17 Douglas Kondziolka, John C. Flickinger and L. Dade Lunsford Stereotactic radiosurgery is used for patients with deep-seated, high-risk cavernous malformations of the brain. If resection is judged to be high risk, we perform radiosurgery for patients with multiple, symptomatic, imaging-confirmed hemorrhages. This chapter examines the long-term results after radiosurgery. Management of brain cavernous malformations (angiographically occult vascular malformations, cavernous angiomas, cavernomas) remains controversial. Since the mid-1980s, there has been improved understanding of their natural history,1–9 as well as increased experience with surgical resection.10–16 In the case of arteriovenous malformations (AVMs), the elimination of the angiographically identifiable anatomic shunt can be demonstrated on imaging and correlates highly with cure. Unfortunately, imaging cannot confirm cure of a cavernous malformation after radiosurgery, as they cannot be defined by angiography. Some patients have cavernous malformations that are not amenable to surgical resection with acceptable risk. When such malformations repeatedly bleed, they warrant management. Stereotactic radiosurgery is a safe intervention that provides a reduction in hemorrhage risk after an initial latency interval for patients with these high-risk cavernous malformations.17–26 These observations confirm the hypothesis that radiosurgical intervention reduces subsequent bleeding rates. The microvasculature of a cavernous malformation ultimately responds to radiosurgery in the same way AVMs respond.27,28 Without an imaging correlate of risk elimination, clinical follow-up remains the standard by which radiosurgery must be judged. High-risk cavernous malformations were managed with stereotactic radiosurgery at the University of Pittsburgh between 1987 and 2003 in a total of 110 patients. There were 58 male and 52 female patients with a mean age of 39 years (range, 4 to 81 years). Almost all patients had multiple hemorrhages (range, 2 to 9), and some suffered a single hemorrhage but had a subsequent stepwise decline in neurologic function. A hemorrhage was defined as a symptomatic, ictal event that consisted of new neurologic symptoms or deficits and imaging confirmation of new blood on magnetic resonance imaging (MRI) or computed tomography (CT). Patients were selected for radiosurgery when the malformation caused functional deterioration due to hemorrhage. Four patients had seizures. In general, the lesions tended to be located in critical brain regions as demonstrated in Table 17-1. Prior to radiosurgery, 30% of patients had surgical interventions that included attempted malformation resection, clot evacuation, biopsy, or shunt placement. One patient had proton beam irradiation and Gamma Knife radiosurgery prior to treatment at our center.
Radiosurgery for Cavernous Malformations
 Clinical Experience of the Authors
Clinical Experience of the Authors
Epidemiology
| Location | Number |
|---|---|
| Pons/midbrain | 60 |
| Thalamus | 18 |
| Medulla | 3 |
| Temporal lobe | 6 |
| Parietal lobe | 7 |
| Basal ganglia | 7 |
| Frontal lobe | 4 |
| Cerebellum | 3 |
| Occipital lobe | 1 |
| Other | 1 |
 Radiosurgical Treatment
Radiosurgical Treatment
Radiosurgical Technique
Prior to radiosurgery, all patients underwent MRI to ensure that the lesion was a typical cavernous malformation. Typically, MRI showed mixed signal change within an outer hemosiderin ring of low signal intensity (Fig. 17-1).28–30 If there was any question about the diagnosis, angiography was performed to exclude an AVM or associated venous malformation.
