of the brachial plexus, namely, predominantly, the inferior trunk. By doing so, the patient observes dysesthesias exactly in the sensitive distribution area of the affected trunk. This area is similar to a dermatome, and completely differs from an autonomous peripheral nerve distribution area; for instance, numbness or dysesthesias of the ulnar nerve present an exact border in the middle of the fourth digit, whereas inferior trunk irritation also involves the third digit, sometimes with a border in the middle of the third digit. Of course, it should not be a matter of discussion that a cervical disc protrusion C6-7 has to be excluded before any thoracic outlet surgery is indicated.
8.2.1.4 Electrodiagnostics
As mentioned above, thoracic outlet syndrome is a controversial topic in the literature. Besides TOS resulting from vascular compression, a neurogenic form occurs. The neurogenic TOS can be further subdivided into a disputed and a true form. The majority of neurogenic TOS belongs to the disputed form (95 %), whereas the true neurogenic TOS is very rare and accounts for less than 1 % of cases [16]. The true neurogenic TOS is characterised by an affection of the inferior trunk of the brachial plexus together with objective diagnostic findings, whereas the disputed neurogenic form of TOS produces only subjective symptoms like chronic pain. In contrast to carpal tunnel syndrome and ulnar neuropathy at the elbow, unfortunately, there are no standardized guidelines regarding the EDX in the diagnosis of TOS at the moment. One reason is certainly its rarity. In this section we will focus on the EDX in the diagnosis of the neurogenic forms only. Regarding vascular TOS, please refer to the specialist literature. Taking into account the anatomy and pathophysiology, the main goal of EDX is to demonstrate a lesion of the inferior trunk, and, on the other hand, to exclude other pathologies which are similar to TOS, e.g. neuropathy of the ulnar nerve or affection of nerve roots C7 and C8. This easily succeeds in advanced stages of true neurogenic TOS with a pre-existing axonal lesion, but those cases where only a transient compression without permanent damage of neural structures occurs are rather difficult to detect (disputed neurogenic TOS). The registration of f-waves of the ulnar and median nerves in neutral position and after provocative maneuvers is unspecific, and a reduction of amplitudes of motor-evoked potentials after provocation was reported in two cases only [17, 18]. The intramuscular anterior scalene block may serve as a diagnostic tool for disputed neurogenic TOS; it temporarily blocks and paralyzes the muscle and allows the first rib to descend, thereby decompressing the thoracic outlet. Pain relief following this diagnostic intervention may serve as a positive predictor when surgery is considered [19]. Demyelinising nerve damage (true neurogenic TOS) instead increases the chances of obtaining electrically abnormal findings. A pathologically changed sensory nerve conduction study of the medial antebrachial cutaneous nerve (sensory latency over 2.4 ms, latency difference of 0.3 ms or more, amplitude under 10 μV, and amplitude ratios of 2.0 or more) is therefore suggested as the most reliable approach. Considering patients with true neurogenic TOS, 40 out of 41 had at least one of these four diagnostic criteria [20]. Furthermore, it can simultaneously exclude a supraganglionic root lesion and a neuropathy of the ulnar nerve. It has to be valued superior to the simple registration of sensory nerve action potential of the ulnar nerve from the fifth finger, which is pathological in the case of true neurogenic TOS as well as in the case of ulnar neuropathy [20]. Other tests have been described in the literature. Fractionated motor nerve conduction studies both of the ulnar and median nerves together with registration of the corresponding f-waves may be helpful. Reduction in segmental motor nerve conduction velocity to <85 m/s of either the ulnar or median nerves across the thoracic outlet should corroborate the clinical diagnosis [21]. A similar test is the registration of compound muscle action potential latencies of the abductor pollicis brevis and abductor digiti minimi muscles after magnetic stimulation of the brachial plexus. Compound muscle action potential latency of the ABP over 11.04 ms and of the ADM over 10.60 ms can indicate a true neurogenic TOS [22]. Another useful diagnostic tool is the registration of dermatomal and mixed nerve somatosensory evoked potentials (SEPs); however, the abnormality rate for both ulnar and C8 dermatomal SEPs was 100 %-significant only in a small group of advanced disease with severe muscular atrophy, whereas in earlier clinical stages, this rate dropped significantly to 50–67 % [23]. If primary demyelization is followed by secondary axonal loss, or if axonal damage primarily occurred, needle electromyography can easily demonstrate these changes by analysis of the distribution pattern of denervation in several muscles (see Sect. 5.6.2). In true neurogenic TOS the abnormalities occur in all muscles supplied by the root C8, and they do not occur in the paravertebral musculature of the cervical spine [24, 25]. This phenomenon helps to differentiate TOS from root C7 and C8 lesions due to cervical disc protrusion. In summary, needle electromyography together with registration of sensory nerve action potential of the medial antebrachial cutaneous nerve plays the key role in the diagnosis of true neurogenic TOS.
8.2.1.5 Imaging
High resolution ultrasound and 3 T-MR imaging provide an important diagnostic tool to detect pathological irritations of the brachial plexus and the cervical nerve roots, whereas EDX is nonspecific. High resolution ultrasound is a cost effective alternative to MR imaging outside the spinal cord [26]. Both high resolution ultrasound and MR are able to define the underlying pathology of brachial plexus involvement, e.g. of neoplasia, traumatic, or inflammatory origin, or due to congenital bone variations and fibromuscular anomalies [26, 27]. Using the transverse processes of the cervical vertebra as a landmark, it is easy to identify the cervical nerve roots with high resolution sonography. Taking into account that the transverse process of C7 has only a posterior tubercle, it becomes easier to determine the root level. In most individuals, this landmark will allow to start the examination of the whole course of nerve roots C4–C7 from the foramen to the interscalene space, and, furthermore, of its trunks above of the clavicle. In non-obese individuals, the root C8/TH1 and the brachial plexus below the clavicle are mostly visible [28]. Normal values of CSA (nerve roots C5–C8 and brachial plexus above of the clavicle) have been published only in a few studies to date [29–31]. At present, experience regarding differential diagnosis between true and disputed neurogenic TOS starts using high resolution ultrasound. Figures 8.2 and 8.3 illustrate imaging abilities. According to our experience in 6 cases, fibrous bands and cervical ribs always demonstrated the same appearance wedge/sickle-shaped with hyperechoic edge of the scalene muscles as well as with a hypoechoic and thickened inferior brachial plexus trunk. External compression with the probe led to an indentation of the subclavian artery (Fig. 8.3). MR imaging additionally provides the opportunity to demonstrate the T2 nerve lesion in the case of entrapment (see Chap. 6) and the administration of contrast agent. This may be helpful in differentiating between entrapment from other types of pathology such as neoplasm or inflammatory immune neuropathies and to localize the site of entrapment precisely – supraclavicular, infraclavicular, and subclavicular entrapment possibilities have at least to be considered [26, 27].
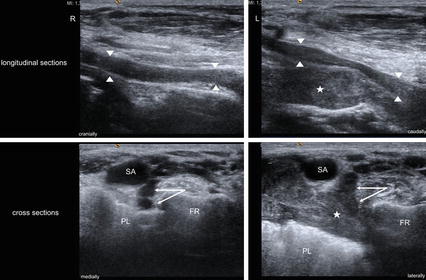
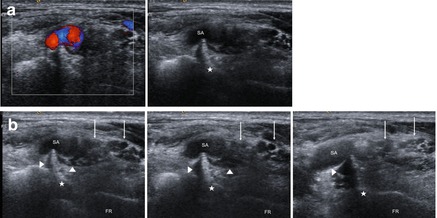

Fig. 8.2
Pancoast-tumor (asterisk) compressing nerve root C8/Th1 (arrows, arrowheads). Right: unaffected side, left: affected side (SA subclavian artery, PL pleura, FR first rib)

Fig. 8.3
Fibrous band at the medial edge of the medial scalene muscle (asterisk) compressing nerve root C8/Th1 (arrowheads) and subclavian artery (SA). (a) Compression of subclavian artery (colour-Doppler and greyscale images). (b) Image sequence (from cranial-left to caudal-right) showing the compression of nerve root C8/Th1 (arrowheads). FR first rib, arrows parts of brachial plexus supplied by roots C5-C7
8.2.1.6 Treatment
As already outlined in the introductory remarks of this book, all young patients want relief of pain symptoms. Besides new diagnostic means, we primarily continue to rely on the Tinel sign which can be elicited when the nerves are pressed by the physician’s fingertips against the irritating structure in the supraclavicular triangle.
