CHAPTER 48 REHABILITATION AFTER STROKE
Stroke, or vascular brain injury, has an incidence of between about 1.1 and 2.6 per 1000 adults1 and a prevalence of 5 to 8 per 1000.2 It is a leading cause of chronic disability worldwide; in the United Kingdom, it is the commonest single cause of severe disability in people living at home.3,4 About 75% of affected patients are older than 65 years, and 10% are younger than 55. Each year in England and Wales, stroke occurs in about 10,000 people younger than 55 and 1000 people younger than 30. Six to 12 months after stroke, only 60% of patients with hemiplegic stroke have achieved independence in personal care, and those with sensory or visual field loss are more disabled; 30% to 40% of survivors are depressed, 10% to 15% severely so; 50% need help with either housework, meal preparation, or shopping; and a similar percentage lack a meaningful social, recreational, or occupational activity during the day.5–8 In addition to the personal consequences, the economic consequences of stroke are also enormous; direct health care costs account for approximately 4% of total health care costs, or about £3 billion in the United Kingdom (about $5.3 billion in U.S. dollars) per annum at 2005 prices9; these costs will increase as the population ages.
Neuroprotective interventions to reverse the immediate consequences of stroke are not widely applicable.10 In these circumstances, when reversal of pathology is incomplete, the majority of stroke survivors need the multidimensional process termed rehabilitation to enhance their functional activity and societal participation and to reduce the effect of limitations in these areas, so that life quality is improved and life is “worth living.”11 Rehabilitation goals are achieved through the prevention of secondary complications; through facilitation of neural protection, restoration, and substitution; and through functional compensation, which involves both behavioral adaptation and substitution, as well as modification of personal, environmental, and social contextual factors (Table 48-1). This process must also be accompanied by often difficult and less frequently admitted adjustment to loss and change, a need that is easily forgotten in the gym or during a functional imaging study. This chapter explores the delivery and effectiveness of these interventions during rehabilitation after stroke.
TABLE 48-1 Mechanisms of Recovery after Stroke
The high incidence and prevalence of stroke have provided an opportunity to test the effectiveness of many components of the rehabilitation process. Trials have shown convincingly that organized care produces better outcomes than disorganized care; this result has far-reaching implications for health care systems in general, let alone those focusing on stroke. Many of the areas addressed in this chapter are included in evidence- and expert opinion–based guidelines produced in the United Kingdom by the Royal College of Physicians12 and the Scottish Intercollegiate Guidelines Network,13 in Australia by the National Stroke Foundation,14 and in the United States by the Department of Veterans Affairs and the Department of Defense.15
COMPLEX ORGANIZATIONAL INTERVENTIONS
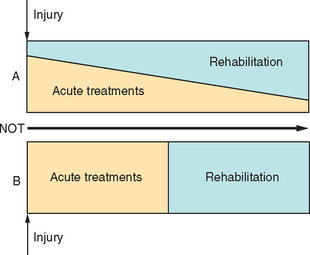
Rehabilitation after single-incident brain injury, including stroke, should be delivered through polymodal inpatient and community-based service delivery systems that differ by structure and process. A classification of these different service options, as a “rehabilitation typology,” remains to be agreed upon, at least cross-culturally, and would facilitate comparisons of different studies.16 The use of each component is determined by a variety of clinical and social factors, including length of time since injury, level of dependency, characteristics of the residual impairment, age of the patient, and resources available.17 Recommendations that different types of care after stroke are incorporated into systems of clinical care (e.g., Schwamm et al18) also emphasize the use of acute and rehabilitation interventions in parallel rather than in series (Fig. 48-1), so that strategies optimizing neural protection acutely are combined with key factors identified in postacute care. The benefits of organized complex polymodal interventions so far reported are likely to reflect mainly the effects of preventing the complications described later in this chapter and shown in Table 48-2, functional interventions that focus on teaching new skills, and the use of aids and appliances and environmental modifications to help patients adapt to their impairments, rather than the effects of neural reorganization.
