CHAPTER 12 Neuro-ophthalmology of Meningiomas
INTRODUCTION
Neuro-ophthalmology is a complex, broad-ranging area of ophthalmology and one of the most important fields that can help in diagnosis and follow-up in neuro-oncology. Recognizing subtle physical signs, such as pupil abnormalities, cranial nerve palsies, or visual field defects may be the key to finding significant intracranial meningioma pathology. Correct diagnosis and referral to neuroimaging all depend on a thorough understanding of neuroanatomy and neuro-ophthalmologic examination. Symptoms may be expressed simply as loss of vision or double vision, or as complex syndromes depending on the location of lesion. A firm understanding of the neuroanatomy and neurophysiology of the eye is essential for correct diagnosis. Neuro-ophthalmologic findings of meningiomas depend on the tumor size and its location along the neuro-ophthalmologic tract. Meningiomas arise from arachnoidal cells, and they may be attached to any of the three layers of the meninges. Problems may occur anywhere along the visual pathway, including the brain stem, cavernous sinus, cerebral cortex, subarachnoid space, and orbital apex, and may affect adjacent structures also. Meningiomas with primary ophthalmic importance are located mainly in the cavernous sinus, tuberculum sella, optic nerve sheath, orbita, sphenoid wing and olfactory groove.1 Other meningiomas located in convexity, falx and parasagittal area, middle cranial fossa, cerebellopontine angle, clivus, foramen magnum, tentorium cerebelli, and ventricles usually present with seizures or focal neurologic signs before they produce neuro-ophthalmologic signs.2
CAVERNOUS SINUS MENINGIOMAS
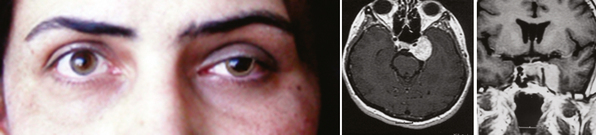
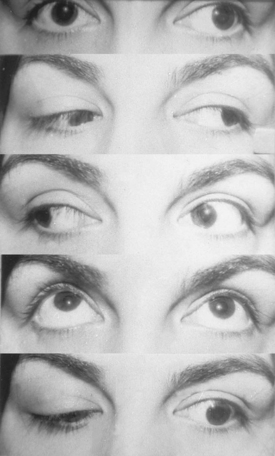
The clinical picture of cavernous sinus meningiomas (CSMs) can arise from compression or damage to one or more of the ocular motor nerves and vessels.3 Patients with cavernous sinus tumors most frequently present with ocular motor deficits: ptosis, diplopia, anisocoria, or complete ophthalmoplegia. The most common symptom is diplopia, resulting from paresis of the ocular motor nerves. Damage to the oculomotor nerve within the cavernous sinus may produce a partial or complete oculomotor nerve palsy. The pupil is usually involved in complete third nerve palsy, and is mydriatic and not reactive to light or accommodation (Fig. 12-1). In partial third nerve palsy, the pupil may be normal and initially may be mistaken for the ophthalmoparesis produced by myasthenia gravis.4 If the pupil is smaller than normal, damage to the oculosympathetic pathway should be considered. Slowly progressive compressive lesions such as meningiomas and aneurysms may cause primary aberrant regeneration between the fibers of the oculomotor nerve.5 The features of aberrant regeneration may be one or a combination of the following: adduction on attempted upgaze, constriction of pupil on lateral gaze and elevation of the upper lid on down gaze (Fig. 12-2).
Sixth nerve palsy is rarely isolated. Undiagnosed chronic isolated sixth nerve paresis or remitting sixth nerve palsy associated with CSM were described mostly before modern imaging techniques were available.6–8 The oculosympathetic pathway runs for a short distance with the abducens nerve in the cavernous sinus; therefore ipsilateral postganglionic Horner syndrome may be associated with unilateral abducens nerve paresis.1 Some patients may complain of dysesthesia and pain in and around the eye, the orbit, or the upper face if the trigeminal nerve is involved. Intermittent exotropia and neuroparalytic keratopathy has been reported as a rare presenting signs of CSM.9,10 Ocular neuromyotonia is an intermittent ocular deviation due to spasm of one or more eye muscles innervated by the same nerve and usually involves the oculomotor nerve. Typically, spasm is triggered by sustained extremes of gaze. Although the radiotherapy remains the most frequent cause, it has been described in patients with CSM who has not received radiotherapy.11,12
The patient may have visual loss and visual field deficits if the optic nerve is damaged by growth of the tumor into the orbit through the superior orbital fissure or by extension of the tumor to involve the intracranial or intracanalicular portions of the nerve. Gaze-evoked amaurosis is a transient monocular loss of vision occurring in a particular direction of eccentric gaze associated with orbital tumors, optic nerve sheath meningioma, and CSM.13 Possible mechanisms for gaze evoked amaurosis include inhibition of axonal impulses or transient optic nerve ischemia.
