Fig. 16.1
Carotid blowout. (a) Contrast cervical axial CT scan showing recurrent laryngeal cancer encasing the right carotid artery (arrow). (b) Pretreatment angiogram showing a carotid-esophageal fistula with manual compression being applied. (c) Inflation of a balloon in the common carotid artery proximal to the fistula to slow flow and reduce blood loss. (d) Carotid occlusion with coil deployment distal, within the fistula, and proximal to the fistula with EVOH deposition to complete the occlusion
Epistaxis
Epidemiology
Idiopathic epistaxis affects at least 60 % of the adult population during a lifetime; however, only 6 % of epistaxis cases require medical attention. Males and females are equally affected with an increase in frequency over the age of 40. Most cases arise from the anterior septal area; however, 5 % of cases arise more posteriorly and are difficult to control [29].
Associated Conditions
Hereditary Hemorrhagic Telangiectasia (Osler–Weber–Rendu syndrome): a rare autosomal dominant, systemic disease in which epistaxis is caused by rupture of telangiectasias and is often refractory to treatment. While embolization will control an acute episode, symptoms generally recur over time [30].
Eroding cavernous carotid aneurysms (see Chaps. 10 and 11): large and giant aneurysms of the cavernous carotid artery may erode into the sphenoid sinus, rupture, and present with epistaxis [31].
Arteriovenous Malformation or Fistula (see Chaps. 12 and 13): is a rare cause of epistaxis but is the subject of several case reports [32].
Trauma: Traumatic maxillofacial injury is sometimes associated with laceration of branches of the external carotid artery and massive oronasal blood loss. More rarely, skull base fractures are associated with laceration of the cavernous carotid artery. Generally this leads to symptoms of carotid cavernous fistulae (see Chap. 15). However, when there is rupture into the sphenoid sinus, oronasal bleeding may be seen [33].
Sinonasal Neoplasm: Juvenile nasopharyngeal angiofibroma (see below) is the neoplasm most commonly associated with epistaxis. Other tumors that may present in this fashion include: hemangioma, hemangiopericytoma, acute myelogenous leukemia, pyogenic granuloma gravidarum, nasopharyngeal carcinoma, esthesioneuroblastoma, malignant fibrous histiocytoma, adenoid cystic carcinoma, and metastatic disease [34].
Stepwise Treatment Algorithm [29]
1.
Nasal pressure
2.
Topical hemostatic and vasoconstricting agents
3.
Anterior packing
4.
Reversal of underlying factors (i.e., platelet inhibition, anticoagulation, or hypertension)
5.
Endoscopic cauterization: chemical, e.g., silver nitrate or electrocautery
6.
Posterior packing with inpatient observation
7.
Surgical or endovascular ligation of arterial supply to posterior nasal fossa
Angiographic Assessment
During the angiographic assessment of epistaxis, selective catheterization and angiography should be performed in bilateral internal and external carotid arteries. At least one vertebral artery injection may be useful to look for evidence of dural arteriovenous fistula or arteriovenous malformation.
Imaging of the internal carotid artery will rule out rare causes of epistaxis including aneurysms, AVMs, DAVF, or traumatic carotid cavernous fistula. In addition the ophthalmic artery provides important vascular supply to the superior nasal cavity via the anterior and posterior ethmoidal arteries. These arteries are not considered targets for endovascular therapy due to the risk of inadvertent embolization of cerebral or ophthalmic vessels.
Assessment of the external carotid artery is particularly important as the target vessels for endovascular treatment arise from it. Moving from the anterior to posterior nasal cavity, these include the superior labial artery (a branch of the facial artery) and the greater palatine and sphenopalatine arteries (branches of the internal maxillary artery). It is also crucial to look for angiographically subtle external to internal anastomoses through the angular branch of the facial artery and branches of the internal maxillary artery (vidian, accessory meningeal, and middle meningeal). At times it may be feasible to remove nasal packing during angiography to look for extravasation of contrast and localize the side of bleeding more precisely.
Surgical Treatment
Surgical treatment is indicated in patients with epistaxis who continue to bleed despite conservative measures including decongestants, application of cautery, hemostatic agents, and short-term nasal packing. The origin of such intractable epistaxis is difficult to visualize and usually arises from the vessels in the posterior and superior nasal cavity, most commonly the sphenopalatine artery. Surgical intervention provides prompt treatment in these situations. It not only prevents pressure necrosis and infections in the nasal cavity but also reduces the hospital stay from prolonged packing. The nature of surgical interventions has evolved both in the technique and the target of ligation from open ligation of the external carotid artery (ECA) to transantral ligation of internal maxillary artery (IMA) to the endoscopic ligation of the sphenopalatine artery (SPA).
Ligation of the ECA through neck exploration is associated with risk of inadvertent injury to the hypoglossal nerve. It can also cause ischemic complications in atherosclerotic patients whose cerebral circulation is dependent on the external to internal carotid system anastomoses. Open ligation of ECA has been replaced by transantral ligation of IMA that is performed through a Caldwell-Luc approach. In this approach, a window is created through the anterior surface of the maxillary sinus via a gingivobuccal sulcus incision. Through this transantral window, the IMA is ligated in the pterygopalatine fossa which lies posterior to the maxillary sinus. A complication rate of 25–30 % is associated with this technique and mainly includes oroantral fistula, cheek and dental anesthesia, and injury to the nasolacrimal duct.
The most favored approach currently is endoscopic ligation of SPA that has less postoperative complications compared to transantral ligation of IMA [35]. A detailed endoscopic examination of the nasal cavity is performed under general anesthesia after adequately preparing the nasal cavity with topical decongestants. Nasal mucosa and, preferably, the greater palatine canal are injected with 1 % Xylocaine with 1:100,000 epinephrine for additional vasoconstriction. The middle turbinate is medialized and followed to its posteriormost aspect. The sphenopalatine foramen is situated just inferior to the posterior end of the middle turbinate and is accessed by a vertical mucoperiosteal incision on the lateral nasal wall. The SPA is clipped and/or cauterized as it exits the foramen, and the mucosal flap is re-approximated, thus completing the procedure. The success rate with this procedure is reported to be over 85 % [36]. The commonly reported complications include minor rebleeding, nasal crusting, palatal numbness, septal perforation, injury to the nasolacrimal duct, and acute sinusitis. Postoperatively, nasal saline irrigation is recommended to reduce crusting.
Rarely, bleeding from the anterior ethmoidal artery (AEA) can be a source of intractable posterior epistaxis. This is seen mainly in patients with a history of midfacial trauma or iatrogenic injury during sinus surgery and often fails to subside with conservative measures. Surgical intervention requires ligation of AEA via either a traditional approach through an external Lynch incision placed over the medial orbital wall or an endoscopic approach. External approach achieves better control of AEA and also avoids the complications that may occur from an endoscopic approach such as cerebrospinal fluid leak and orbital injury.
Endovascular Treatment
Endovascular procedures to control epistaxis are most often performed under general anesthesia to reduce patient movement and protect the patient’s airway from blood and saliva. Femoral artery access is obtained in the standard manor and a 5 or 6 F guide catheter is navigated into the origin of the external carotid. This vessel is prone to catheter-induced spasm, and consideration should be given to topical application of 1 inch of nitroglycerin to the angle of the ipsilateral jaw prior to catheterization. Intra-arterial nitroglycerine should also be available to relieve spasm that occurs during the case. A large inner diameter microcatheter (e.g., a 0.021 Rapid Transit, Codman) is then navigated over a microwire and positioned within the facial artery distal to the submandibular artery. The authors rely on microparticles for embolization of the ECA branches. We utilize 250–355 μm PVA particles suspended in a contrast slurry ipsilateral to the site of bleeding. Injection is performed under negative roadmap imaging. Brief puffs are administered with careful attention paid to anterograde penetration of the nasal cavity. Once reflux is noted, embolization of these vessels is complete. The particle-contrast slurry must be constantly agitated and attention must be paid to accumulation of microparticles within the hub or the microcatheter in order to avoid occlusion. A new microcatheter is utilized to select the contralateral ECA, is navigated in the internal maxillary artery, and is positioned distal to the deep temporal vessels. The process above is then repeated. The authors will embolize the contralateral ECA to reduce collateral supply to the area of hemorrhage. However, larger-diameter PVA particles (500–710 μm) are utilized in this case to avoid excessive penetration and devascularization of the nasal cavity and skin overlying the nose.