TABLE 48-2 Complications after Stroke during Inpatient Rehabilitation and Their Management
| Cause and Effect | Stroke Unit Frequency % (95% Confidence Interval) | Management |
|---|---|---|
| Comorbid Conditions | ||
| Hypertension/hypotension | 15/8199 | Antihypertensive/treat cause |
| Cardiac events | 13199 | Specific medical treatments |
| Fever | 20199 | Antipyretics |
| GI bleed | 3196/4199 | ? Prophylaxis; avoid NSAIDs |
| Drug side effects | 9198 | Drug withdrawal |
| Musculoskeletal pain | 14198/38197 | Physical therapies |
| Physical Dependency | ||
| Pressure sores | 4198 | Pressure care, 24-hr handling and positioning |
| Sores and skin breaks | 21 (16-25)195/18196 | |
| Contractures | ||
| PEs and DVTs | 3 (0-3)195/5198 | Thromboprophylaxis |
| Falls | 25 (21-30)195/20256/11198 | Risk assessment |
| Shoulder pain | 9 (6-12)195/24256 | Handling and positioning |
| Neurological Damage | ||
| Malnutrition and dehydration | 5198 and 10198/10199 | NG/PEG feeding and dietetics |
| Chest infections | 22 (18-27)195/4199 | Dysphagia management |
| Urinary tract infections | 23 (18-28)195/15256/31198 | Toileting program |
| Confusion and agitation | 36 (30-41)195/4199 | Environmental management ± drugs |
| Epilepsy | 3 (1-5)195/2256/2198 | AEDs if no other trigger |
| Sleep-disordered breathing | 62257 | CPAP |
| Fatigue | 40-60223 | ? Retraining |
| Vascular Brain Injury | ||
| Mass effects | Neurosurgical decompression | |
| Hydrocephalus | Shunting | |
| Catastrophic illness | ||
| Depression | 16 (12-21)195/26256/13198 | CBT ± drugs |
| Anxiety | 14 (10-18)195/8198 | CBT ± drugs |
| Family breakdown | Information + training | |
AED, antiepileptic drug; CBT, cognitive-behavior therapy; CCF, congestive cardiac failure; CPAP, continuous positive airway pressure; DVT, deep vein thrombosis; GI, gastrointestinal; NG/PEG, nasogastric/percutaneous endoscopic gastrostomy; NSAID, nonsteroidal anti-inflammatory drug; PE, pulmonary embolism.
Until about 1995, 20% to 50% of patients after stroke were managed acutely at home in some European countries,19 despite recommendations to the contrary.20 The benefits of stroke unit care and the gradual introduction of thrombolysis and neuroprotective agents are, alone, likely to reduce this percentage dramatically. Furthermore, patients randomly assigned to receive acute care at home after moderately severe stroke (sufficient to cause persistent neurological deficit affecting continence, mobility, and self-care, and necessitating multidisciplinary treatment), rather than to receive care in an acute and rehabilitation stroke unit or a general ward with stroke team support, were significantly more likely at 1 year to be dead or institutionalized (24% versus 14%; P = 0.03) and less likely to be alive without severe disability (85% versus 71%; P = 0.002) than were patients admitted to the stroke unit.21
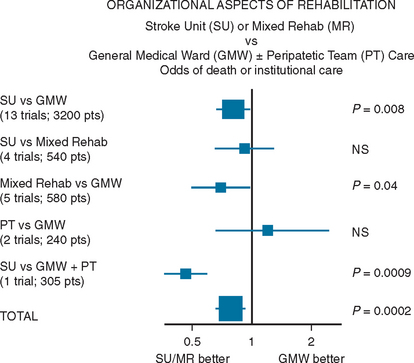
In their meta-analysis of 23 trials, the Stroke Unit Trialists’ Collaboration22 compared care in stroke units, nine of which were “comprehensive” and included both acute and rehabilitation components, with care given by alternative services, usually in a general medical or geriatric ward with or without a visiting stroke team. The meta-analysis showed that patients who receive unit-based inpatient care do not stay longer in hospital and are more likely to be alive, independent, and living at home 1 year after the stroke, regardless of gender, age, and stroke severity (Fig. 48-2). Thus, in comparison with alternative services, stroke unit care produced reductions in the odds of death at 1 year (odds ratio, 0.86; 95% confidence interval, 0.71 to 0.94; P = 0.005), of death or institutionalized care (odds ratio, 0.80; 95% confidence interval, 0.71 to 0.90; P = 0.0002), and of death or dependency (odds ratio, 0.78; 95% confidence interval, 0.68 to 0.89; P = 0.0003).