The literature is full of advice on how to approach the proximal part of the brachial plexus in the case of mechanical irritation. The history of this development can be derived from Atasoy’s extensive presentation [32]. The thoracic outlet syndrome is defined as a focal irritation. Consequently, the approach has to remain as small as possible. Personal experiences over more than two decades regarding the disputed and true neurogenic cases are as follows. First rib resection as often advocated is not mandatory; this also holds true in cases with associated vascular symptoms which can even consist of artery embolism or venous incidents like pulmonary embolism in patient’s history. Inferior trunk damage following rib resection as sporadically observed means an intolerable complication; it has therefore to be avoided in all cases. Partial cervical rib resection was necessary in about 50 %. Complete scalenotomy is unnecessary because muscle fibers do not compress nerves. However, ligamentous fibers hidden between muscles have to be identified and transected completely. In a single case of middle trunk compression, an elongated transverse process of C6 was shortened. When the internal margin of the first rib is particularly sharp, it can be rounded off by means of punches in order to reduce its additional irritating role. The supraclavicular approach was always sufficient to achieve vascular and nerve decompression and thus relief of symptoms. However, there is one restriction to be stated: all cases we operated on in this period always had neurogenic symptoms and, infrequently, additional vascular ones. Pure vascular thoracic outlet syndromes were not included in our series because, in our region, they are included in the field of vascular and thorax surgery.
Theoretically, three different approaches of different extensions to the thoracic outlet region are available, and, of course, for several decades all of them have been considered to be equally successful: the superior approach through the lateral neck triangle [33–35], the transaxillary approach which needs first rib resection [36, 37], and the posterior subscapular approach which is restricted to recurrent cases [34, 38]. Few authors recommend a combination of these options [39]. The whole history of recommendations since 1861 can be followed up – as already mentioned – in the comprehensive contribution by Atasoy in 1996 [32].
The main objections to the transaxillary approach are possible damage of the inferior trunk and a rather hidden localized cervical rib if existent [40], to the supraclavicular approach damage of the phrenic nerve [40]. Probably due to the surgeon’s experience, these complications remain rare. Nevertheless, in reality, they are intolerable. Another intolerable complication is reported by Millesi et al. in 2007: sliding capacities of roots and trunks have to be preserved. The fat tissue which surrounds the nerves allows this sliding during extensive arm motion. This tissue has to be kept aside and never removed [41]. Secondary bleedings leave behind scar tissue and cause severe adhesions. A postoperative suction drainage of 2 or even 3 days is therefore mandatory in our experience. Intra-operative looping of roots and nerves irritates mechanically; it could cause the surgeon to induce damage on previously untouched and normally functioning nerves. We should remember that the myelin sheath of the nerve fiber is rather sensitive; its damage may end up as grade I lesion in accordance with Sunderland’s classification detailed in Sect. 3.1.
A small skin incision, 2–3 cm parallel to the clavicle, gives enough space. The skin in the neck region is extremely flexible and elastic so the actual approach site can easily be extended by two self-holding retractors. Of course, these retractors should not touch any nerve structure. Particular care has to be taken with the expected course of the phrenic nerve at the opening of the exposure. Adjacent, we find the above-mentioned package of loose fat tissue which we enter by using spreading movements of our scissors. We don’t cut through the tissue both in order to identify small venous vessels which have to be coagulated and individually transected and in order to avoid injury to the important structures lying underneath. By doing so, the omohyoid muscle is isolated within the fat package and can be looped and kept away. Above the muscle, by palpation with the finger tip, it will be helpful to feel and identify where superior and middle trunks run laterally and downward. A little more medial, the anterior scalene muscle can be exposed. By longitudinally spreading movements a careful opening of its surrounding membrane finally leads to the phrenic nerve. Spreading movements of scissor tips must always remain parallel to the expected structure. Instruments that keep nerves such as the phrenic nerve away must always to be placed in such a way that avoids harm or even kinking. During the surgery, all these instruments have to remain under permanent control. We use Langenbeck retractors and never loop nerves. The phrenic nerve acts as border: the whole exposure remains lateral to it because, if medial to it, the surgeon is in danger of damaging the internal jugular vein and the vagus nerve. The superior trunk – previously identified by fingertip – is now carefully isolated far enough, with the middle trunk next to it. At this point, both trunks can be slightly retracted upwards and lateral. Under them, palpation with fingertip will help, first, to find the irritating structure such as a fibrous band, and, second, to identify the subclavian artery which runs parallel to and under the clavicula, but sometimes also abnormally cranial convex curved. Before transecting or even removing any structure which you suppose to be responsible for the symptoms, a reliable identification of the inferior trunk and its division into the two centrally running roots C8 and T1 is mandatory. In most cases, the inferior trunk is flattened, and it rides across the band to lateral. Anatomical variations at root level, e.g. pre- and post-fixation of the brachial plexus, or a missing medial trunk are confusing [42, 43]. Prior to transecting the disturbing fibrous band, and sometimes, additionally, to opening a hypertrophic supra-pleural membrane you must have identified and isolated each structure. Along with this most important step, you can take a punch either to round off the internal margin of the first rib or to remove a cervical rib piece by piece, the latter however as extensively as possible. Rough bone edges left behind can cause new irritations. Before starting rib removal, its periost has to be carried off in order to skeletonise the bone. This procedure avoids injury to the parietal pleura and therefore reduces risks of pneumothorax decisively. Retracting instruments to keep nerves away again have to be under your permanent control. The suction in your left hand can aid as retractor in addition to instruments operated by your assistant.
As previously mentioned, a suction drain for 2 or even 3 days is necessary to exclude secondary bleeding and scarring. Before wound closure, the fat tissue is replaced into the supraclavicular groove. In addition, within this space are many small lymph vessels from which extravasations of lymph liquid will occur; this quickly stops when the wound sticks under suction (Fig. 8.4).
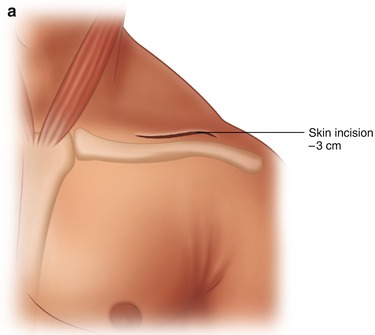
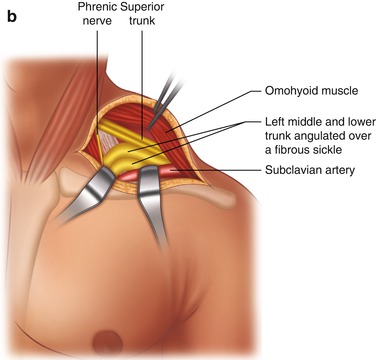
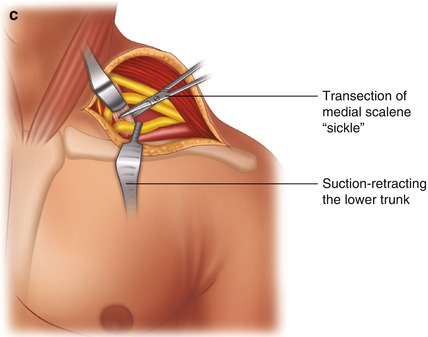



Fig. 8.4
Surgical treatment of TOS. (a) Supraclavicular skin incision. (b) Exposure of nerve trunks. (c) Transection of a disturbing fibrous “sickle”
We therefore prefer hospitalization of these patients for no less than 4 days. In only one case a pneumothorax complication and the need for thorax drainage extended hospitalization. We have had no cases of phrenic nerve lesion nor recurrences. In one of our recent patients, a female physiotherapist with a fuller figure, the elicitable pain disappeared at once; however two days later there was a sudden onset of a neuropathic pain in the neck and lateral triangle region. We are aware of criticism but these selected patients are in permanent contact with each other via the internet; thus problems come to light very quickly nowadays.