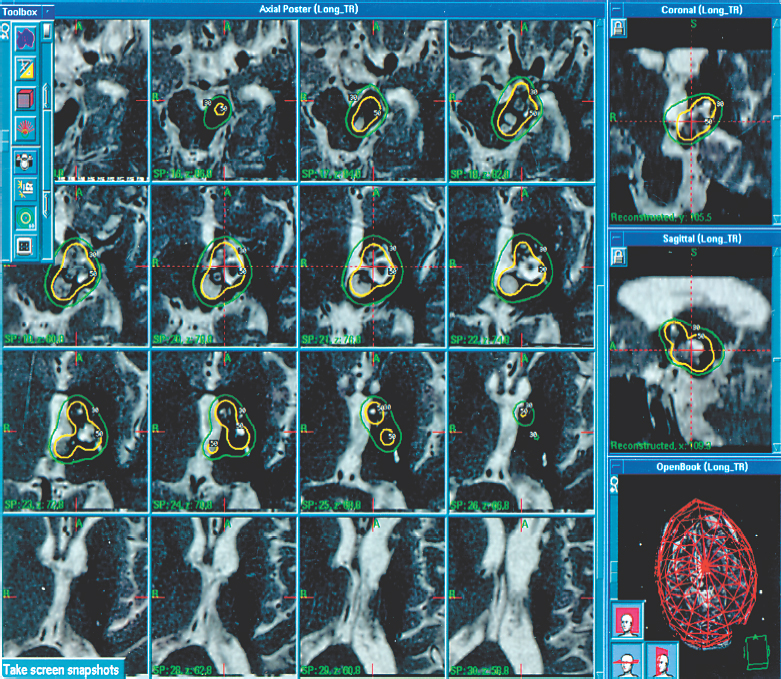
Figure 17-1 Axial magnetic resonance scan at the time of radiosurgery showing a left thalamic and superior midbrain cavernous malformation. This 22-year-old man had five prior symptomatic hemorrhages and three prior subtotal resections. Radiosurgery was planned with seven 8-mm isocenters. A margin dose of 14 Gy and a maximum dose of 28 Gy were administered. Six months later, the lesion was smaller as much of the blood had been resorbed.
Radiosurgery was performed with the use of the Leksell model G stereotactic frame (Elekta Instruments, Atlanta, GA, USA). The frame was applied after mild sedation and local anesthesia was administered. General anesthesia was reserved for patients under 12 years of age. After frame application, all patients had stereotactic imaging. CT was used for planning in all patients prior to 1990. Patients treated from 1988 through 1992 had both CT and MRI. Since 1992, stereotactic MRI alone has been utilized because MRI is superior to CT in defining cavernous malformations and is equally accurate. A sagittal short-repetition time (TR) scout image acquisition was obtained, followed by axial short-and long-TR images obtained at 3-mm image intervals. Finally, repeat axial and coronal short-TR images with volume acquisitions (1 to 1.5 mm slices) and contrast enhancement were obtained.
Images were transferred to the dose planning workstation of the Gamma Knife (GammaPlan; Elekta Instruments). A team composed of a neurosurgeon, radiation oncologist, and medical physicist selected the target and composed the dose plan. Single or multiple isocenter (range, 1 to 9) plans were constructed to give a conformal irradiation volume for the cavernous malformation margin (Fig. 17-2). The mean number of isocenters was 3.2. The target nidus was defined as the region characterized by mixed signal change within an outer hemosiderin ring, typically of low signal intensity. Hematoma eccentric from the malformation was excluded from dose planning. In all patients in this series, the 50% isodose or greater was used for the target margin. The radiosurgical dose selection was set just below that advocated for angiographically identifiable vascular malformations and was, therefore, dependent on the location and volume of the cavernous malformation.20,22 The volume was calculated as the sum of the voxels within the isodose used to envelop the malformation margin. The mean volume was 1.36 mL (range, 0.12 to 9.5), and the mean maximum and marginal doses were 30 Gy (maximum = 40) and 15.7 Gy (maximum = 20 Gy), respectively. Radiosurgery was performed with a 201-source cobalt-60 Leksell Gamma Knife, models U, B, or C (Elekta Instruments). After radiosurgery, all patients received 40 mg methylprednisolone and were discharged from the hospital within 24 hours.