A subsequent systematic review of results from nine postacute units, which admitted patients more than 1 week after stroke, in comparison with an alternative service, revealed benefits similar to those resulting from combined acute and postacute units,23 and in the United States, compliance with postacute but not acute stroke unit guidelines (Table 48-3) has been shown to be correlated with outcome.16 Secondary analysis has indicated that the reduction in stroke unit deaths probably results from a reduction in the complications of immobility rather than in neurological or cardiovascular complications. The increased number of patients discharged home from stroke units and the reduced requirement for institutional care were attributable largely to an increase in the number of patients returning home physically independent (Rankin score, 0 to 2), rather than dependent (Rankin score, 3 to 5).24
TABLE 48-3 Dimensions of Acute Rehabilitation after Stroke
Compliance with these dimensions of process during postacute rehabilitation after stroke is correlated with outcome.
After Duncan PW, Horner RD, Reker DM, et al: Adherence to postacute guidelines is associated with functional recovery in stroke. Stroke 2002; 33:167-178.
Langhorne and Pollock25 identified aspects of care common to 11 stroke units, 8 of which included acute care, reporting beneficial effects between 1985 and 2000 in the Stroke Unit Trialists’ Collaboration’s systematic review.22 These included comprehensive medical, nursing, and therapy assessments; integration of nursing care within the multidisciplinary team; early mobilization and treatment of hypoxia, hyperglycemia, and suspected infection; avoidance of urinary catheterization; and formalized goal-oriented multidisciplinary team care, with early discharge planning and education and involvement of caretakers.
Evans and associates26 found that many of these aspects of care were not delivered by a peripatetic specialist stroke team (PSST). They compared care delivered to 304 patients randomly assigned to an acute and rehabilitation stroke unit (n = 152) or to general wards supported by a PSST (n = 152). Patients in the stroke unit were monitored more frequently, and more of those patients received oxygen, antipyretics, measures to reduce aspiration, and early nutrition than those in general wards. Many aspects of multidisciplinary care occurred more frequently in the stroke unit, in which complications including stroke progression, chest infections, and dehydration were less frequent. Early feeding, stroke unit management, frequency of complications, and measures to prevent aspiration independently affected outcome. In 1984, a PSST’s input to general ward care was shown to nonsignificantly reduce mortality rates and significantly improve functional recovery in men.27 More recently, in a head-to-head comparison at 1 year of patients randomly assigned to receive care from the PSST with those receiving care in a combined acute and rehabilitation stroke unit, more patients in the PSST group were dead or institutionalized (30% versus 14%; P < 0.001) and fewer were alive without severe disability (66% versus 85%; P < 0.001).21 Retrospective data collected by van der Walt and colleagues28 before and 2 years after the introduction of a PSST to a general ward showed significant improvements in prophylaxis for deep vein thrombosis, incontinence management, premorbid function documentation, frequent neurological observations, and early occupational therapy, with fewer severe complications (9% after versus 24% before; P = 0.004), reduced median length of stay (12.0 after versus 18.5 days before; P = 0.003), and more patients independent at discharge (32% after versus 9% before; P < 0.001). It is thus possible that practice developed in stroke units can now be delivered more effectively by a PSST, and further comparison of these two methods of service delivery remains legitimate.
More recently, high-dependency care has been introduced immediately after stroke, providing continuous rather than manual monitoring for hypoxia, hyperglycemia, hypotension, cardiac arrhythmias, and elevated body temperature during the first 48 to 72 hours after admission.29,30 Currently, its effectiveness is relatively unexplored, but there is a suggestion that it may reduce mortality rates at 3 months and 1 year among patients with severe stroke, without increasing dependency.31,32
Remarkably, the differences between stroke unit and alternative care persist for many years. Indredavik and colleagues33 found that even 10 years after random assignment to an acute and rehabilitation stroke unit or a general ward, fewer patients in the stroke unit had died (75.5% versus 87.3%; P = 0.008), more were at home (19.1% versus 8.2%; P = 0.018), and more were at least partly independent with a Barthel Index score of 60 or higher (20.0% versus 8.2%; P = 0.012) or independent with a Barthel Index score of 95 or higher (12.7% versus 5.4%; P = 0.061). Increased survival times 5 years after random assignment to a stroke rehabilitation unit, versus a general medical or geriatric ward, were also reported by Lincoln and colleagues.34
Early discharge, supported by a multidisciplinary outreach team, of medically stable patients after mild and moderate stroke, with an admission Barthel Index score of more than 9, supplements initial stroke unit gains. A meta-analysis of individual patient data from 11 trials of early supported discharge versus conventional care showed that the patients with early supported discharge had a reduced risk of death or dependency (odds ratio, 0.79; 95% confidence interval, 0.64 to 0.97; P = 0.02), a hospital stay shortened by 8 days (P < 0.0001), and significant improvement in extended activities of daily living (P = 0.05), although not in subjective health status or mood in either patients or caretakers. Patients were moderately disabled at discharge with a median discharge Barthel Index score of 15.35 One study revealed that these gains can include better life quality, assessed by the Nottingham Health Profile, at 1 year,36 and another study revealed that gains in domestic and extended activities of daily living are still evident after 5 years.37
Once in the community, patients are known to be at risk of deteriorating as a result of multiple health problems, including falls, depression, and physical and social inactivity and isolation, in addition to age-related symptoms and comorbidity, and health-related quality of life has been shown to significantly decline in the 6 months after discharge.38 Nursing home care does not substitute for stroke unit care,39 but the place for and the optimal process in other service systems for patients later after stroke (e.g., nurse-led wards, nursing homes, or residential placements) remain to be examined.
