Fig. 1
Gamma aminobutyric acid (GABA A ) modulators (eszopiclone and zolpidem) dose-responsively alter electroencephalogram (EEG) spectral frequency of sleep/wake states in inactive-phase dosing, whereas DORA-22 produces sleep more akin to vehicle-treated animals. Shown are EEG spectral changes (as a ratio of treatment over vehicle) from 1 to 100 Hz within each sleep/wake state for each treatment at respective doses (low, mid, and high). Horizontal shaded area represents 1 (no change from vehicle) ±5 % as observed baseline dosing effect. Ninety-five percent confidence bounds were calculated, and circles indicate nonoverlapping areas above/below baseline dosing effect and 95 % confidence intervals for each sleep/wake state. All treatments and doses were collected as independent 3-day crossover study designs in male Sprague-Dawley rats (n = 16) during the inactive phase (ZT23). [Figure reproduced with permission from Fox et al. (2013)]
5 GABAA-Positive Allosteric Modulators and DORAs: Potential Differences in Cognitive and Behavioral Effects
As alluded to above, GABA is the major inhibitory neurotransmitter in the brain and the GABAA α1 subunit–containing receptor, upon which the standard-of-care therapeutics interact, is found throughout the brain. Therefore, it is not surprising that these compounds produce a variety of effects in addition to impacting sleep. Most notably, compounds such as zolpidem and eszopiclone have been shown to produce cognitive disruption in human subjects, including deficits in attention and memory (Warot et al. 1987; Kuitunen et al. 1990; Balkin et al. 1992; Berlin et al. 1993; Roehrs et al. 1994; Allain et al. 1995; Wesensten et al. 1995; Rush and Griffiths 1996; Verster et al. 2002; Leufkens et al. 2009), and motor disturbances, such as ataxia and loss of balance (Drover 2004; Allain et al. 2005; Zammit et al. 2008; de Haas et al. 2010). These findings are aligned with the high level of GABAA α1 subunit-containing receptor found in the hippocampus and cortex, key regions involved in cognition, and the cerebellum and striatum, important for motor function. Many of these effects are also quite similar to those of ethanol, which also nonselectively impacts GABA receptor function.
Very few studies have directly compared the effects of GABAA-positive allosteric modulators versus DORAs on cognitive performance, but those that have suggest DORAs might be better tolerated. In a series of studies in rat and nonhuman primates, we evaluated the effects of zolpidem, eszopiclone, and DORA-22 on recognition memory in rats and working memory and attention in rhesus macaques (Uslaner et al. 2013). Compound testing was conducted shortly after dosing, while drug levels were at or near their maximal levels. The GABAA-positive allosteric modulators produced cognitive disruption in all three measures in both species at doses that were below or equal to those that promoted sleep. In contrast, DORA-22 increased sleep at doses 30-fold lower than the dose that impacted recognition memory, and no dose of DORA-22 tested impacted working memory or attention in rhesus macaques (Fig. 2). These findings are very similar to those of a more recent publication by Morairty et al. (2014), who demonstrated that, at doses producing equivalent levels of sleep, another DORA, almorexant, had no effect on spatial reference or spatial working memory in rats, whereas the effect of zolpidem was impairing. Finally, in rhesus macaques we have demonstrated that DORA-22 produced no next-day effects on working memory or attention, whereas diazepam produced next-day effects on both measures and eszopiclone produced effects on attention (Gotter et al. 2013). Importantly, in humans a dose of suvorexant five- or ten-fold greater than that necessary to promote sleep had effects on next-day subjective alertness (Sun et al. 2013). Future studies aimed at better characterizing the cognitive effects of these different mechanisms of action are warranted to determine the therapeutic window of each approach.