8.3 Costaclavicular Syndrome
We must assume that a compression of nerves and vessels between clavicula and first rib is a more or less historical idea. At first, it was described by Falconer and Weddell in 1943 [44]. They reported about soldiers who sustained symptoms in their arms after carrying heavy loads on their shoulders. The therapy they advised was first rib resection. Following Falconer’s first paper, literature presented a few further papers in 1949 and 1962 [45, 46].
However, our own experiences derived from conventional arteriography and modern MR-angiography demonstrated every time that a vessel stenosis during functional positioning of the arm occurred more proximal than the costaclavicular space is located. This phenomenon actually indicates that we always dealt with a classical thoracic outlet syndrome. Subsequently, our approach was the supraclavicular one without first rib resection as described in the thoracic outlet chapter. Falconer and Li described very comprehensively 50 years ago that they indeed preferred this approach and even succeeded in doing a first rib resection via the supraclavicular space; in their paper, the authors recommended to place the surgeon’s fingertips between clavicula and first rib and to depress the shoulder downwards and backwards as an important maneuver to consider first rib resection in selected cases [46]. However, we think that these considerations are more or less historical ones.
The only realistic focal brachial plexus neuropathy that can occur in the costaclavicular space in any case, is the one related to hypertrophic callus after clavicula fracture. The treatment then consists of the reduction of the callus only or, in the case of a pseudoarthrosis, of a secondary stabilization of the clavicula.
8.4 Hyperabduction Syndrome
Comparable to the costaclavicular syndrome, the question again rises as to whether the hyper-abduction syndrome really exists. We found the first description in the literature by Wright in 1945 [47]. Radiating dysesthesias were reported as depending on hyper-abducted arms during work and general daily activities.
We have experience with one such case. A few years ago, a young man presented repeated discomfort irradiating downwards into the sensitive area of the radial nerve, indicating an irritation of the posterior cord. A characteristic Tinel sign could be elicited in the infraclavicular groove as the only available guiding symptom to suggest surgery. At that time, functional neuroimaging was still not on the way.
The nerve irritation at cord level of the brachial plexus originates from the attachment of the pectoral minor muscle tendon at the coracoid process of the scapula. Therefore, surgery achieves relief by transecting this tendon. The skin incision can be angled at 90° to the clavicula and leads downward towards the axilla; the exposure should try to define the margins of the pectoral major and deltoid muscles with the cephalic vein in between. This vein indicates where you can easily enter into a sheath of fat tissue. Through this fatty package the pectoral minor muscle runs belly transverse to the course of the pectoral major muscle fibers. Of course, the pectoral minor muscle should then be isolated in its entire circumference. By doing so, you are able to identify the cord structures of the brachial plexus which lie underneath the pectoral minor muscle and parallel to the skin incision. You are still allowed to transect the tendon without any risk to the nerves.
This single surgery was done before qualified imaging experience as described in chapter 6 came up. The indication to operate on the patient was exclusively a clinical one.
8.5 Long Thoracic, Dorsal Scapular and Thoracodorsal Nerves
Mechanically caused focal lesions of these three nerves are rare and comparatively unknown. The nerves play a role in selected cases of trauma-related brachial plexus lesions as axon donor (the dorsal scapular nerve) or special target nerves. Iatrogenic lesions rarely occur after extensive axillary lymph node extirpation. Nerve repair then requires such a great deal of effort that reconstructive surgery is usually preferred.
In particular, when a patient’s history is characterised by sudden and severe pain with diffuse involvement of the shoulder region, then by pain disappearance, and next by motor deficits and quick atrophy with discomfort and inability to control and move the shoulder, the diagnosis of an inflammatory brachial plexus disease (Parsonage-Turner syndrome) has to be suggested as we describe in detail in Sect. 10.3. The onset of motor deficits is often outlined as occurring overnight. The symptoms may affect one of the three nerves separately or all three nerves together, and, additionally, the axillary (circumflex) nerve. MRI then reveals rapid fatty muscle substance degeneration and significant signal alterations of brachial plexus nerves. This important differential diagnosis must always be considered very seriously. Surgery should then be unconditionally avoided.
8.5.1 Clinical Symptoms
Independent of these diagnostic considerations, weakness of the serratus anterior muscle due to a long thoracic nerve lesion is considered a severe discomfort in patients’ eyes. Active arm abduction then stops at 90° because active forces to turn the inferior margin of the scapula outward to support further abduction have been lost.
8.5.2 Electrodiagnostics
As stated above, a lesion of long thoracic, dorsal scapular and thoracodorsal nerves is rather uncommon, therefore reports about EDX and imaging of the three nerves remain limited. Nevertheless, all three nerves can be examined using Erbs´s point for surface stimulation and by means of surface or needle recording electrodes placed over or in the muscles supplied by these nerves (long thoracic nerve – serratus anterior muscle, thoracodorsal nerve – latissimus dorsi muscle, dorsal scapular nerve – rhomboid muscles). Thereby, a distal motor latency and amplitude of the compound muscle action potential can be derived. Generally, the upper limit of normal decrease in amplitude from one side to the other is 50 % [48]. The normal values of distal motor latency depend on the recording technique and also on the distance between stimulation and recording electrode. For details please refer to Le Monaco et al. [48]. In addition, needle electromyography can demonstrate the axonal damage in the specific muscles mentioned above, and it can especially help to detect the comparably frequent inflammatory neuropathies which likely involve the three nerves – e.g. a Parsonage-Turner syndrome, or, alternatively, a C5 root lesion [49].
8.5.3 Imaging
Unfortunately, there are only a few reports about imaging of the three nerves. The long thoracic and dorsal scapular nerves were visible under ultrasound assessment within or superficial to the middle scalene muscle after division of the root C5 in 90 % of cases. The dorsal scapular nerve pierces the middle scalene muscle similar to the musculocutaneous nerve which pierces the coracobrachialis muscle. The nerves were located at depths similar to the perceived C6 nerve root, but a reliable distinction between the two nerves was only possible with the aid of electrostimulation [26, 50]. No knowledge exists of normal values of the CSA of the three nerves, nor of ultrasound imaging trials of the thoracodorsal nerve at the moment. Recently normal values of the anterior-posterior diameter of the long thoracic nerve (1.6 ± 0.3 mm) as well as examples of pathological involvement (e.g. Parsonage Turner Syndrome) have been described in a small retrospective study [51]. MR imaging may also become effective in recognising neuropathies of the shoulder girdle involving one or several of the three nerves [26].
Accordingly, trials of EDX and imaging on the long thoracic, dorsal scapular and thoracodorsal nerves will still continue at centers with specific research interests at the moment.