A meta-analysis of trials of therapy-based outpatient or domiciliary rehabilitation, delivered by either a multidisciplinary team or by a physiotherapist or occupational therapist, with the goal of improving task-oriented behaviors, has shown that deterioration is prevented (odds ratio, 0.72; 95% confidence interval, 0.57 to 0.92; P = 0.009) and dependency in personal care reduced (95% confidence interval, 0.02 to 0.25; P = 0.02).40 The effective components and best location for this type of service need further exploration, but benefit has been consistent in trials of community-based occupational therapy, which have provided sufficient data for a meta-analysis of eight trials that showed that intervention was associated with improved personal, extended, and leisure-based activities of daily living, depending on the intervention target.41 These findings were confirmed in a more wide-ranging systematic review of occupational therapy for stroke patients by Steultjens and associates,42 who also noted the need for further studies of the effectiveness of splinting.
Most studies of physiotherapy in patients in the community after stroke investigate the effect of a particular physiotherapy treatment on upper or lower limb function at the level of impairment and mobility, which may improve, rather than on limitations in activity and independence, which, if examined, may not improve. Domiciliary physiotherapy within 6 months after stroke has been shown to reduce probability of readmission after an average of only 2.9 (range, 1 to 8) visits43 and to reduce dependency at less cost than for day hospital attendance.44,45 In patients more than 1 year after stroke, only four to five physiotherapy sessions produced a clinically small but significant improvement in mobility.46
Informal caretakers should be recognized as an important resource: They enable patients to remain in the community,47 their support is likely to facilitate patient outcomes,48 and depression is more severe in caretakers who feel poorly supported.49 Formal support for caretakers is difficult to obtain in the United Kingdom. Trials of psychosocial interventions to support caretakers of patients with stroke, involving information packages, specialist nurses, a mental health worker, or family support workers, have failed to show functional or psychological benefit in patients and only modest psychosocial benefit for caretakers.50–57 In contrast, there is evidence that caretaker adjustment is increased by education and counseling or by training in social problem-solving skills.58 Kalra and colleagues59,60 demonstrated that training informal caretakers in basic nursing skills and facilitation of personal care techniques reduced costs and caregiver burden and improved psychosocial outcomes for the caretaker and the patient, although there was still no change in patients’ rates of mortality, institutionalization, and disability.
MANAGEMENT OF NEUROLOGICAL IMPAIRMENTS
Evidence of dysphagia, with consequent risk of dehydration, further malnutrition, and chest infections resulting from aspiration, has been reported to occur clinically in about 50% of all patients with stroke admitted to a hospital, with videofluoroscopic evidence of a swallowing abnormality in up to 65% of patients and of aspiration in about 20%.61,62 Videofluoroscopic and flexible endoscopic evaluation of swallowing increase the reliability with which aspiration can be identified63,64 when clinicians select which patients need tube feeding. However, whether other aspects of dysphagia management, including dietary modification and compensatory swallowing techniques, reduce the need for tube feeding or the risk of aspiration pneumonia remains unclear despite assertion to the contrary.65 The Feed or Ordinary Diet (FOOD) trials66 revealed that in 859 patients randomly assigned to receive nasogastric feeding within a week, in contrast to more than a week, after stroke, absolute mortality rates were reduced by 5.8% (95% confidence interval, −0.8 to 12.5; P = 0.09) in the group fed early; in 321 patients randomly assigned to undergo percutaneous enteral gastrostomy (PEG) feeding or nasogastric tube feeding a median of 1 week after stroke, PEG feeding was associated with an absolute increase in risk of death of 1.0% (95% confidence interval, −10.0 to 11.9; P = 0.9) and an increased risk of death or other poor outcome of 7.8% (95% confidence interval, 0.0 to 15.5; P = 0.05). Thus, as is recommended current practice in hospitalized adult patients in general,67 nasogastric feeding should be used for dysphagic patients soon after stroke, whereas PEG feeding is reserved for patients who do not tolerate nasogastric feeding and as required in the longer-term care of dysphagic patients.