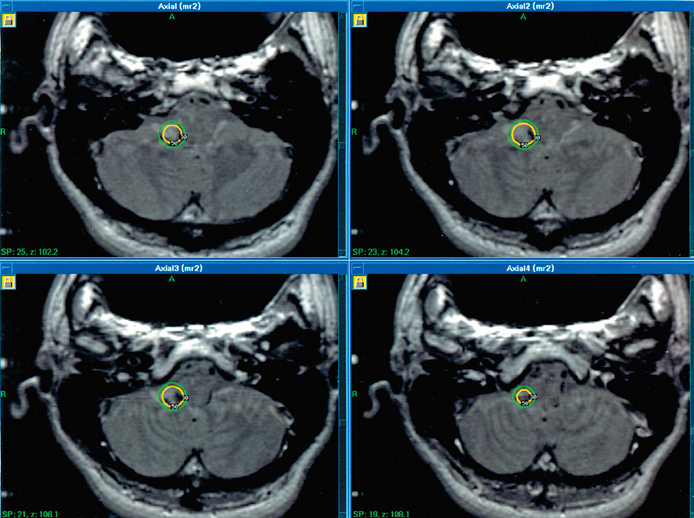
Figure 17-2 This 33-year-old patient presented with multiple hemorrhages (vertigo and vomiting) in association with a cavernous malformation of the right inferior cerebellar peduncle. Radiosurgery was performed with one 8-mm isocenter to deliver a margin dose of 14 Gy.
Follow-up
Clinical follow-up data was obtained from either the patients or their referring physicians if they lived at a distance from Pittsburgh. Where necessary, patients were contacted by telephone to update their outcome for the purposes of this study. Imaging follow-up was requested at 6-month intervals for the first 2 years after radiosurgery, and then annually. The following equation was used to determine hemorrhage rates:

Hemorrhage rates were compared before and after radiosurgical intervention using a paired t-test. A hemorrhage was defined as a new neurologic symptom or sign associated with new blood detected on MRI.
Hemorrhage Rates before Radiosurgery
Patient observation before radiosurgery began with the first symptomatic, image-documented hemorrhage and ended with radiosurgery. At our last comprehensive review, a total of 354 patient-years were observed by this definition, giving a mean observation time of 4.33 years per patient (range, 0.17 to 18 years). During this period, 202 hemorrhages (2.46 per patient) were observed. Multiple hemorrhages were documented in 76 patients (range, 2 to 7), whereas six patients had one hemorrhage. The first hemorrhage of the 202 hemorrhages was excluded, leaving 120 subsequent hemorrhages observed in 354 patient years. This gave an annual hemorrhage rate of 33.9%, a rate that remained fairly stable over five separate annual observations. After the first bleed, the annual hemorrhage rates in years 1 through 5 were 52%, 35%, 39%, 24%, and 32%, respectively.
Hemorrhage Rates after Radiosurgery
The mean follow-up after radiosurgery was 4.89 years per patient (range, 0.42 to 12.08 years), with 57 patients having at least 2 years of follow-up and a total of 401 patient-years of follow-up. During this period, 19 hemorrhages (0.22 per patient) were identified in 15 patients. Of these hemorrhages, 17 occurred in 13 patients during the first 2 years after radiosurgery, representing 138 patient-years of observation, for an annual hemorrhage rate of 12.3% per year. After the expected latency period, two hemorrhages were identified during 262 patient-years of observation, giving an 0.76% per year hemorrhage rate from years 2 to 12.20 One patient had neurologic deterioration accompanied by increased edema on T2-weighted MRI and increased high signal on T1-weighted MRI, suggestive of new blood, at 5 years. The other patient’s bleed was asymptomatic, but follow-up imaging at 10 years showed an increase in size and high signal intensity in T1-weighted imaging. There was no significant difference between the maximum dose received, the margin dose received, the number of isocenters, or the number of hemorrhages prior to treatment between the group who hemorrhaged after radiosurgery and those who remained hemorrhage free.