The decision regarding treatment is often difficult because of poor accessibility and the frequent involvement of the cavernous carotid artery. Cranial nerve morbidity is a critical issue on the resection of cavernous sinus meningiomas. Extraocular muscle functions may be impaired because of cranial nerve III, IV, and VI injuries. The reported incidences of permanently impaired extraocular muscle function developing after resection of cavernous sinus meningioma is 14% to 58%.14–18 Initially, with the advent of skull base approaches, the authors thought that these tumors could be removed with an acceptable cranial nerve morbidity. However, surgical treatment of these tumors with radical excision using all the modern skull-base surgery techniques is usually an unnecessary effort that leads to complications and decreases the quality of life, which could have been avoided with more conservative strategies. Extraocular muscle impairment is so disturbing that it becomes the main determining factor for the quality of life in the postoperative period. With time, authors favored more conservative strategies with cytoreduction of extracavernous tumor to protect brain function and to contain the tumor within the cavernous sinus to preserve cranial nerve function.19–21 Repair of the third through sixth cranial nerves injured during surgery can be pursued in suitable patients.22
Gamma-knife radiosurgery was found to be an effective low morbidity–related tool for the treatment of cavernous sinus meningioma.23 Although it cannot eradicate the neoplasm, it makes a favorable change in its natural history and helps clinical healing or stabilization of the symptoms. From a clinical perspective, the authors indicated stable or improved neurologic status in more than 90% of cases.24–27 Postoperative cranial nerve palsies are extremely rare (0% to 1% post-radiosurgery cranial nerve palsy).25,26 However, radiation sensitivity of the optic apparatus is one of the radiosurgery-limiting issues in the management of cavernous sinus meningiomas. Duma and colleagues28 reported 2 of 34 patients with radiation-induced neuropathy after radiosurgery. Removing the extracavernous part near the optic nerves also may eliminate this problem; a distance may be maintained between the radiated tumor margin and the closest optical anatomic structure to preserve optic nerve function.
The prognosis for vision depends on the extent of compression on optic nerves. Removal of compressing part of tumor may help; however, ischemia to the optic nerve is also an issue in this regard. Delayed ischemic optic neuropathy in two cases after surgery on skull-base meningiomas have been reported and successfully treated with nimodipine and rheologic therapy.29 In addition, some patients may experience spontaneous regression of symptoms and signs.30,31
OLFACTORY GROOVE MENINGIOMAS
Olfactory groove meningiomas (OGMs) arise from the midline of the anterior fossa between the crista galli and the tuberculum sella. Although these tumors arise in the midline, they may extend predominantly to one side. They are usually bilateral but may be asymmetric and reach a large size before causing symptoms.32 They can be differentiated from tuberculum sella meningiomas because OGMs arise more anterior in the skull base and displace the optic nerve and chiasm inferiorly rather than superiorly.33 An intracranial mass is often not suspected until the patient develops symptoms and signs of increased intracranial pressure (ICP), slowly progressive visual loss from pressure on the intracranial optic nerves or chiasm, or evidence of frontal lobe compression. In some cases, OGMs grow between the optic nerves, forcing them against the internal carotid and anterior cerebral arteries. Compression of the temporal portions of the optic nerves by these vessels produces bilateral nasal field defects.34 Less commonly, acute visual loss may occur when hemorrhage develops.1 Optic atrophy is a frequent finding, which may be unilateral or bilateral and is often asymmetric. Optic atrophy associated with contralateral papilledema and anosmia is a well known triad of Foster Kennedy syndrome which is caused by optic nerve compression and increased ICP due to OGM.35 Visual loss can be bilateral with unilateral optic disc swelling. Sequential anterior ischemic optic neuropathy can produce a similar picture with acute optic disc swelling in one eye and optic atrophy on the contralateral side, called pseudo-Foster Kennedy syndrome.36
With current microsurgical techniques, the results of OGM resection are excellent, with a high rate of total resection and a low incidence of complications.33 Authors indicated high rate of visual improvement (80% to 100%)33,37,38 with no visual deterioration.