After stroke, early incontinence is predictive of poor outcomes, including death, lengthy hospital stay, institutionalization, and severe disability.68 New urinary incontinence occurs in 40% to 50% of patients during the first week after admission and in 10% to 20% by 6-month follow-up68,69; it appears to remain at this level in the community over the long term.70 The proportions of stroke patients with new fecal incontinence on admission and at 6-month follow-up are lower, at 30% to 40% and 5% to 10%, respectively.68,69 Causes and predictors68,71,72 of fecal and urinary incontinence include age; stroke type and severity; “functional incontinence” resulting from stroke-related cognitive and communication difficulties and mobility problems; autonomic neuropathies, usually diabetic in origin; medications causing bladder and bowel hyporeflexia; preexisting bladder outflow obstruction in men or stress incontinence in women; and perhaps depression and lowered self-esteem that result from the incontinence itself.73,74
Current guidelines (e.g., Royal College of Physicians12) for the management of incontinence after stroke recommend initial definition of its cause and associations through a full clinical assessment. The neurogenic cause after stroke is usually disruption of suprapontine inhibition of bladder contractility resulting in detrusor hyperreflexia, but bladder hyporeflexia may occur after cerebellar stroke.75 Evidence for the effectiveness of methods promoting continence after stroke is currently derived largely from trials in other (nonstroke) patient groups. Management of urinary incontinence76 includes a combination of anticholinergic drug therapy, bladder training by prompted and/or timed voiding77,78 or biofeedback,79 and appropriate management of outflow obstruction and stress incontinence, for which one small study of 26 stroke patients randomly assigned to receive treatment or to control conditions showed no benefit from a 12-week pelvic floor muscle training program.80 Similar general principles apply to the management of fecal incontinence,81 which in inpatients or in discharged patients is associated with needing help to use the toilet82 and thus might be addressed by improving functional independence, whereas constipation but not fecal incontinence after stroke is improved by a single clinical/educational nurse intervention up to 6 months later.83
Impairment of trunk control and sitting balance after acute stroke is common, and recovery is a reliable predictor of functional outcome after stroke. Trunk control, measured by the trunk control test at rehabilitation admission a median of 5 weeks after stroke, accounted for 71% of the variance of the motor component of the Functional Independence Measure at discharge 10 weeks after stroke84; as measured by the Postural Assessment Scale for Stroke Patients within 2 weeks after stroke, trunk control was strongly correlated with functional outcome, rated by a combined score of both personal (Barthel Index) and instrumental (Frenchay Activities Index) activities of daily living at 6 months after stroke.85 A positive correlation has also been found between sitting balance and the Barthel Index score86 and between sitting balance and gait at 6 months.87 Mudie and associates88 found that three specific treatment approaches implemented early after stroke improved sitting balance in the short term in comparison with a nonspecific control approach. Whether early targeting of sitting balance for treatment could improve mobility over the long term is an interesting question that remains to be explored.
Disordered motor control of the limbs after stroke is usually caused by the positive and negative effects of the upper motor neuron syndrome, in which the central abnormality is usually muscle weakness and co-contraction resulting from a failure of coordinated high-frequency motor neuron firing during movement, rather than high tone resulting from spasticity and contracture found at rest,89 which is seen clinically in only 20% to 30% of hemiparetic patients 3 to 12 months after stroke.90,91 Spasticity at rest, when it is a velocity-dependent increase in tonic stretch reflexes with exaggerated tendon jerks, is of diagnostic use but does not contribute significantly to impairment during active voluntary movement and function in either the leg or the arm92,93 and is only one factor contributing to increased muscle tone in the upper motor neuron syndrome, in which co-contraction and biomechanical changes contribute significantly to the resistance to passive movement.94 It has thus been easier to show that pharmacological treatments that reduce the hypertonus of spasticity and co-contraction benefit passive rather than active functional activities, including hand and perineal hygiene, dressing, pain, and limb position,95–100 and even correction of equinovarus at the ankle to improve weight bearing by the affected leg is unlikely to translate into functional improvements in gait101,102 unless it is followed by physical measures to reduce soft tissue shortening and a program of task-related training.103,104
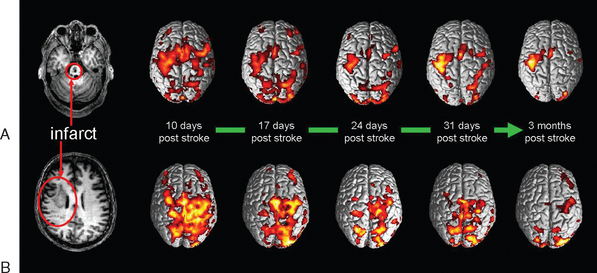
Thus, although treatments for spasticity after stroke achieve passive functional goals by preventing contracture or reduce pain and are of particular value in dependent patients unable to participate in skill learning, the results of treatments for spasticity in general and after stroke have emphasized the fundamental truth of Landau’s statement in 1974105 that “If the major disability of the upper motor neuron syndrome is due to diminished neural input to segmental control of the final common path (negative symptoms), it follows that only some method of provoking [central nervous system] neuronal regeneration or of improving the potency of and control by surviving upper level neurons could provide a direct remedy.” Landau’s next sentence was “No such approach is on the horizon,” and at that time the focus in rehabilitation was on training in compensatory strategies and social and environmental modification. Since the mid-1990s, however, there have been investigations of neurobiological rehabilitation strategies designed to modify the neural processes and behaviors that occur during recovery (Fig. 48-3) (Dobkin, 2004).106
Meta-analyses of physiotherapy after stroke have revealed significant improvements in independence in activities of daily living and reduction in impairments with higher intensities of physiotherapy.107–109 Traditional physiotherapeutic strategies emphasize cutaneous and proprioceptive stimulation, reduction of tone, and therapist-induced central facilitation of normal movement, rather than voluntary activation of proximal or distal musculature to relearn functional tasks. This type of task-related motor relearning was first shown to be effective in improving the ability to balance during seated reaching activities late after stroke.110 Although this approach has not yet been consistently shown to improve functional outcomes and resource use early after stroke,111,112 its principles of repetition, task orientation, attention, and reward are similar to those that result in motor learning and cortical neural reorganization in nonhuman primates with and without ischemic cortical damage.113 It is thus a “neurobiological” approach to skill relearning in which repetition of graded and motivating task-related learning programs is envisaged to drive the acquisition of motor, linguistic, and cognitive skills and more complex behaviors through activity-dependent neural reorganization, regrowth, and replacement.114
Although these techniques can be shown to produce neuroplastic effects—in humans, usually by functional imaging techniques—these changes should not be confused with the potential clinical usefulness of these techniques, which requires a robust behavioral effect if measurable functional benefit is to occur. The effects of task-related training are seen most obviously in the function targeted for treatment; change has been demonstrated after stroke in upper and lower limb function and linguistic skills. Factors that optimize skill learning, including implicit rather than explicit feedback115 and a random rather than blocked learning schedule,116 are likely to affect the rate at which a task is learned and the extent of carryover into novel environments. In the arm, training is focused on practicing graded motivating functional tasks, including grasping, reaching, leaning, and manipulating clothing by the affected limb; in the trunk and leg, training is focused on the recovery of bed mobility, sitting and standing balance, transfers, gait stability and velocity, and stair climbing. Kwakkel and colleagues117 showed that leg training of this sort for an average of 30 minutes of therapy 5 days a week for 20 weeks after stroke onset, in contrast to immobilization of the limb in an inflatable cuff for a similar time, increased performance in activities of daily living (measured by the Barthel Index), walking ability, and also hand dexterity, whereas arm training increased arm dexterity alone.