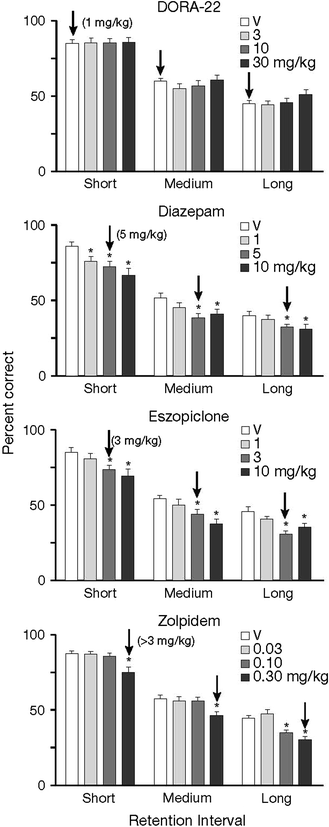

Fig. 2
Percentage correct on a working memory task, delayed match to sample, in rhesus monkeys. Animals were given stimuli and needed to remember them over a short, medium, or long duration. Eszopiclone (3 and 10 mg/kg), diazepam (1, 5, and 10 mg/kg), and zolpidem (0.3 mg/kg) impaired performance, whereas no dose of DORA-22 impacted performance (asterisk indicates significantly different from vehicle). The minimum effective doses to produce sleep as measured by electroencephalogram were as follows: DORA-22 1 mg/kg, eszopiclone 3 mg/kg, diazepam 5 mg/kg, and zolpidem >3 mg/kg (as indicted by arrows). [Figure reproduced with permission from Uslaner et al. (2013)]
In addition to potential differences in the cognitive impact of these drugs in subjects who are awake, it appears that DORAs and the GABAA-positive allosteric modulators may also differentially impact the ability of salient stimuli to arouse the individual when asleep. Specifically, it is important for survival and safety to be able to wake to a meaningful stimulus, such as the calling of one’s name, a fire alarm, or the sound of a predator, but to sleep through irrelevant stimuli, such as a bird chirping or background noise. We have demonstrated that dogs given vehicle or DORA-22 are much more likely to be awakened from sleep when exposed to a relevant, salient stimulus that has been previously paired with reward versus a nonrelevant, neutral stimulus (Tannenbaum et al. 2014). In stark contrast, rhesus macaques given eszopiclone or diazepam displayed no difference in their ability to awaken to a salient versus neutral stimulus, rarely waking to either (Tannenbaum et al. 2013) (Fig. 3). These results suggest that, at least in nonhuman primates, DORAs protect the ability to respond to salient stimuli when awake, whereas GABAA-positive allosteric modulators do not. Furthermore, these findings indicate that alternate arousal pathways responding to strong alerting signals remain intact while orexin signaling is silenced, as is observed when unmedicated subjects are awakened during normal sleep (and when endogenous orexin signaling is silent). This could have important consequences, as experimental data in human subjects show that GABAA-positive allosteric modulators impair the ability to arouse to salient stimuli, such as a fire alarm (Johnson et al. 1987; Mendelson et al. 1988).
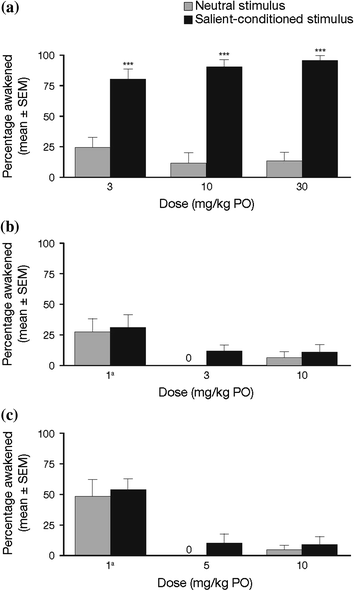

Fig. 3
Arousal from sleep to emotionally salient-conditioned or neutral acoustical stimuli at maximal nighttime drug exposure of a DORA-22, b eszopiclone, or c diazepam. a During DORA-22 sleep, rhesus monkeys (n = 12) woke to salient-conditioned acoustical stimuli significantly more than they woke to neutral stimuli. b During eszopiclone treatment, monkeys did not discriminate between salient and neutral stimuli; monkeys tended to sleep through both stimuli. c During diazepam treatment, monkeys did not discriminate between salient and neutral stimuli; at doses that induce sleep (5 and 10 mg/kg), monkeys slept through both stimuli. Data shown are mean ± standard error of the mean. ***P < 0.001. aNonsedating daytime dose as measured by electroencephalogram. [Figure based on data from a study presented by Tannenbaum et al. at the 27th Annual Meeting of the Associated Professional Sleep Societies (APSS), June 2013, Baltimore, MD (Tannenbaum et al. 2013]
In addition to potentially differentiating with respect to cognitive performance, there is some evidence that the effects of these two mechanisms of action differ with respect to motor impairment. GABAA-positive allosteric modulators have been shown to produce motor disturbances, including loss of balance and an increased likelihood of falls, particularly in the elderly (Vermeeren 2004; Drover 2004; Allain et al. 2005; Zammit et al. 2008; de Haas et al. 2010). Far fewer clinical studies have characterized the motor effects of DORAs in humans. A very high dose of almorexant (1000 mg) was found to impact body sway; the effect was less pronounced than for zolpidem (10 mg), and it appeared to tolerate with repeated dosing (Hoever et al. 2010, 2012). In addition, whereas zolpidem produced reports of abnormal coordination and “feeling drunk,” no dose of almorexant produced such an effect (Hoever et al. 2010). However, other measures of sensorimotor processing, such as the ability of the eyes to smoothly follow an object or saccade, appear to be more sensitive to almorexant. Finally, interactions between alcohol and almorexant or suvorexant appear to be additive, but not synergistic, to any of the clinical effects produced by almorexant or suvorexant alone (Hoch et al. 2013).
Preclinical studies also suggest that DORAs might have less impact on motor coordination than GABAA-positive allosteric modulators. Using the rotarod performance test, in which the ability of an animal to maintain its balance on a rotating accelerating rod is assessed, two studies (Steiner et al. 2011; Ramirez et al. 2013) reported that two different DORAs, DORA-12 and almorexant, did not disrupt rotarod performance in rats at doses well above those necessary to produce sleep. In contrast, both studies showed that zolpidem and eszopiclone produced significant impairment in rotarod performance (Fig. 4). Finally, both studies showed that DORAs administered in the presence of ethanol did not impair rotarod performance, whereas Ramirez et al. (2013) showed that ethanol produced synergistic impairment when combined with zolpidem or eszopiclone. The interaction between ethanol and GABAA-positive allosteric modulators is likely due to the fact that these compounds act on the same receptor, whereas DORAs act on a distinct set of neuronal circuits.
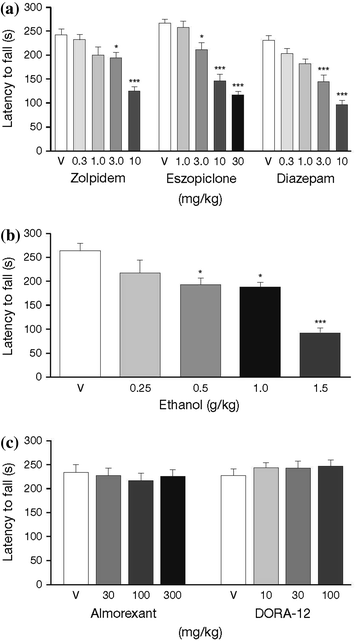

Fig. 4
Dose—response curve for latency to fall from rotarod for rats administered a GABAA receptor modulators, b ethanol, and c orexin receptor antagonists. Acute administration of zolpidem, eszopiclone, diazepam, and ethanol dose-dependently impaired rotarod performance. In contrast, the orexin receptor antagonists almorexant and DORA-12 did not impair rotarod performance after acute administration. Data shown are mean ± standard error of the mean. *P < 0.05, ***P < 0.001 versus vehicle-treated group. [Figure reproduced with permission from Ramirez et al. (2013)]
6 Summary and Future Directions
Insomnia is a debilitating disorder that affects a significant proportion of the population and has substantial health and economic consequences. As reviewed in this chapter, the current standard of care, the GABAa positive allosteric modulators, were discovered before much was understood regarding the neural mechanisms underlying sleep. These compounds produce a range of effects in addition to increasing sleep, including reducing REM and disrupting the spectral frequency observed during normal sleep, impairing cognition and disrupting motor coordination. In comparison, the DORAs have only recently been developed and as a result have less clinical characterization, but may offer some potential advantages. The discovery and development of orexin receptor antagonists was a concerted effort of rational drug design stemming from a core understanding of orexin biology, genetics, sleep physiology, and neuroanatomy. It was hypothesized that DORAs would promote sleep without disrupting spectral frequency and impairing cognition and motor coordination, and preclinical and clinical data appear consistent with this hypothesis. More clinical experience is necessary to better understand the effect of DORAs in humans and how they compare with the standard of care. Suvorexant is currently under evaluation by regulatory agencies for the treatment of insomnia, and if approved, would represent the first DORA available for patients suffering from this sleep disorder.
References
Balter MB, Uhlenhuth EH (1991) The beneficial and adverse effects of hypnotics. J Clin Psychiatry 52(Suppl):16–23PubMed
Bettica P, Squassante L et al (2012) Differential effects of a dual orexin receptor antagonist (SB-649868) and zolpidem on sleep initiation and consolidation, SWS, REM sleep, and EEG power spectra in a model of situational insomnia. Neuropsychopharmacology 37(5):1224–1233PubMedCentralCrossRefPubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







