Fig. 1
Preparation of the rabbit subarachnoid hemorrhage model. (a) Cardboard with a hole for head fixation to keep a bent-neck forward position. (b) Sagittal section image and three-dimensional volume-rendering image of the rabbit head from computed tomography scan, showing the puncture point and direction of the needle (white arrows) for puncture of the cisterna magna. (c) Tilting the head 30° down on an adjustable bed with the feet of the rabbit strapped to the bed frame using silicone tubes. (d) Clothespin to stem bleeding from the central auricular artery and marginal auricular vein
Causes of Death in the Procedure
Deaths occurring during the procedure are mainly caused by a failure of puncture and rapid elevation of intracranial pressure (ICP). Failure to achieve accurate puncture, such as brainstem needling or intraparenchymal blood injection, often leads to the death of the animal. To avoid puncture failure, the following factors may be important: having an appropriate posture, not evacuating too much CSF, and directing the needle slightly rostrally and puncturing as the tip of the needle runs through under the foramen magnum. A rapid increase in ICP causes respiratory arrest or cardiac arrest during the procedure. These problems tend to occur with the second injection. Pain stimulation or thoracic compression often restores respiration and cardiac rhythm. A slow injection is the most important factor to consider. We usually perform the injection over the course of 1 min. However, we sometimes take a much longer time for the injection, depending on the respiratory condition during the injection. We often encounter twitching of the roots of the ear when the injected blood reaches approximately 2.5–3.0 ml. This response may be caused by compression or stretching of the facial nerve by the injected blood. We consider this as a warning sign of reaching the maximum amount of blood able to be injected. A 2.5-ml syringe is appropriate for injection, because we can feel the resistance of the ICP through the syringe. After the injection of blood, observation of the respiratory condition for 10 min is important, because respiratory arrest mostly occurs in the first few minutes after injection.
Epidural, Subdural, and Intraparenchymal Hematomas
On rare occasions, blood is injected into spaces other than the subarachnoid space because of accidental movement of the needle chip induced by body movement, loss of traction of the subarachnoid membrane induced by excessive evacuation of CSF, or intense negative pressure during CSF evacuation. To avoid body movement, appropriate control of the depth of anesthesia is important, especially in our procedure, which does not require strict head fixation using instruments. Sodium pentobarbital regulates the depth of anesthesia. We confirm the depth of anesthesia by providing painful stimulation just before puncture. If the anesthesia is weak, finger pressure on the skin around the puncture point easily induces body movement. Deep anesthesia easily induces respiratory arrest following injection of blood.
After Blood Injection
Immediate withdrawal of the puncture needle causes leakage of injected blood with CSF from a pinhole in the dura mater. An indwelling venous catheter should also be maintained during the entire process in case additional anesthesia is required. It is therefore important to not withdraw all of the needles during the entire process. Preservation of the artery and vein in the ear is important, especially in the double-injection method, because we often use the same vessel repeatedly. After withdrawal of the needle, firm pressure should be applied on the pinhole to stem bleeding, so as to not damage the ear vessels by hematoma formation. We use clothespins to completely stem bleeding (Fig. 1d).
Outcome Measures
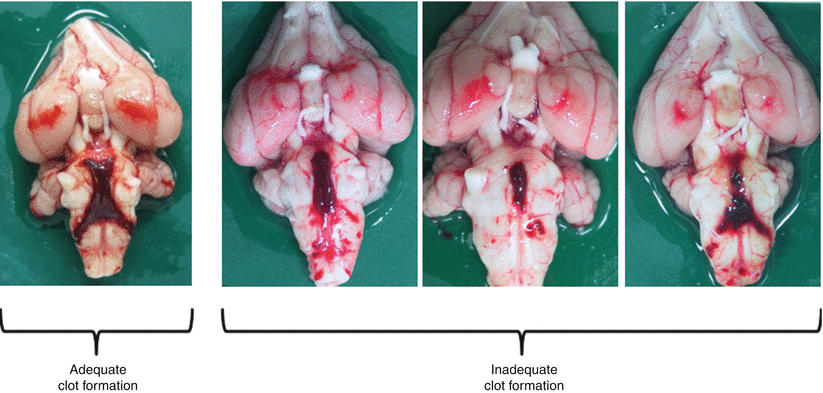
Blood clot formation, degree of vasospasm, cerebral perfusion, neurological and behavioral deficit, and appetite loss have been evaluated as outcome measures of the rabbit SAH model. Brief assessment of clot formation is important for onsite determination of the propriety of the SAH model just after sacrifice. A single report by Zhou et al. [16] described a scoring method of residual clots along the basilar artery of the rabbit SAH model. In our laboratory, when clots do not form sufficiently (Fig. 2), those models are excluded from the following experiments using the basilar artery. Expansion of clot formation can be evaluated quantitatively using imaging software [5]. For the evaluation of arterial narrowing, digital subtraction angiography (DSA), measurement of crosssectional area using a perfusion fixation sample, direct observation, and computed tomography (CT) angiography are available in rabbits. DSA and crosssectional area measurement are the most popular in rabbits [10]. Evaluation of the external diameter of the basilar artery through direct observation is readily applied on sacrifice day [6, 7]. A potential disadvantage of this method is underestimating arterial narrowing because the thickness of the arterial wall is included in the value. In our model, vasospasm occurs on day 3 after the first injection of blood and peaks on day 5 (data not shown). Recently, the usefulness of CT evaluation of vessel diameter and cerebral perfusion in rabbits has been reported [8, 11]. Few reports have described neurobehavioral scoring of the rabbit SAH model. The grading systems by Endo et al. [3] and Zhou et al. [16] are well known [3, 16]. Our rabbits show some slight loss of appetite on days 3 and 5, but rarely show neurological deterioration (data not shown), suggesting that the rabbit double-injection model does not tend to be symptomatic.