As described above, some interventionalists rely on proximal vessel occlusion of the IMAX through deployment of detachable or pushable coils rather than microparticles. Coils have the advantage of speed and simplicity but do not penetrate the capillary bed and obstruct re-treatment should symptoms recur. Liquid embolic agents are enjoying increasing favor but are higher cost, may be more prone to travel through external to internal collaterals, and also inhibit re-treatment if needed.
Some practitioners will have nasal packing removed in the angiography suite to assess the need for further treatment before concluding the procedure. Others will remove nasal packing the next day.
Efficacy
Embolization is associated with a 90–100 % efficacy in idiopathic epistaxis; however, eventual recurrence is the rule in HHT patients. When early rebleeding (within 30 days) is taken into account, rates of effective treatment are between 70 and 90 % [29].
Risks
While the overall risk of embolization for epistaxis is low, there is risk of serious adverse events such as blindness and stroke that may occur due to reflux of the embolic material into cerebral or ophthalmic collaterals. In addition, external to internal collateral may lead to inadvertent cerebral artery embolization, making the assessment of collaterals crucial. Finally, excessive devascularization of the nasal cavity may lead to erosion and ulceration of the nasal mucosa or skin overlying the nose.
Illustrative Case 2: Epistaxis
A 55-year-old woman was admitted with several days of epistaxis. Upon admission she underwent posterior nasal packing but upon removal once again experienced severe bleeding, requiring transfusion of two units of packed red blood cells. She was then brought to the angiography suit where she underwent PVA particle embolization of both distal facial and internal maxillary arteries. She tolerated the procedure well and was discharged, free of further bleeding, on postoperative day number 2 (Fig. 16.2).
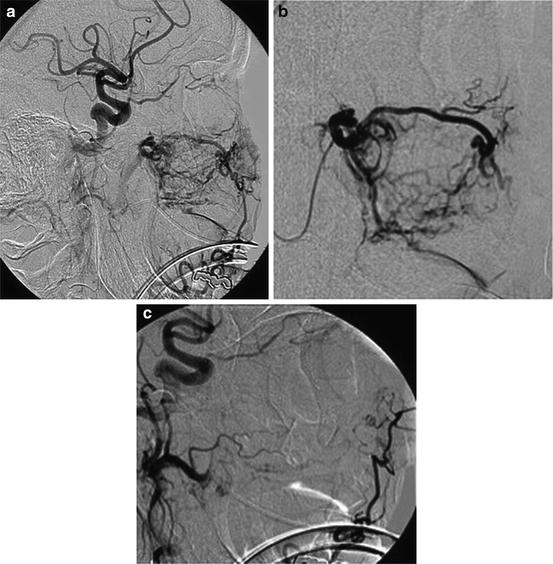

Fig. 16.2
Epistaxis. (a) Guide catheter angiogram showing vascular supply to the nasal cavity (sphenopalatine arteries: bracket, inferior orbital: open arrow, superior labial: small arrow). (b) Microcatheter angiogram of the distal internal maxillary artery (IMAX). (c) Post-PVA embolization showing occlusion of the distal IMAX
Preoperative Tumor Embolization
Risks
The risks associated with tumor embolization are similar to those seen in the treatment of epistaxis and include stroke due to reflux of the embolic agent into the cerebral vasculature or transit through external to internal or external to vertebral artery connections. There is also risk associated with unintentional devascularization of cranial nerves, skin, and mucosa. When large tumors are treated, perioperative edema and mass effect may occur. This risk can be minimized through the judicious use of postoperative steroids and timing the embolization procedure in close proximity to surgical resection.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