8.5.4 Treatment
The long thoracic nerve is the one and only nerve which is said to suffer from entrapment near the thoracic outlet region. The nerve arises at root level from C5 to C7, and runs posterior to the superior trunk downwards where it pierces the medial scalene muscle from outside to posterior. There are not previously published papers by Nath et al. in 2007 which report on 50 cases of long thoracic nerve decompression and neurolysis with post-operative significant improvement in 46 cases [52, 53]. The authors describe their usual approach as through the supraclavicular groove for exposure of roots and primary plexus trunks. They identify the long thoracic nerve posterior and lateral to the superior trunk. They add an external and internal microsurgical neurolysis. They mention that the nerve has several fascicles, and that it can also divide early into two or three branches at the location where the nerve traverses the middle scalene muscle. Its muscle fibers were finally transected as far as the situation required. This series seems to be the largest in literature. The rate of postoperative significant improvement of scapular winging within 1 day to 3 months is reported as 88 %. A few patients had a postoperative seroma similar to our experiences following thoracic outlet surgery, a relatively minor important detail which nevertheless emphasizes that suction drainage should be left for long enough. Similar experience with four cases of long thoracic nerve neurolysis was described by Disa et al. in 2001 [54]. The authors also stated that reconstructive surgery by means of muscle transfer would be the method of choice if recovery failed to appear or if nerve repair was being considered after nerve injury.
It is correct that Nath and his co-authors mention the Parsonage-Turner syndrome as one of considerable etiologies of functional loss of the long thoracic nerve (see also Sects. 6.1.8, 6.2.8 and 10.3). Half of their patients experienced pain; unfortunately, the paper does not reveal any detail of its onset, time course and localization. It should not be neglected that the long thoracic nerve mainly consists of efferent fibers. Nobody knows whether the few afferent fibers from muscle spindles are somehow able to generate pain sensations which the patient then feels, a question which is discussed when referring to the interosseus anterior nerve in the work of Birch and co-authors in 1998 [55]. The selection of appropriate cases to operate on after occurrence of severe scapular winging was determined again (in Nath’s 50 cases) according to patient history: particular attention was focussed on “injuries or any associated events” [52, 53]. It remains to be debated how to choose between inflammatory plexus neuritis and long thoracic nerve entrapment. Our personal impression was that our patient histories predominantly presented similarity to a plexus neuritis disease. It would be of immense help if surgeons could compare their series of operated and non-operated cases with each other in order to detect guiding clinical details which may help with the differential diagnosis. The development of modern imaging probably techniques will further contribute to the remaining diagnostic uncertainties. Findings of long thoracic nerve entrapments by imaging seem impossible because of the extremely thin nerve caliber and its hidden localization posterior to the superior trunk. Instead, indications of nerve entrapment can obviously be derived clinically, whereas differential-diagnostic evidence of plexus neuritis can be visualized by means of imaging.
8.6 Suprascapular Nerve
The suprascapular nerve can be involved in the field of focal neuropathies very frequently so we have to deal with it rather intensively.
8.6.1 Anatomy
The nerve arises from the superior trunk in the supraclavicular groove, turns to lateral and posterior, and runs through the incisura scapulae. The incisura has a ligamentous roof, the transverse superior scapular ligament. The nerve then has a kink of 60° to run downward where it divides into the supraspinous and infraspinous branches. The supraspinous branch turns again to lateral and supplies the supraspinous muscle which is situated in the supraspinous groove. Its tendon blends into the fibrous capsule of the shoulder joint which forms the rotator cuff. The infraspinous branch runs through the spinoglenoid notch into the infraspinous groove to supply the infraspinous muscle. Its tendon also blends into the fibrous rotator cuff. The course of the inferior branch has an additional roof between the superior and inferior fossa, the transverse inferior scapular ligament.
8.6.2 Damaging Factors
The main location of a focal nerve entrapment is the incisura scapulae with its transverse ligament as roof. Intensive sports, particularly volleyball or weightlifting, but also special exercises during ballet, can involve the nerve at the superior ligament where it kinks upwards during special maneuvers. Antoniadis et al. reported on their experiences with 28 cases in 1996; they outlined similar patients’ histories [56].
8.6.3 Clinical Symptoms
Most complaints consist of weakness during abduction in the shoulder joint. For the rest, further complaints are more varied, e.g. impairment during daily activities and sports in the affected shoulder region. Clinical findings then are weakness of initial upper arm abduction via the supraspinous muscle and weakness of a rotation outwards via the infraspinous muscle, in a position of a 90° flexed elbow joint. Behind the scapula we sometimes find a small area of numbness; very rarely the patient presents pain in this area. It should not be neglected that a separate infraspinous branch entrapment can occur under the ligamentous roofs in the spinoglenoid notch [57]. The resulting clinical finding is then presented as outward-rotation weakness only without pain or numbness. Never to be neglected is the important differential diagnosis to a partial rotator cuff tear which has similar clinical symptoms but presents with severe pain [58]; considerable pain that the physician induces by palpation of the rotator cuff area leads to the supposition of such degenerative periarthrosis symptoms, whereas if pain is not present during palpation, such a periarthrosis can be rather reliably excluded. MRI of the shoulder joint area is then definitively able to describe the conditions of the supra- and infraspinous tendons in the rotator cuff. Electromyography of the affected muscles with pathological results instead acts as an aid to diagnosing an alternative suprascapular nerve entrapment.
No less frequently, a so-called ganglion turns out to be the origin of nerve symptoms. Its location is predominantly the suprascapular notch, and there it sits, occupying space along with the irritating ligamentous roof of the incisura scapulae. Moreover, and rather rarely, such a ganglion can be situated within the spinoglenoid notch and can alone cause entrapment of the infraspinous branch [59]. Logically, imaging as a mandatory procedure leads to these findings and helps to decide about different treatment modalities.
8.6.4 Electrodiagnostics
In suprascapular neuropathy, the highest abnormality rate could be derived from nerve conduction studies (88 %), whereas needle electromyography showed pathological alterations in only 33 % of cases. Surface stimulation was always performed at Erb’s point, and compound muscle action potentials together with distal motor latencies were recorded from the supraspinous and infraspinous muscles. Therefore, in the majority of studies, concentric needle electrodes have been used [60]. The upper limit of distal motor latency to the supraspinous muscle was 2.71 ms, while data for the infraspinous muscle yielded an upper limit of distal motor latency of 3.42 ms in healthy individuals. Much more important than the distal motor latency prolongation are changes in the compound muscle action potential configuration in comparison to the healthy side [60]. The same test can also be performed with surface recording electrodes. The upper limit of distal motor latency is then 3.7 ms to the supraspinous and 4.2 ms to the infraspinous muscle, respectively [61]. In an advanced disease with axonal damage, needle electromyography shows denervation potentials either in both muscles, or only in the infraspinous muscle [62]. Furthermore, and again rather important, needle electromyography can exclude other pathologies, similar to suprascapular entrapment neuropathies, like a C5 lesion or an inflammatory affection of the upper brachial plexus (e.g. Parsonage-Turner syndrome; see Sects. 6.2.8 and 10.3) by means of examination of the muscles of the cervical spine or other muscles, e.g. the deltoid muscle, supplied by the root C5 but not by the suprascapular nerve. Abnormalities in needle electromyography and/or nerve conduction studies exclusively in the infraspinous muscle indicate entrapment neuropathy at the spinoglenoid notch, whereas the involvement of both muscles is compatible with entrapment neuropathy at the suprascapular notch [62].