What determines when, in whom, and how task-oriented training should be carried out after stroke, in an individual as well as in a population, remains open to investigation. The duration in training can be increased: for example, to an impractical 6 hours a day for 10 weekdays late after stroke. Training can be performed solely with a therapist or also automated with a robotic,118,119 most commonly as a treadmill with or without body weight support for gait training120 or in a virtual environment,121 or it can be encouraged, probably at both cortical122 and functional levels, and also in animals,123 by constraint of the unaffected upper limb.124–126 It can also be combined with a variety of impairment-based adjunctive therapies, including muscle-strengthening exercises,127 an orthotic,128 electrical stimulation of the arm or leg,129–131 sensory stimulation,132 mental imagery,133 and musical feedback.134
The available evidence for the effectiveness of these and other physical therapies was systematically reviewed in 2004 by van Peppen and colleagues.135 Which therapy modalities should be used, to what purpose, how they should be combined, at what stage after stroke, at what intensity and for how long, and in which patients are questions that are beginning to be answered clinically with an evidence base, rather than at best theory and at worst belief. The need to be able to do this is emphasized particularly by the evidence beginning to show long-term persistence of task-related training effects136,137 and the useful effects of interventions late after stroke (e.g., Werner and Kessler138 and van der Lee et al139). It is accompanied by a need to explore how the new technologies, which may not always yield benefit (e.g., Richards et al140), can best be put best to work.
Similar principles are beginning to drive trials of rehabilitation therapies of the focal, rather than global,141 cognitive disorders observed after stroke. According to results of clinical measures, language impairments affect about 40% of the stroke population at stroke onset142 and about 10% at 6 months after stroke.143 Minimal spontaneous improvement in language functions occur more than 1 year after stroke. An early meta-analysis144 and review145 of trials of speech and language therapies concluded that there was evidence of effectiveness, but restriction of the evidence to randomized controlled trials, in comparison with no therapy, informal support, or another speech and language therapy, failed to yield clear evidence of effectiveness.146 However, Bhogal and colleagues147 found that increased intensity of therapy was associated with improvement, which occurred in four studies that provided an average of 9 hours of therapy per week for 11 weeks, in contrast to four studies with negative results that provided approximately 2 hours per week for 23 weeks. Although the type of therapy required remains unclear (e.g., Doesborgh et al148), Pulvermüller and colleagues149 suggested that the principles of (a) massed practice—for example, 30 to 35 hours of therapy over 10 days rather than the same therapy time over a longer period—and of (b) constraint (in this case of maladaptive methods of communication), and of (c) everyday behavioral relevance are important factors in generating neuroplastic and thus functional changes in language skills.
The presence of other cognitive problems in the acute phase are important independent predictors of long-term outcome, in addition to neurological deficits rated by standard measures, particularly unilateral neglect150,151 and executive dysfunction.152 Neglect, as currently measured, is seen in more than 60% of patients assessed within 2 to 3 days of admission for stroke, more commonly after right than left hemisphere stroke.153 Of patients with neglect 2 to 4 weeks after stroke, 80% still exhibit aspects of neglect on testing at 6 months,154 and the poor functional outcome predicted by the presence of early neglect is still seen if neglect has resolved on testing,151 possibly as a result of residual problems with nonlateralized attention.155 Neglect, like dysphasia, is heterogeneous; different combinations of lateralized and nonlateralized attentional and motor deficits are identifiable in each patient. A number of techniques have been shown to improve lateralized spatial representation, including caloric stimulation156,157; contralesional neck muscle vibration, in one trial with scanning training158; prism adaptation159; and contralesional limb activation,160 as well as alerting techniques to increase nonlateralized loss of attentional capacity.161 Some of these techniques have also been reported to improve activities of daily living, notably neck muscle vibration with scanning training158 and prism adaptation,162 and contralesional limb activation in one study reduced length of stay by 24 days.150 Whether a treatment battery tailored to the neglect subtypes present in each patient163,164 will prove sufficiently practical and robust to produce significant functional benefits, not yet evident in the literature on systematic review,165 remains to be explored.
