Fig. 1
Secure ligatures of the subclavian artery catheter. (a) The subclavian artery is dissected and secured with a proximal and distal ligature. One ligature is kept in place to secure the catheter. (b) After arteriotomy and retrograde cannulation, the catheter is fixed. (c) The catheter is fixed with one ligature (hollow arrow) and additionally secured and kept in placed by fixation to the distal ligature (solid arrow). The proximal ligature (asterix) is kept in place in case of slippage of the catheter during repositioning
Angiography
Angiography can be either performed non-selectively through retrograde intra-arterial bolus injection of non-ionic Iopamidol, or selectively through an angiographic catheter inserted into the rabbit’s left vertebral artery (Fig. 2). Each angiogram is calibrated according to an external sizing device (stainless steel skin stapler) placed over both mandible angles at the time of baseline DSA. Measurement of the vessel is performed three times (along a predefined length from the tip of the basilar artery) in a blinded fashion using automatic measurement analysis software, and mean values are determined (Fig. 3). Owing to these measures, we were able to keep intra- and inter-observer variability low.
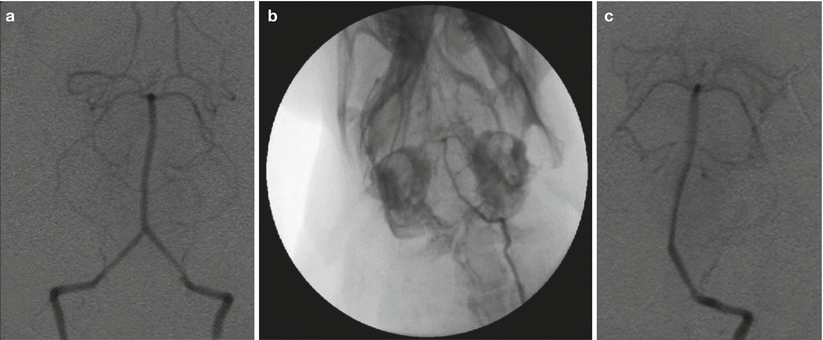
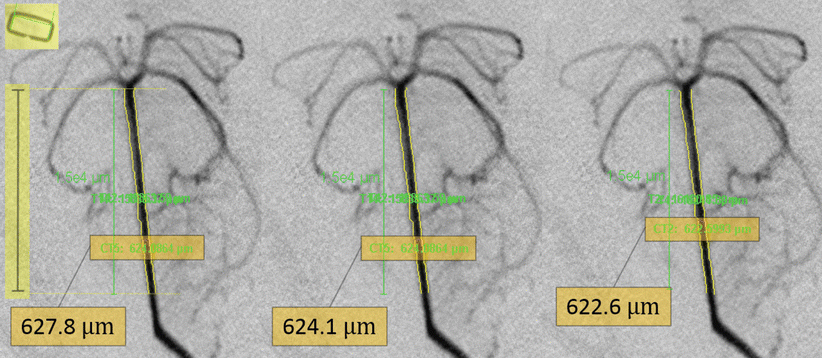

Fig. 2
Non-selective and selective angiography. (a) Non-selective angiography through retrograde intra-arterial bolus injection via subclavian artery. (b, c) Selective angiography through an angiographic catheter inserted into the rabbit’s left vertebral artery

Fig. 3
Calibration and measurement of angiograms. Each angiogram is calibrated according to an external sizing device (skin stapler, highlighted left upper corner). The vessel diameter is measured along a predefined length (highlighted scale on the left) from the tip of the basilar artery using automatic measurement analysis software. The measurements are performed three times and the mean value is determined
Thirteen studies addressed the delayed occurrence of vasoconstriction after a single blood injection into the cisterna magna and showed maximal narrowing on day 2–4 [12]. In accordance with this body of literature, we performed follow-up angiography on day 3 after blood shunt-induced SAH on day 0. However, the exact time course using this novel approach remains to be determined by a study of serial DSA.
Closed Cranium Condition and Cisterna Magna Puncture
To preserve the closed cranium condition, it is necessary to seal all burr holes with a thick plug of bone wax before SAH induction. After ICP reaches its nadir or intentional termination of the bleeding, the spinal access needle should be kept in place until ICP returns to a steady state close to baseline values. Incorrect positioning of the spinal access needle can result in significant morbidity and mortality. Therefore, one should be extremely careful with this step. After confirmation that the rabbit is fully anaesthetised, the needle can be slid down the down bony external protuberance until a gap is felt. Once having entered the cisterna magna, the needle should not be pushed any further unless spontaneous dripping of cerebrospinal fluid (head tilted down at 20° for at least 1 min) does not occur.
Outcome Measures
Neurological Grading
The four-point neurological grading system reported by Endo et al. [3] is a feasible tool often applied for evaluation of clinical signs after experimental SAH in rabbits. We graded neurological performance at 6, 12, 24, 48, and 72 h after SAH as follows. Grade 1, no neurological deficit; Grade 2, minimal or suspected neurological deficit; Grade 3, mild neurological deficit without abnormal movement; and Grade 4, severe neurological deficit with abnormal movement. Aggravation of neurological deficits was noted on days 1–3 after SAH induction. Previous studies confirmed that this grading system in rabbits correlates well with the onset of DCVS [4, 5]. More recently, other neurological scales developed to assess myelopathy in rabbits and SAH in dogs have been applied as clinical endpoints after single [8] and double [20] cisterna magna blood injection in rabbits.
SAH Bleeding Scale
An ultrasound flow probe mounted on the blood shunt allows quantitative assessment of haemorrhage volume. Although one needs to be aware of the rabbit’s ability to rapidly clear subarachnoid blood, the severity of SAH based on the amount of blood can also be roughly estimated by grading the subarachnoid clots at the time of brain harvest [18, 20].
Applicability for Studying Early Brain Injury and Delayed Cerebral Vasospasm
The shunt model was originally developed to mimic pathophysiological mechanisms of acute SAH more closely in a closed cranium, thereby triggering moderate to severe degrees of DCVS [15]. Subsequent larger series using the model demonstrated an average of 30 % basilar artery narrowing on day 3 after SAH (unpublished data). These figures tempered the initial enthusiasm of >50 % of DCVS but are still at the upper end of what can be expected after single cisterna magna blood injection in rabbits [10, 12].
The blood shunt model has been shown to provoke acute hemodynamic disturbances and consistent early damage to the hippocampus, basal cortex, and cerebral vasculature 24 h after procedure [11]. Thus, the model seems to qualify for the study of the pathophysiology of EBI and their sequels after SAH. In light of the assumption that early damage to endothelial cells [1, 21] may trigger, aggravate, and maintain DCVS, the rabbit could play an increasingly important role in research of both the pre-vasospasm and DCVS phase after SAH, especially for the screening of potential preventive DCVS treatment modalities that act early in the pre-vasospasm phase.
Advantages and Limitations
Species-specific advantages of the rabbit model are the relatively low cost of the animal, the non-aggressive behaviour and therefore easy handling, and the fact that DCVS monitoring techniques including DSA [19], computed tomography angiography [7], and transcranial Doppler [17] are more readily applied than in smaller animals. A specific advantage of the shunt model is that the acute pathophysiological changes reflect those events seen during aneurysm rupture. Accordingly, SAH induction causes a rapid increase in ICP with a subsequent drop to almost zero in cerebral perfusion pressure (CPP), along with marked and equal reduction of regional cerebral blood flow in both cerebral hemispheres. The bleeding results in characteristic blood distribution with severe grades of SAH, with pronounced and consistent early ischemic brain and vessel damage on day 1. Furthermore, it provokes moderate to severe DCVS on day 3 after the onset of SAH. The shunt model allows for quantitative assessment of haemorrhage volume and corresponding time course. The model is reproducible, and “spontaneous” SAH induction is performed examiner-independently while simultaneously creating potential varying degrees of “controlled” SAH severity.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