8.6.5 Imaging
In order to apply peripheral nerve blocks the suprascapular nerve was depicted using high resolution ultrasound within the supraspinous fossa or at the suprascapular notch, however, at this site, the nerve is located deep under the trapezoid and supraspinous muscle bellies, and hence it is hardly visible there [63]. It can be visualized at the division of the nerve root C5, running posterior over the middle scalene muscle and then passing underneath the omohyoid muscle at a level slightly above the clavicle (Fig. 8.5). At this point diagnostic nerve blocks can be performed precisely [64]. However, this site is not the location of the classical entrapment. Its normal CSA ranges from 1.9–2.0 mm2 [65].
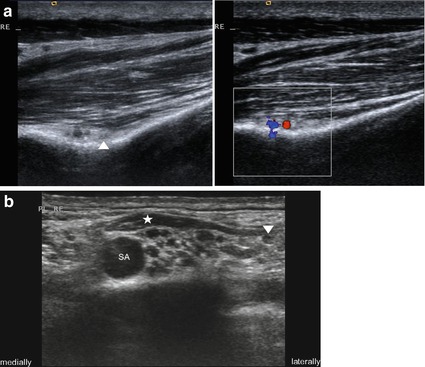

Fig. 8.5
Normal anatomy of the suprascapular nerve in HRUS. (a) Greyscale and colour-Doppler images of the suprascapular nerve (arrowhead) and the accompanying vessels at the level of the suprascapular notch below trapezoid and supraspinous muscle bellies in a slender individual. (b) Suprascapular nerve (arrowhead) at level of the supraclavicular brachial plexus below the omohyoid muscle (asterisk), SA subclavian artery
Therefore 3 T MRI should be preferred over high resolution ultrasound if a suprascapular nerve entrapment is suspected. MRI is especially able to demonstrate a T2 lesion at and above the site of entrapment (regarding spinoglenoid notch or suprascapular notch) and the fatty atrophy in the muscles supplied by the suprascapular nerve in the case of a chronic axonal loss. Moreover, MRI reveals special entrapment causes such as anomalous or thickened transverse scapular ligament, space-occupying lesions like a ganglion cyst or a soft tissue tumor (Fig. 8.6). It differentiates severe and painful traction injuries occurring with retraction of a large rotator cuff tear. In addition, mimicking conditions as mentioned in the EDX section in Chap. 5 can be excluded using MRI [62, 66, 67].
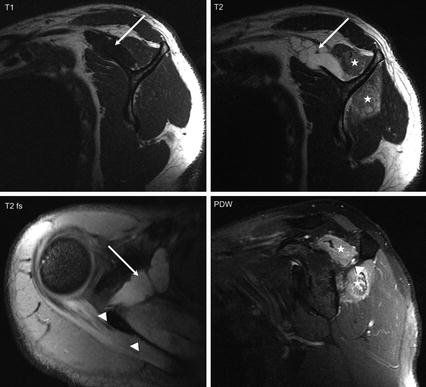

Fig. 8.6
MRI images of suprascapular nerve pathology caused by a ganglion-cyst (arrows) compressing the nerve at level of the suprascapular notch. Particularly on PDW images the nerve appears hyperintensive (arrowhead). Note also the denervation changes both of the supraspinous and infraspinous muscles (asterisk) compatible with a compression at the suprascapular notch (Images courtesy of Bergit Boy, MD and Marc Ewing, MD (department of radiology, Friederikenstift Hannover))
8.6.6 Treatment
The literature is full of recommendations on how to approach the suprascapular nerve and notch. Nerve entrapments are predominantly located under the superior transverse ligament, which the surgeon has to transect. The suprascapular artery is mostly lying upon the ligament whereas the nerve runs beneath. It may rarely happen that the supraspinous nerve branch runs together with the artery on the ligament. These details have to be clarified before ligament transection starts. Different approaches to the suprascapular notch are described in the literature: a lateral approach with the patient in a prone position [68], from above with skin incision transverse to the margin of the scapula and with respect to skin creases, additional detachment of trapezoid muscle fibers from the clavicula [69], transmuscular approach from behind with patient in supine position, fibers of the supraspinous muscle belly are split [56, 70], as with the next sub-periostal approach, patient again in supine position, supraspinous muscle belly kept upwards [71, 72]. We personally prefer the latter for two reasons: first, a transmuscular approach with splitting of the supraspinous muscle belly may damage microscopically small intramuscular branches of the supraspinous main nerve branch, and, second, early identification of the infraspinous branch and its course through the spinoglenoid notch is allowed. The frequent muscle belly atrophy facilitates the approach [73], and it provides enough space to follow the infraspinous branch to proximal in order to identify the supraspinous branch which runs more to lateral. The suprascapular notch usually lies more laterally than was at first thought. Early on we use the microscope after skin incision parallel to the spina scapulae, and after transection of the fascia near the spina scapulae, consequently at the moment when the supraspinous muscle fibers come into view, and when the subperiostal approach starts. The downwards running infraspinous nerve branch is the first nerve structure you can identify at the bottom of the supraspinous groove. It is embedded in fat tissue under the muscles fibers. Spreading movements of the scissor tips have to be as careful as possible so as not to injure the nerve. As a last step, transection of the superior transverse ligament is carried out.
Isolated infraspinous nerve branch entrapments have been reported [57, 74]. Incision, patient’s positioning and approach along the infraspinous surface of the spina scapulae comparable to the approach along the supraspinous surface should be suitable.
Pretty often, ganglia arising from the glenohumeral joint may be found to be similar to these manifestations near the tibiofibular joint. The pathogenesis of these ganglia remains controversial, but there is more and more evidence that occurrence of intra- and extraneural ganglia is connected with the articular nerve branch, here the branch to the glenohumeral joint. Therefore, the surgeon has to address and ligate this connection between joint and ganglion [74]. Nevertheless, a high rate of recurrence is known among surgeons at every location where the ganglia can occur. Our last three cases with multi-cystic ganglia in the supraspinous fossa were punctured CT (computed tomography)-guided. Re-puncturing, if necessary, is less risky than reoperation.
Not to be ignored is the rare possibility that ganglia may also be located within the spinoglenoid notch [74]. Imaging then has to decide between puncturing or surgical approach through the supraspinous or the infraspinous fossa. In any case, imaging is mandatory before suprascapular nerve entrapment surgery starts, namely, because of the relatively high frequency of space occupying ganglia in the region near the glenohumeral joint.
After open surgery, the fascia of the supraspinous or infraspinous grooves has to be reattached at the spina scapulae. The suction drain can be removed 1 day later.
8.7 Axillary (Circumflex) Nerve
The axillary (circumflex) nerve can rarely suffer from a focal entrapment within the so-called quadrilateral space. most axillary nerve lesions are instead related to trauma such as subcondyle humerus fractures or dislocations with luxation of the glenohumeral joint. The resulting severe functional loss needs to be examined after a few months and very frequently nerve repair by grafts.
Alternatively, frequent functional involvements happen in the context of the inflammatory Parsonage-Turner syndrome as described in detail in Sect. 10.3. Imaging modalities for assessing the differential diagnosis are discussed in Sects. 6.1.8 and 6.2.8. Initially, very severe shoulder girdle pain is noted, then weakness. Recovery takes several months.
8.7.1 Clinical Symptoms
The few entrapments that were observed as single cases and that were reported in the literature must be mentioned here [75, 76]. Over-trained muscles are described as origin. The quadrilateral space is formed medially by the triceps, laterally by the humerus bone, above by the teres minor muscle and below by the teres major muscle. Patients’ complaints consisted of pain and numbness in the distribution area of sensory axillary fibers at the lateral aspect of the shoulder. The symptoms could be provoked by lateral rotation of the shoulder or hyperabduction. With the aid of today’s modern imaging techniques, functional MR-angiography might delineate occlusion of the posterior circumflex humeral artery in abduction and lateral rotation of the shoulder joint.