The mean number of hemorrhages per patient was significantly reduced after radiosurgery (2.43 vs. 0.22, P < 0.0001) as well as after the 2-year latent interval (0.19 vs. 0.02, P < 0.01). We compared a group of 52 patients who had their first hemorrhage more than 2 years before radiosurgery (group 1) with 30 patients who had their first symptomatic bleed within 2 years of radiosurgery (group 2). During the 2 years after radiosurgery, the annual bleeding rates were 16.6% and 4.2% for groups 1 and 2, respectively. Two years after radiosurgery, the rates were 1.1% and 0%. This data indicated that the hemorrhage rate after radiosurgery was independent of the time from the first hemorrhage.20
Morbidity of Radiosurgery
Twelve patients (12.4%) had new neurologic symptoms without hemorrhage after radiosurgery. Such new symptoms are suspected to be adverse radiation effects (AREs). AREs have been uncommon since 1992, when we instituted lower margin doses and switched exclusively to MRI-based targeting. Seven of the new deficits were minor and six of these were temporary. Complications were seen with 8 of 51 lesions located in the pons or midbrain, 2 of 13 in the thalamus, and 1 of 7 in the medulla. All radiosurgical complications were seen within a year of radiosurgery. Patients with AREs received a small, but significantly higher marginal dose (17.45 vs. 16.05, P < 0.03), delivered by a lower number of isocenters (1.64 vs. 3.06, P< 0.01), and tended to have more previous hemorrhages (3.18 vs. 2.32, P< 0.001). There were more complications observed with malformations in the brain stem or diencephalon compared with other sites.
Patients were chosen for radiosurgical management because they had progressively symptomatic cavernous malformations, located in areas that are associated with unacceptable surgical risk of morbidity. In general, they did not present at a pial or ependymal surface. Morbidity after radiosurgery is higher when the malformation is located in an area of critical brain function. We suspect that AREs are related to the hemosiderin ring surrounding the malformation, corresponding with a region of normal brain stained with iron pigment, which is a potential radiation sensitizer.
 The Goal of Radiosurgery: Reduction in Hemorrhage Risk
The Goal of Radiosurgery: Reduction in Hemorrhage Risk
We believe that radiosurgery on such high-risk, cavernous malformations must improve outcomes compared with the natural history of these lesions. Any treatment modality must rely on clinical follow-up to demonstrate its effectiveness and justify its use. Studies of the natural history of asymptomatic cavernous malformations have suggested that they have a relatively low yearly risk of hemorrhage.1-9 A study at the University of Pittsburgh concluded that the overall annual risk of hemorrhage was 2.6%.5 However, when these patients were stratified into those who had previously suffered a hemorrhage and those who had not, the former group appeared to be at higher risk. Patients with one previous hemorrhage had a yearly 4.5% risk of hemorrhage, whereas those without had a 0.6% yearly risk. Patients with two or more hemorrhages had a bleeding rate of ~30% per year.23 It was for this reason that the patients with prior hemorrhage were chosen for radiosurgery. It follows, then, that their results may differ from the former groups at lower risk. Barker et al. hypothesized that symptomatic cavernous malformation hemorrhage may occur in a pattern of temporal clustering.31 They noted a 2.4-fold decline in the hemorrhage rate after 2.5 years and suggested that this alone may be responsible for the reduced bleeding rate seen after radiosurgery. These data were derived from a series of 141 patients who had resection or proton beam irradiation of their malformation over an 18-year period. However, only 63 patients had a second hemorrhage. This series is different from ours in that most patients were not observed to see if they would rebleed without treatment.
We believe that the observed 33.9% yearly risk of hemorrhage prior to radiosurgery in our series warranted intervention. Within 2 years of radiosurgery, this risk of hemorrhage was reduced by one-third to 12.3% per year. After the anticipated 2-year latency interval, the yearly risk of hemorrhage was further reduced to 0.76%. This risk approximated the 0.6% yearly rate of hemorrhage seen in patients who harbor asymptomatic cavernous malformations.5 Radiosurgery was associated with a greater than 30-fold reduction in the baseline risk of symptomatic bleeding.
 The Radiobiological Effect of Radiosurgery
The Radiobiological Effect of Radiosurgery
It is not clear why the risk for hemorrhage is reduced after radiosurgery. Our hypothesis is that the endothelial-lined channels undergo progressive hyalinization leading to thickening and eventual luminal closure, perhaps via the chronic inflammatory response typical of radiation-induced vasculopathy.3,23,32 Unfortunately, there are few reports about the histology of cavernous malformations after radiation. Gewirtz et al. reported pathologic changes in 11 patients who underwent surgical resection after irradiation.33 Of these lesions, eight were identified as cavernous malformations, one was identified as a true AVM, and two were not identified adequately. No malformation was completely thrombosed; this is not surprising as most had rebled necessitating surgical resection. Six lesions showed a combination of marked vessel fibrosis, fibrinoid necrosis, and ferrugination. In addition, Karlsson et al. showed a cavernous malformation specimen that was treated with Gamma Knife radiosurgery, revealing that more than 70% of the lesion had been obliterated.21
Chang et al. reported 57 patients with surgically inaccessible cavernous malformations treated by helium ion or linear accelerator radiosurgery.18 They found an annual hemorrhage rate of 9.4% was reduced to 1.6% after 36 months elapsed after radiosurgery. Complications included symptomatic radiation edema (7%), necrosis (2%), and increased seizure frequency (2%). Amin-Hanjani et al. reported 95 patients with 98 cavernous malformations who were treated with stereotactic Bragg-peak proton beam therapy.10 They found that the annual hemorrhage rate was reduced from 17.3% before treatment to 4.5% after a latency period of 2 years with a 16% incidence of permanent neurologic deficit and a 3% mortality rate. Karlsson et al. reported a hemorrhage rate after radiosurgery of 8%, but they cautioned on the use of this technique because of side effects (27%).21 Some of their patients were treated without MRI guidance and at doses that currently would be considered excessive.21 Some may have had associated venous anomalies.
In contrast, Steinberg et al. reported results after microsurgical resection in 56 patients with 57 deep angiographically occult vascular malformations (AOVMs).15 They also reported a long-term neurologic morbidity rate of only 5% and a complete lesion resection of 93% after the initial planned resection. This figure may have been overestimated because two of their patients had rebleeding despite postoperative MRI that suggested complete resection. Compared with surgical resection, the obvious disadvantage of radiosurgery is the latency interval necessary to achieve a reduction in the bleeding rate. This disadvantage is outweighed by the lower risk of radiosurgery for hemorrhagic malformations completely located within critical brain parenchyma. Although Pollock et al. reported a reduction in the bleeding rate after radiosurgery in their series of 17 patients (reduced from 40% before radiosurgery to 2.9% two years later), they found a radiation-related morbidity rate of 41%.26 This may have been due to the high median margin dose of 18 Gy.
It is still unclear whether radiosurgery should be offered to a patient after one symptomatic hemorrhage. Good clinical decision-making is difficult in this group because their overall yearly hemorrhage rate is ~4 to 5% per year.5 Any intervention for this group must have a lower overall morbidity associated with it and offer a clear benefit. Perhaps the best patient for radiosurgery after a single bleed is a younger patient whose first hemorrhage caused disabling symptoms. Because the morbidity of radiosurgery for cavernous malformations may be much lower than previously thought, and the rate of hemorrhagic risk reduction dramatic, we think that after a single-bleed, radiosurgery is reasonable for selected patients. A decision analysis model should help to elucidate the crossover points of morbidity and hemorrhage rate that would make radiosurgery an appropriate treatment strategy for patients with a first hemorrhage from a cavernous malformation.
References
14. Pozzati E. Thalamic cavernous malformations. Surg Neurol 2000;53:30–40
26. Pollock BE, Garces YI, Stafford SL, Foote RL, Schomberg PJ, Link MJ. Stereotactic radiosurgery for cavernous malformations. J Neurosurg 2000;93:987–991
< div class='tao-gold-member'>




