Fig. 1
Rat endovascular perforation model of SAH. (a) The technique used for SAH induction (Adapted from Bederson et al. [1]); (b) SAH surgery; arrow cannula placed in the cisterna magna for ICP recording
Advantages/Limitations
Advantages
A number of advantages have made the endovascular perforation model a favorite for studying early injury after SAH, such as:
Replicates early hallmark events of SAH: Accumulation of blood in the subarachnoid space, rapid rise in ICP, fall in CBF, a transient increase in MABP (Cushing response), and fall in cerebral perfusion pressure (CPP) are acute hallmark events of SAH. Because of the sudden nature of the injury, in patients, these hallmark events are mostly observed during rebleed. A major advantage of the endovascular perforation model is that it replicates all acute hallmark events of SAH.
Allows real-time recording of early physiological changes after SAH: This helps determine the intensity of hemorrhage and prevents pooling data from animals with different SAH intensity, which might lead to large standard deviations, making results difficult to accurately analyze. As described above, in the endovascular perforation model, catheters placed in the cisterna magna measure ICP (Figs. 1b and 2a); laser Doppler flowmeters placed on the temporal side of the skull, away from the large vessels, measure changes in CBF; and catheters placed in the femoral artery measure changes in MABP (for review see [1, 8]) after SAH.
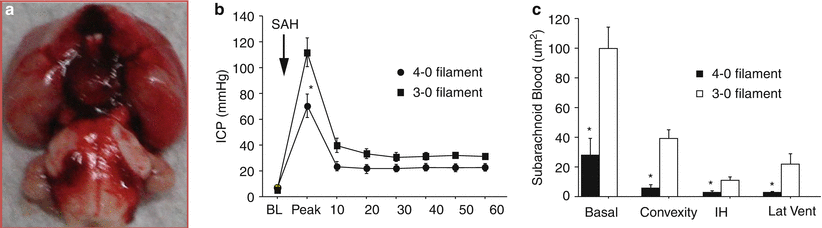

Fig. 2
Controlling and establishing severity of SAH. (a) Representative image showing blood accumulation in brain after SAH. In (b) and (c), note that filament size can be manipulated to create SAH of different severities; ICP rise and blood accumulation are greater when the ICA is perforated by 3’0 as compared with 4’0 suture. Data is from five animals per filament group. * Significantly different at P <0.05
Replicates mechanical trauma: Flooding of subarachnoid space with blood after aneurysm rupture exposes the brain and cerebral vasculature to mechanical trauma. Blood compresses adjacent cerebral arteries, stretches subarachnoid space, and obstructs the passage of CSF. Because SAH in the endovascular perforation model is induced by rupturing/perforating an intracranical artery (see above), it replicates the mechanical trauma experienced by the brain tissue and cerebral vasculature during SAH because blood is released under pressure and pools in basal cisterns [10].
Limitations
SAH surgery: A major limitation of the endovascular perforation model is that it requires complicated and extensive surgery. Hence, extensive hands-on training is required to master the procedure. Animal mortality in this model is as high as in SAH patients (45 % within the first 48 h).
Hemorrhage severity: Another major limitation is the inability to control the amount of bleeding and hemorrhagic severity, which, as explained above, can lead to wide standard deviations, making it difficult to draw firm conclusions when evaluating the effects of therapeutic agents [7, 8]. One reason underlying this limitation is that the severity of the bleed depends on the speed and force used to perforate the artery, which can vary from surgery to surgery and from surgeon to surgeon. However, with training, a surgeon’s hand gets settled and he/she can begin to produce SAHs that have ICP rises of similar intensity.
Lack of proper sham control: The sham control in the endovascular perforation model is an animal that undergoes all the procedures that are performed in a SAH animal, including introducing a monofilament into the ICA, except for the perforation [8]. What makes this sham a poor control is that, unlike the sham control in an injection model, the sham control in this case does not experience any change in physiology.
Technical considerations
A number of considerations can help a surgeon master the endovascular perforation SAH model and reduce acute deaths. Some are as follows:
Acute death: The rapid rise in ICP at the time of SAH can increase the pressure at the respiratory centers to the point that the animal may stop breathing spontaneously. Use of a respiratory support, such as intubation or placement of a nose cone, ensures that the animal continues to breathe and does not die as SAH is induced [9].
Injury to brain tissue: Brain injury may occur because of the stabbing of the brain tissue by the filament used to perforate the artery. This injury occurs if the surgeon continues to advance the filament after the artery is perforated and can be avoided if a conscious attempt is made to retract the endovascular filament as soon as the artery is perforated. The event of perforation is felt by the surgeon and is confirmed by the rise in ICP; hence a quick look at the real-time ICP recording will confirm that artery is perforated and that the endovascular filament needs be retracted.
Controlling severity of SAH: Several methods to control the severity of SAH have been attempted. Veelken et al. [15] tried occluding the ipsilateral common carotid artery or left the endovascular filament in place to control SAH severity [15]. This mechanism however, hinders normal perfusion through the ipsilateral ICA, and creates a superimposed regional ischemia. Schwartz et al. manipulated the size of the monofilament to control the size of the bleed and the SAH severity [7]. They succeeded in creating smaller bleeds and smaller ICP rises (a low-intensity SAH) with 4’0 instead of 3’0 suture (Fig. 2). We frequently use an unsharpened, blunt 3’0 suture to create a low-intensity (≤40 mmHg) and a sharpened 3’0 suture to create a high-intensity SAH (≥40 mmHg) in rats.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