8.7.2 Electrodiagnostics and Imaging
Lesions of the axillary nerve can be assessed by means of motor nerve conduction study using Erb’s point for surface stimulation. Placing the surface recording electrodes over the deltoid muscle, the upper limit of distal motor latency is 5.4 ms and the lower limit of compound muscle action potential is 4,6 mV with an upper limit of normal decrease in amplitude from one side to the other of 54 % [61]. Furthermore in the case of axonal loss, needle electromyography helps to differentiate from other conditions (upper brachial plexus lesion, C5 root lesion) due to analysis of the denervation pattern in different and neighbouring muscles. Additionally, myography can demonstrate whether reinnervation has started or not [49].
Ultrasound imaging of the axillary nerve can be performed via a posterior approach at the proximal part of the upper arm. Within the neurovascular space bordered by teres minor, deltoid, and triceps muscles as well as the shaft of the humerus, the nerve can be localized and successfully blocked with local anaesthesia beside the posterior circumflex artery (Fig. 8.7). However, it is rather difficult to visualize the small nerve directly, and hence there are no normal values of CSA available [77]. Recently a new sonographic approach has been described turning the upper-arm in a position of abduction and eversion. Traumatic lesions of the axillary nerve could be assessed using this method [78]. For that reason, 3 T-MR imaging seems to be the better approach to examine the whole course of the axillary nerve and its damaging factors [79].
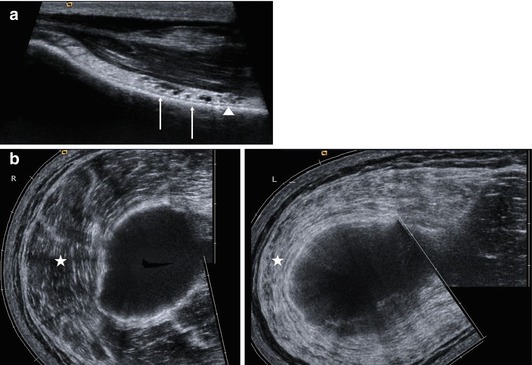

Fig. 8.7
(a) Ultrasound imaging of the axillary nerve performed via a posterior approach at the proximal part of the upper arm. Within the neurovascular space bordered by teres minor, deltoid, and triceps muscles as well as the shaft of the humerus, the axillary nerve (arrowhead) and the accompanying vessels (arrows) are clearly to be seen. (b) Denervation changes of the left deltoid muscle (asterisk) compared to the unaffected right side in a patient after motorcycle accident (panoramic view)
8.7.3 Treatment
The suitable surgical approach can best be derived from the atlas of peripheral nerve surgery by Kline et al. [80]. Nevertheless, the quadrilateral space is much more deeply located than thought when looking at illustrations of atlas specimens. Each patient reported in the literature was a sportsman with hypertrophic musculature. Experience with the approach from behind to the axillary nerve is described in our previous book, referring to trauma related nerve lesions where a bi-portal approach is always applied [81].
8.8 Musculocutaneous Nerve
With regard to the purpose of this book, a short overview will be sufficient because entrapment pathologies of the musculocutaneous nerve are unknown. Most of its fibers arise from root C6, and run within the lateral cord which divides at the level of the median fork. The musculocutaneous fibers turn to lateral and pierce the coracobrachialis muscle. Its fibers supply the biceps and brachialis muscles, and they contain sensory fibers for the lateral cutaneous nerve of the forearm.
The nerve has great importance in the case of brachial plexus reconstructive surgery as the target nerve of primary value. Injuries which hit the nerve in its upper arm course are either of a perforating character or happen in the context of proximal humerus fractures. Exploration and nerve repair is then almost ever indicated.
Difficulties can arise when the surgeon is confronted with the anatomical anomalies that were described in chap. 2.1 and which we found in several cases: the musculocutaneous fibers do sometimes not separate from the tateral cord proximal the shoulder joint level, but instead arise from the median nerve still at upper arm level. Similar observations are also described in literature [82]. We found these anomalies in search of the musculocutaneous nerve as the target in brachial plexus repairs.
The musculocutaneous nerve can quite frequently be severely be affected by inflammatory neuropathies such as the Parsonage-Turner syndrome as described in Sects. 6.2.8 and 10.3. Surgical decompression without some injury in the patient’s history is actually never indicated because, as mentioned, entrapment-related spontaneous deterioration of musculocutaneous function never occurs.
8.8.1 Electrodiagnostics
Lesions of the musculocutaneous nerve can be assessed with a motor nerve conduction study using Erb’s point for surface stimulation. Placing the surface recording electrodes over the biceps brachii muscle, the upper limit of distal motor latency is 5.6 ms and the lower limit of compound muscle action potential is 4.0 mV with an upper limit of normal decrease in amplitude from one side to the other of 33 % [61]. In addition, the terminal branch of the musculocutaneous nerve (lateral antebrachial cutaneous sensory nerve) can also be examined with a sensory nerve conduction study [83]. Furthermore, in the case of axonal loss, needle electromyography helps to differentiate from other conditions (upper brachial plexus lesion, C6 root lesion) due to analysis of the denervation pattern in different muscles. Additionally, electromyography can demonstrate whether a reinnervation takes place or not [49].
8.8.2 Imaging
The whole course of the musculocutaneous nerve can easily be visualized with high resolution ultrasound. At axilla level, the nerve enters the coracobrachialis muscle and runs within it (Fig. 8.8). After exiting the coracobrachialis the nerve lies between the brachialis and the biceps brachii muscles. Even its terminal branch (lateral antebrachial cutaneous sensory nerve) may possibly be visible, piercing the superficial fascia at elbow level in non-obese individuals (Fig. 8.9). The mean CSA of the musculocutaneous nerve was reported as 2.5 ± 0.4 mm2. Owing to its small size and out-of-plane course, the MCN may be more reliably depicted with ultrasound imaging than with MR imaging [84]. It should be mentioned that many anatomical variations of the musculocutaneous nerve were observed [85]. Ultrasound can show them before performing a nerve block or another intervention. Taking into account the scientific literature and our own experience with different traumatic lesions, a neuroma especially in continuity is easily detectable using sonography [84]. Moreover, we could observe a single case of external compression both of the median and musculocutaneous nerves at the axilla due to an arm prosthesis (Fig. 8.16). If ultrasound imaging is not available, 3 T-MR imaging may be a sufficient alternative, especially if administration of contrast agent is necessary [79].
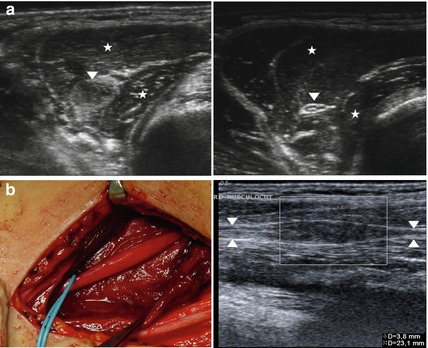
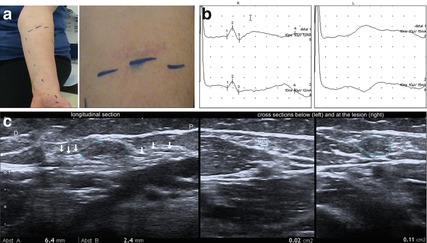

Fig. 8.8
(a) Cross sections of musculocutaneous nerve (arrowhead) piercing the coracobrachialis muscle (asterisk) at the axilla. Note the destroyed echotexture and nerve swelling as well as muscle denervation alterations after major lesion in a young man after motorcycle accident at the left side. In comparison to that at right side normal nerve structure preserved. (b) Corresponding intraoperative images and longitudinal sections of the pathologically altered nerve. Note the focal, fusiform, non-vascularised nerve enlargement, representing a neuroma in continuity (Intraoperative image courtesy of Ralph Schoen(MD), department of neurosurgery, Hospital Dessau-Rosslau)

Fig. 8.9
Major lesion of the left lateral antebrachial cutaneous sensory nerve after suicide attempt. (a) Clinical representation of numbness below the scar. (b) Nerve conduction studies revealed an absent sensory nerve action potential (SNAP) of the left lateral antebrachial cutaneous sensory nerve. (c) Ultrasound images demonstrating the neuroma in continuity and the normal appearance of the nerve below the lesion below the lesion (arrows: lateral antebrachial cutaneous nerve enters and exits the neuroma in continuity)
8.8.3 Treatment
Focal musculocutaneous entrapment lesions commonly do not occur, whereas traumatic lesions of this nerve appear rather frequently. But the treatment of nerve injuries is not a main purpose of this book, therefore we refer here to nerve trauma literature [80].
8.9 Radial Nerve at the Upper Arm
8.9.1 Anatomy
After separation of the axillary (circumflex) nerve from the posterior cord, remaining nerve fibers continue as radial nerve into the medial aspect of the upper arm. Behind the humerus, the nerve transverses through the spiral groove to the lateral side of the upper arm. A few centimeters distal it pierces the lateral intermuscular septum. Two or three proximal branches which supply the triceps muscle arise from the radial nerve below the armpit, whereas, after running transverse behind the humerus, the first motor branch runs to the brachio-radial muscle. Knowledge of these anatomical details helps to interpret clinical findings and to assess the location of the nerve lesion.
8.9.2 Damaging Factors
In terms of entrapment neuropathies, focal neuropathies at upper arm level occur rather rarely. The radial nerve may be easily compressed against the humerus bone within the spiral groove. Popular terms to describe this mechanism are “saturday night palsy” or “park bench palsy”. Be that as it may, an entrapment behind the humerus is always related to a special incident, and it never occurs spontaneously.
Quite another and extremely rare mechanism of nerve lesion can affect the radial nerve at the site where it pierces the lateral intermuscular septum. We have observed a case of spontaneous nerve rotation with hourglass-like aspect intraoperatively. Literature is full of single case reports of nerve rotation. Affected nerves were the radial nerve but also the posterior interosseus and anterior interosseus nerves [86, 87]. The single case of rotated radial nerve trunk we experienced had no special incident as possible explanation in its history [88]. Nevertheless, a spontaneous origin seems to be inconceivable. The latest scientific findings perhaps suggest a real context with hereditary forms of the Parsonage-Turner syndrome to which we refer in Sects. 6.2.8 and 10.3.
Trauma- or injury-related radial nerve lesions are commonly due to humerus fractures. Almost always the triceps muscle function remains preserved despite severe injury to the extremity. It will surely become of great interest if, particularly behind the humerus, imaging techniques and further improvements can distinguish between nerve continuity or discontinuity; furthermore, question of intraneural reactive scarring on one hand, or of the nerve gap distance after disruption on the other, will become an issue in future.
8.9.3 Clinical Symptoms
Loss of triceps muscle function indicates a highly proximal lesion, preservation of triceps function but loss of brachio-radial muscle function indicates a lesion between the two branching levels. Therefore, the clinical differentiation concerning the level of lesion should not be difficult. Electromyography supports the clinical assessment, and it can moreover differentiate between partial or complete functional loss, a question of great importance for deciding adequately whether surgical treatment is indicated.
8.9.4 Electrodiagnostics
Electrodiagnostic workup in radial neuropathy of the upper arm includes motor and sensory nerve conduction studies, needle electromyography and motor and sensory nerve conduction studies of the ulnar nerve and the median nerve in order to confirm the isolated neuropathy of the radial nerve and to exclude other kinds of neuropathies. A fractionated motor nerve conduction study with recording from the extensor indicis proprius (EIP), stimulating at forearm, at elbow, as well as below and above the spiral groove bilaterally, is recommended. With surface electrodes the normal CMAP recorded from the EIP is 2–5 mV. In the case of axonal loss 3–5 days after the lesion, a decreased distal CMAP results when comparing the involved side with the contralateral one. Measuring CMAP with surface electrodes often results in an initial positive deflection; therefore, this testing only allows approximations to solve the question of axonal loss. It presents additional difficulties when side-to-side comparison with neighboured radial muscles is tried, because the EIP is not a well-isolated muscle. Compared to the median and ulnar nerve, the radial nerve has a less straight course; this fact results in difficulties in the distance measurement. Together with the initial positive deflection of the CMAP, it can lead to considerable inaccuracies in measuring true conduction velocities. Therefore, the conduction velocity is not of real value concerning the radial motor function, but a focal conduction block between the proximal and distal sites and the determination of the relative CMAP amplitude are useful to assess eventual axonal loss [89].
In contrast to motor conduction evaluation, study of the sensory superficial radial nerve (SRN) is easy to perform. The most common technique is the antidromic excitation of the nerve 10 cm proximally on the distal radius while recording takes place distally from the first digit with ring electrodes. Comparing the SNAP on both sides is important. A decreased or absent SNAP suggests axonal damage at some level of the superficial radial nerve fibers. An interesting phenomenon in exclusively or predominantly proximal demyelination cases can be a normal SNAP although the patient reports marked numbness in the distribution of the superficial radial nerve [89]. Normal SNAP of the SRN may also occur in incomplete axonal proximal radial nerve lesions which spare fibers of the SNR on one hand [90], and particularly in the case of exclusive axonal loss in the posterior interosseus nerve (PIN) on the other.
Needle electromyography examination is required for a relatively accurate determination of lesion location and differential diagnosis. In PIN abnormalities the lesion will be limited to those muscles innervated by the posterior interosseus nerve (extensor indicis proprius, extensor digitorum communnis and extensor carpi ulnaris). In radial neuropathies at the spiral groove, brachioradialis and the long head of the extensor carpi radialis are involved in addition to PIN-innervated muscles. If the lesion is at the axilla, the triceps muscle is included in the lesion. A far proximal lesion of the posterior cord shows additional abnormalities which include the deltoid muscle (axillary nerve) and latissimus dorsi muscle (thoracodorsal nerve). In plexus brachialis lesions the cutaneous antebrachii lateralis nerve may also be involved. A C7/C8 radiculopathy will show abnormalities of the cervical paraspinal muscles and of non-radial-innervated C7/C8 muscles (pronator teres, flexor carpi radialis, interosseus dorsalis manus I) and a normal neurography of the sensory SRN.
8.9.5 Imaging
High resolution ultrasound can visualize the origin of the nerve injury, and it helps to locate the lesion by examining the entire nerve course from the axillary region down to the wrist. The nerve is easily detected in a transverse scan at the lateral aspect of the mid-humerus and can be followed from that point upward and downward [91]. Color Doppler sonography allows the visualization of the accompanying deep brachial artery, which serves as an anatomical landmark to identify the nerve. HRUS was meanwhile even proven to be useful for accurate evaluation of patients with nerve palsy associated with humeral shaft fracture [91]. Electrodiagnostic studies are instead less able to localize the site of proximal radial neuropathies [92]. Following a trauma, HRUS can assess primary lesions related to the trauma (e.g. a major lesion concerning Sunderland Grade V with discontinuity of the nerve after transection or severe traction) or secondary reactive alterations during the healing process of the humerus fracture (e.g. callus compression or nerve riding on a metal plate). Comparable cases are shown in Figs. 6.8b and 8.10b. In our experience, the typical compression syndrome among drinkers (“Saturday night nerve palsy”) causes either no changes detectable by high resolution ultrasound or a segment of the radial nerve at the site of compression with normal nerve caliber proximally and distally to it (Fig. 8.10a), whereas we never detected a focal entrapment at upper arm level with spontaneous onset so far. However, important is the fact that, in some non-traumatic and non-entrapment radial nerve palsies, HRUS can demonstrate either unspecific long-distance thickening of nerve fascicles and caliber changes (e.g. as a result of brachial plexus neuritis or diabetic mono-neuritis multiplex) or specific morphological changes such as peripheral nerve sheath tumors, tumor like lesions, or even a torsion or rotation neuropathy (see Chap. 12). Figure 8.10b illustrates nerve damage due to trauma, Fig. 8.10c a malign nerve sheath tumor, and Fig. 8.11 a nerve torsion. We experienced the curious radial nerve torsion neuropathy as characterized by an hourglass-like stenosis with congestion of the nerve more proximally from it. Similar changes have been described by other authors [93].