In the context of stroke, use-dependent neural reorganization after damage is facilitated by other extrinsic and intrinsic inputs that modulate neural excitability and augment long-term synaptic potentiation. Use-dependent network remodeling at cellular and systems levels is also likely to be needed for the successful application of techniques to promote neural regrowth and/or replacement.166,167 Adjunctive facilitators of this process include manipulation of attention168; increase or decrease of somatosensory input, shown to improve swallowing169 and leg170 and arm122 function; neuromodulatory drugs171; electrical172 and magnetic173 stimulation of the brain; and possibly exercise.174
Pharmacological facilitation and inhibition of neurological recovery after stroke are also likely to be of clinical relevance. The use-dependent plasticity that underpins motor training and perceptual learning is dependent on N-methyl-D-aspartate receptor activation and γ-amino butyric acid–mediated (GABAergic) inhibition,175,176 and these are in turn modulated by cholinergic, noradrenergic, serotonergic, and dopaminergic transmitter systems.171 Early experiments by Feeney and coworkers177 in rats showed that motor recovery after suction ablation of the sensorimotor cortex was improved by amphetamine before practice on a beam-walking task; neither practice nor amphetamine alone had any benefit. Subsequently, α2 antagonists, increasing noradrenergic effects, were found to facilitate motor recovery, and α1 antagonists and α2 agonists, which decrease norepinephrine release or block its postsynaptic effects, reinstate deficit in clinically recovered animals or slow their recovery.178,179 There is also evidence that modulation of other neurotransmitter systems has similar effects.180 The beneficial effect of these pharmacological interventions is dose dependent, and the timing of administration is likely to be important, the effect being dependent on close temporal linkage to behavioral experience. In animals, there is a strong correlation between the functional effects of altering levels of various neurotransmitters and their ability to facilitate increases in synaptic efficacy through long term potentiation.180
Attempts have been made to translate these findings into treatments for stroke patients. In the first clinical trial of noradrenergic enhancement coupled with physical therapy, patients who had suffered from hemiplegic stroke were randomly assigned to receive either 10 mg of dextroamphetamine or placebo 45 minutes before physiotherapy.181 Follow-up assessment 24 hours after treatment revealed a 40% improvement from baseline scores with dextroamphetamine in comparison with placebo. Small numbers of patients in this study made interpretation difficult. A subsequent failure to replicate these findings by Borucki and associates182 was attributed to a different experimental design, particularly a failure to schedule physiotherapy immediately after amphetamine treatment. More recent studies with dextroamphetamine have had conflicting results,183,184 but positive results have been published for daily doses of levodopa (100 mg) used in conjunction with physiotherapy.185 Studies of the effects of drugs used in conjunction with speech therapy have yielded some success.186 Although larger scale studies are under way, the use of pharmacological agents to facilitate the effects of physical and speech therapy is currently only experimental. Conversely, there is some evidence to suggest that patients receiving drugs that reduce the effects of norepinephrine or dopamine, or increase the effects of GABA, at the time of stroke or shortly afterward have poorer outcomes than do patients who do not receive any of these drugs187; thus, care must be taken in their prescription.
Maladaptive or ineffective neural reorganization after stroke may contribute to emotionalism, central poststroke pain (CPSP), and involuntary movement disorders, which include hemiballismus, hemichoreoathetosis, distal resting and/or action tremor, and proximal postural tremor. CPSP, originally described in 1906 as a phenomenon after thalamic stroke by Dejerine and Roussy,188 was found in 16 (8%) of 207 hospitalized stroke patients 6 months after stroke by Andersen and colleagues.48,49 Pain may result from stroke involving spinothalamocortical afferent pathways in the medulla, pons, midbrain, thalamus, subcortical white matter, and cortex. Leijon and colleagues189 found in 27 patients with CPSP that the thalamus was involved in only 9, lesions were in the lower brainstem in 8 and lateral and superior to the thalamus in 6, and onset of the pain was delayed by more than a month in 13 patients. The pain was described as “burning, aching, pricking, and lacerating”; the intensity was increased by external stimuli, either noxious (hyperalgesia) or not noxious (allodynia), the most common being joint movements, cold and light touch, and, in five patients, emotional stimuli. All patients had decreased temperature sensation. Hypersensitivity to cutaneous stimuli was found in 88% of the patients, and sensations of touch and vibration were abnormal in only 52% and 41%, respectively. There were also relatively low incidences of weakness and ataxia, which were present in 48% and 62%, respectively. Treatments in individual patients include all the drugs and physical methods used for neuropathic pain in general; descending modulation of pain pathways by electrical motor cortex stimulation,190 rather than deep brain stimulation,191 and more recently repetitive TMS over the hand area of the motor cortex192 have shown some promise in the treatment of CPSP. Both deep brain stimulation and electrical motor cortex stimulation have been used with some success to treat poststroke movement disorders,191 whereas antidepressants are effective in treating emotionalism.192–194