OPTIC NERVE SHEATH MENINGIOMAS
Optic nerve sheath meningiomas (ONSMs) arise either primarily from meningeal cells within the orbit or in the optic canal, or are an extension of intracranial meningiomas that are invading the optic canal. They are also known as primary optic nerve meningiomas or perioptic meningiomas. Most primary ONSMs are unilateral, and 6% are reported to be bilateral; of those, 60% were canalicular.39–41 Secondary ONSMs originate intracranially, usually in the region of the planum sphenoidale or tuberculum sella, and gradually extend into the optic canal. In many cases, it may be difficult to determine whether a meningioma began in the posterior orbit or optic canal and then spread intracranially, or whether the tumor began intracranially and spread into the optic canal.
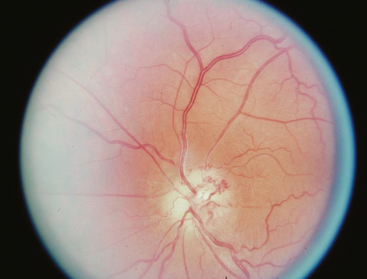
Occasionally these tumors cause no initial changes in the appearance of the optic disc. Examination of color vision, relative desaturation of red compared with the fellow eye, and relative afferent pupillary defect may give a clue to the presence of an optic neuropathy. The optic disc is usually swollen when the tumor surrounds or compresses the intraorbital portion of the nerve. When the patient gradually lose vision, the optic disc swelling begins to resolve, and optociliary shunt vessels may appear on the surface of the disc42 (Fig. 12-3). Optociliary shunt vessels are abnormal blood vessels on the disc, directing blood from retinal circulation to choroidal circulation, which indicate elevated retinal venous pressure with good compensatory circulation. The triad of visual loss, optic atrophy, and optociliary shunt veins is characteristic of an optic nerve sheath meningioma; however, it can also occur with optic nerve gliomas, sphenoid wing meningiomas, advanced glaucoma, and central retinal vein occlusion. The shunt vessels are seen in only a third of optic nerve meningiomas. They often become subtler or resolve as atrophy progresses.43,44
When the tumor originates at the apex of the orbit or within the optic canal, there is slowly progressive visual loss without orbital signs, usually with a normal-appearing optic disc. Although such patients almost always have evidence of an underlying optic neuropathy, the diagnosis may be delayed and the cause of the visual loss may be obscure. In rare cases, small tumors located within the optic canal are impossible to detect using currently available neuroimaging procedures. ONSMs should be suspected in any patient with slowly progressive, unilateral loss of vision associated with signs of optic neuropathy. Intraocular invasion can occur in ONSMs but it is rare.41,45 Metastatic tumors, lymphoma, aneurysm, and inflammatory lesions such as sarcoid or sclerosing orbital inflammation may mimic ONSMs, and these should be considered in the differential diagnosis of a patient with a presumed ONSM.40,46–48
The presence of enlarged, aerated, posterior ethmoid and sphenoid sinuses, a condition known as pneumosinus dilatans, has been reported in association with spheno-orbital meningiomas and considered to be a sign of an adjacent meningioma.49
The diagnosis of an ONSM may be made by a variety of imaging studies. High-resolution computerized tomography (CT), thin-section magnetic resonance imaging (MRI), or ultrasonography make an early diagnosis possible and obviate the need for tissue biopsy in most cases.40,50
CT scan with contrast is an excellent imaging technique for evaluation of ONSM. Thin slices (1.5–3 mm) must be taken to assess the extent of the tumor. ONSM is confined to the dura mater; therefore, it often appears as a distinct, fusiform thickening of the optic nerve. ONSMs usually enhance homogeneously. In addition, linear, diffuse, or patchy calcifications within or along an optic nerve mass are commonly seen.51 ONSMs have three main morphologic patterns on imaging: tubular, fusiform, and globular. Meningiomas surround the optic nerve. The diameter of the nerve is attenuated within the surrounding mass, giving a bull’s eye appearance on coronal images and a tram track appearance on axial images.52 It can be differentiated from an optic nerve glioma, in which the nerve itself is expanded and there is no bull’s eye appearance on coronal section. These changes are particularly well seen after intravenous injection of iodinated contrast material; however, calcifications surrounding the nerve may be masked by contrast enhancement. They are best identified on precontrast soft tissue and bone windowed images. MRI with gadolinium-enhanced fat-suppression, T1-weighted pulse sequences allow visualization of meningiomas as a localized or tubular enlargement with significant contrast enhancement. Compared to brain tissue, meningiomas may appear hypointense on T1-weighted images and hyperintense on T2-weighted images. MRI also provides sufficient tissue detail that one can assess intracranial extension. Ultrasound evaluation of an ONSM can be helpful to show an enlargement in the diameter of the nerve and shadowing from internal calcification.40,41