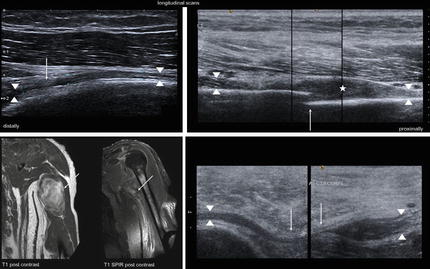
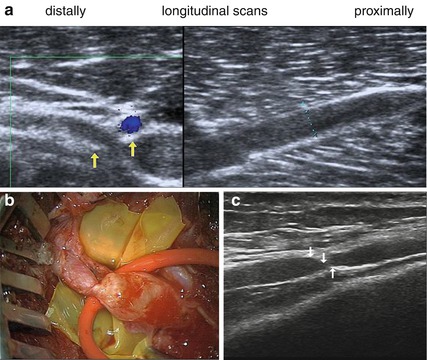

Fig. 8.10
Several lesions of the radial nerve at upper arm. (a) Saturday night palsy in HRUS. Longitudinal scan along the radial nerve (arrowhead) at the spiral groove shows spindle-like swelling of the radial nerve (caliber 2 mm, proximal of it only 1.2 mm) above the site of compression (arrow). (b) HRUS, fracture of the humerus (arrow) with a bone fragment piercing the radial nerve (arrowhead) with secondary development of a neuroma in continuity (asterisk). (c) MRI, Malign peripheral nerve sheath tumor of the radial nerve in a patient with Neurofibromatosis type I before and after resection (left). HRUS; scarring after excision-induced nerve entrapment (arrows) of the radial nerve (arrowheads) (MR images courtesy of Karsten Stock, MD (department of radiology, Dessau-Rosslau))

Fig. 8.11
HRUS, multiple torsions of the radial nerve. (a) Substantial caliber change and hourglass-like constriction (arrows) with deep brachial artery just above the constriction as well as long-segmental enlargement of nerve diameter proximal to the constriction (3.6 mm). (b) Corresponding intraoperative image. Only after removal of adventitia the twisted constriction and proximal bulging of the nerve is clearly visible (Image courtesy of Thomas-Nicolas Lehmann, MD (department of neurosurgery, Hospital-Bad Saarow/Germany)). (c) A second distally located spontaneous torsion of the radial nerve in the same patient month after surgical repair of the torsion depicted in (a) and (b) (arrows: site of nerve torsion)
3 T-MR neurography is also suitable for the diagnosis of radial neuropathies. Especially in early stages of the lesion it already shows a T2 lesion of fascicles [94, 95]. The advantage is that this type of imaging makes it possible to administer contrast agent if a tumor is suspected. However, the majority of cases do not need such expansive means.
8.9.6 Treatment
Spontaneous radial nerve trunk entrapments behind the humerus are not to be expected as just mentioned. In our experience, surgical exposure following a supposed nerve compression was never indicated. The same holds true in the literature.
The very rare nerve rotation cases have hardly an incident in patient history, and in contrast to missing causative factors, a complete functional loss mostly from the brachio-radial muscle downwards. The symptoms occur suddenly, and nerve exploration, e.g. via a longitudinal incision at the lateral aspect of the lower half of the upper arm, is unavoidable. To facilitate the exposure, the surgeon can start at the level of the elbow skin crease. A direct approach between the brachio-radial and biceps muscle bellies and the biceps tendon is suitable to identify the radial nerve most easily. From that point, the exposure has to progress in a proximal direction. An hourglass-like nerve aspect, if it really has been found, needs a microneurolysis under microscope and mostly a nerve repair by grafts as demonstrated in Chap. 12 and [88]. It should be noted that graft repair of the radial nerve carries the best prognosis of all extremity nerves.
8.10 Posterior Interosseus Nerve
8.10.1 Anatomy
Several aspects of anatomy are of great clinical importance and must be known: the radial nerve runs downwards between the biceps tendon and the brachioradial muscle, the superficial radial nerve branch separates at different levels, the posterior interosseus branch enters the supinator channel under a tendon-like arcade which is referred to as the arcade of Frohse. This is the most common site of focal nerve entrapment. Different names, depending of the site of compression, are common: “PIN syndrome” or “supinator syndrome” [96]. The entrapment results in pure motor deficits. It is characterized by loss of ability to extend the digits as well as radial wrist deviation due to weakness of the extensor carpi ulnaris but preserved wrist extension because the short radial wrist extensor remains supplied. This muscle mostly gets a small branch that separates from the superficial nerve branch or from the main radial trunk. A very small sensory branch runs downwards supplying a small skin area between the first and second digit on the extension side.
“Radial tunnel syndrome” is sometimes said to be a distinct clinical entity without motor weakness and occurring as a result of repetitive motion injuries to the elbow [97]. The term “algetic supinator tunnel syndrome” is unfortunately confusing and should not be used [98]. It is associated with a tendinosis such as epicondylitis humeri radialis.
8.10.2 Damaging Factors
Focal compression sites from proximal to distal include fibrous bands from the radio-capitellar joint, the tendinous edge of the extensor carpi radialis brevis muscle adjacent to the radial recurrent artery and its branches (leash of Henry), the arcade of Frohse (tendinous proximal end of supinator), and a fibrous band at the distal end of supinator muscle [96, 99]. Entrapment neuropathies of the PIN occur frequently. The patient normally does not remember any incident and not even the time of weakness onset. Sometimes a lipoma compresses the nerve branch against the arcade of Frohse. It remains controversial as to why such lipomas emerge exactly under the supinator muscle. They are accidentally found during surgery, or they are evaluated by HRUS (high resolution ultrasound) or MRI (Multi-Resonance-Imaging) when done pre-operatively. PIN palsy can be a very rare complication of rheumatoid arthritis affecting the elbow joint. Eighteen cases of PIN palsy due to rheumatoid arthritis have been reported so far in the literature [100]. Trauma related PIN palsies result from radius fractures or its osteosynthesis. The functional loss is then almost ever complete.
As a differential-diagnostic problem with the PIN syndrome, with slowly proceeding palsy and without numbness, the rare multifocal motorical neuropathy (MMN) has to be kept in mind. It can quite easily simulate an entrapped motor nerve as described in detail in Chap. 10. PIN torsion (rotation) as a spontaneously occurring incident has been sporadically reported in literature [86, 101]. We observed one case with multi-segmental torsions of the radial nerve, including one incident in the supinator tunnel. Not to be ignored is the fact that severe PIN palsies can generally be the single manifestation of a Parsonage-Turner syndrome, sometimes even with nerve torsion (see Sects. 6.2.8 and 10.3; Chap. 12). We have experienced in two such patients initial and heavy shoulder pain as important anamnesis factor and, following these attacks, a relatively sudden weakness.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