The treatment of primary optic nerve sheath meningiomas is controversial because they rarely represent a life-threatening process. Surgical excision usually results in complete visual loss due to the damage of the perineural pial vessels of the optic nerve, causing an ischemic infarction. Patients with useful vision are generally not candidates for surgical excision. Surgical excision is considered to minimize secondary orbital problems, such as progressive proptosis, only if the tumor is confined to the orbit and the eye is blind. When intracranial extension is present the decision to excise a tumor depends on (1) whether the contralateral optic nerve or chiasm is threatened; (2) the size, location, and growth of the tumor; and (3) remaining sight in the affected eye.40,51,53,54
For tumors that have already involved the chiasm, surgery is an unacceptable risk to the patient’s remaining vision, and radiotherapy is the only treatment option. Conventional radiotherapy is not preferred because it can damage the optic nerve, retina, and pituitary gland if high doses of radiation are given and lower doses are not effective.55 Fractionated stereotactic conformal radiotherapy appears to be the treatment of choice, and has been demonstrated to improve or stabilize vision in progressive or advanced cases.56 Other options are new frameless radiosurgery devices such as the robotic CyberKnife, an image-guided radiosurgery system that can provide the conformality and accuracy of frame-based systems and at the same time can deliver radiation in several sessions.57
ORBITAL MENINGIOMAS
Orbital meningiomas that do not originate from either the optic fascicle or the intracranial meninges are exceedingly rare. Primary orbital meningiomas originate from ectopic (extradural) meningeal tissue within the orbit.58–60 Such tumors occur in an extradural location but they can compress the optic nerve, the intraorbital contents, the contents of the superior orbital fissure, the cavernous sinus, and the frontal and temporal lobes. Visual loss and proptosis are characteristic findings. Diplopia can occur due to ocular motility limitation, if the vision is intact. Chorioretinal folds are a rare presentation of a meningioma involving the right orbit and right anterior middle cranial fossa.61
Orbital meningiomas are different from those that are located within the dural sheath of the optic nerve (ONSM). Microsurgical removal with subsequent improvement in vision is more common in orbital meningiomas.1 Intraosseous orbital meningioma is a rare cause of chronic optic neuropathy and exophthalmos. Because of their unusual presentation, there are case reports initially misdiagnosed as intraosseous meningioma62 and fibrous dysplasia.63
Surgical resection is the first choice of treatment.64 Frameless computer-assisted images guide the surgeon in the safe resection of these tumors.65 Image guidance is particularly useful when resecting the osseous portion of the tumor because the tissue does not shift with respect to the calibration frame.
SPHENOID RIDGE (WING) MENINGIOMAS
Meningiomas of the sphenoid ridge are traditionally divided into three types: outer, middle, and inner (medial). Outer ridge meningiomas can grow from the pterion toward the sylvian fissure or the tumor infiltrates the dura and invades the bone of the sphenoid ridge, producing marked thickening or hyperostosis (en plaque). Differential diagnosis includes all lesions that can be associated with focal hyperostosis (e.g., osteomas, Paget disease, neurosarcoidosis, tuberculosis, lymphoma, and fibrous dysplasia).51,63
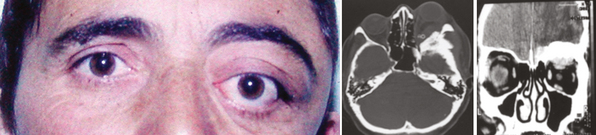
Outer ridge meningiomas often become quite large before producing any symptoms or signs. Most patients eventually present with headache, papilledema, and signs of increased ICP. Some patients develop visual hallucinations. Despite their lateral origin in the region of the pterion, they eventually spread medially across the sphenoid ridge to produce symptoms and signs identical with those produced by inner ridge tumors. The patient develops slowly progressive, unilateral proptosis with periorbital edema. A characteristic swelling in the ipsilateral temporal region develops from hyperostosis and thickening of the greater wing of the sphenoid over a period of several years (Fig. 12-4). As the tumor continues to grow and the posterior orbital bones become increasingly thickened, proptosis becomes severe, and diplopia in the lateral gaze may develop. Loss of vision from optic nerve compression is rare and occurs late. It is ultimately associated with optic disc pallor. Although optic disc swelling occasionally occurs, it is directly related to compression of the orbital portion of the optic nerve and is not papilledema caused by increased ICP.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree