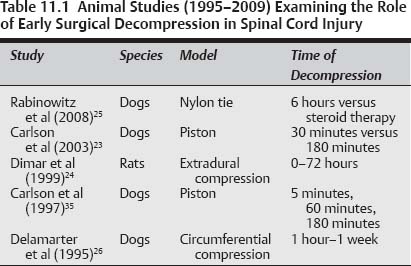
11 The role of early surgical decompression following acute spinal cord injury (SCI) is embedded in an evolving understanding of the pathophysiology of this devastating condition. Specialized SCI treatment centers date back to the mid-1900s, and despite advances in biomedical knowledge regarding the pathobiology of SCI, the prognosis for neurological recovery in severe SCI is poor. The personal and familial suffering related to impaired ambulation, sensation, and bowel, bladder, and sexual function is incalculable. To make matters worse, of the ~10 to 12,000 patients that suffer SCI each year in the United States, most are young and contribute to the economy in a meaningful way.1 The goals of treating a patient with acute SCI can be summarized as follows: preservation of life, preservation and maximization of neurological function, and provision of a stable and painless vertebral column.2 The first of these three goals involves acute life-saving measures that begin at the scene of the traumatic event and are carried through to the acute care facility as mandated by the Advanced Trauma Life Support (ATLS) protocol. Only after airway and hemo-dynamic stability is maintained can attention be turned to neurological function. In fact, hemodynamic instability and hypotension are important contributors of secondary SCI. Following medical stabilization, the clinical team can focus on preserving and maximizing neurological function and subsequently ensuring a stable and painless vertebral column. This chapter briefly reviews the pathophysiology of SCI as well as the definition of timing with regard to surgical decompression. Following this we will outline the role for conservative treatment in SCI along with the available evidence. The remainder of the chapter provides a systematic review of the literature with regard to the role of early surgical decompression following SCI. We break the evidence into levels I, II, and III and highlight both animal and human studies. Finally, we conclude with recommendations based on our evolving understanding of the pathobiology and the available evidence. To understand how one might intervene to preserve and maximize neurological function one must understand the pathophysiology that occurs following injury. Primary SCI refers to the initial physical deformation forces that may result in the severing of axons and subsequent cellular death. Secondary injury is more complicated and refers to the cellular mechanisms that occur in the minutes to weeks to years following the initial injury. Briefly, these mechanisms include ischemia, vasospasm, delayed axonal loss, apoptosis, ion-mediated cell damage, excitotoxicity, neuroinflammation, mitochondrial dysfunction, and oxidative cell damage. A thorough understanding of these secondary mechanisms provides a framework for intervention and the possibility of better outcomes by mitigating the destructive effects that follow such cascades. Of particular importance to this chapter is the notion that persistent compression and instability are key contributors to secondary injury and exacerbate ischemia. This provides the key rationale underlying the concept that (1) persisting compression of the cord contributes to secondary injury and (2) earlier decompression affords the injured cord the optimal opportunity for repair and recovery. For a more complete understanding of these secondary mechanisms one should refer to a recent review.3 As our understanding of these mechanisms advanced throughout the 1990s and into the twenty-first century, so too did clinical trials aimed at halting these mechanisms with the proposed benefit of improving outcomes via a “neuroprotective” effect. The National Acute Spinal Cord Injury Studies (NASCIS II and NASCIS III) have shown that high-dose methylprednisolone affords a modest benefit if given within 8 hours of injury in patients with SCI4,5 and have suggested that treatment within 3 hours may be better than treatment initiated 3 to 8 hours after trauma.6 Although the use of methylprednisolone is controversial (as discussed in an earlier chapter) these studies set the framework for future studies of novel neuroprotective agents and highlight the important concept that timing of treatment following injury is important. Alongside clinical trials of neuroprotective therapeutic agents there have been surgical trials of acute decompression and stabilization. The role of early surgical treatment is controversial and firmly embedded in the notion that the surgeon must balance the risks of therapy with the potential benefit of neurological recovery. In the pages to follow we will review the evidence for early surgical treatment in SCI and outline an approach to consider the evidence, why some of the evidence is controversial, and the important considerations that must be taken into account to offer patients with an acute SCI optimal care. Prior to discussing the level of evidence and drawing conclusions from such evidence, it is important to review the definition of “early” surgery because this certainly varies among surgeons. The notion of early surgery stems from an increased understanding of secondary mechanisms of SCI that is largely deduced from animal data. In such animal studies the timing of surgical decompression is most commonly in the range of 8 to 24 hours,7,8 an experimental model that is rarely possible to reproduce in clinical situations where many factors limit this possibility. There are, however, two justifications for early surgery that exist within the clinical literature (with varying degrees of evidence): neurological function (maintenance and potential improvement) and reduction of medical comorbidities (such as respiratory tract infections and decubitus ulcers) by shortening hospital stay. A group of highly specialized spinal surgeons were questioned to determine an appropriate range of timing of early surgery,9 and a range from 8 to 72 hours was considered appropriate. This same group concluded that more clinical research is needed to properly address this important question. Based on the biology of secondary mechanisms in SCI, the Spine Trauma Study Group has operationally defined early intervention as occurring within 24 hours, with the important caveat of avoiding hypotension and hypoxia because each contributes to a cascade of secondary cellular mechanisms that results in progressive tissue damage. This group recognizes that there is unlikely a specific threshold as to when surgical decompression provides clinical benefit but rather a continuous relationship, and the details of this continuous relationship are certainly multifactorial and as yet unknown. For the purposes of this chapter we chose to limit our review to studies that performed surgical decompression within 72 hours. We also chose to disclose the time of decompression along with the reference so the reader would be aware of the outcomes relative to the time of decompression. Some authors have reported10 spontaneous neurological recovery with nonoperative treatment and that anatomical realignment does not provide the benefit of preserving neurological function. Incomplete cervical SCI, studied more recently in 1996,11 revealed that 10% of patients deteriorated neurologically while undergoing conservative treatment. These conflicting views penetrate the literature with varying degrees along the spectrum of complete to incomplete cord injury. One important exception to this is the reduction of bilateral locked facets, of which there is good evidence12 for early reduction to preserve neurological function. All clinical studies that examine the role for nonoperative treatment are noncontrolled and retrospective in nature. Laminectomy was the treatment of choice in these clinical studies, and it was later demonstrated that laminectomy alone provides inadequate decompression13 and may in fact exacerbate underlying instability. The same authors advise that in the setting of traumatic injury, fusion is usually required. Another theme that exists in the literature, from the 1960s to the present, is the fact that spinal surgical techniques have significantly advanced, especially in the past 2 decades, beyond the use of laminectomy alone. One should therefore be cautious in interpreting studies that made use of surgical techniques that would not be considered standard practice today. Early reduction of fractures is supported by several studies that demonstrate both improved neurological function and safety of early closed reduction.14–16 The most compelling evidence supports early reduction of bilateral locked facets in patients with incomplete tetraplegia or worsening neurological function.12,17–20 Tator et al,21 however, warned against the potential complications of closed reduction by reporting an 8.1% rate of neurological decline in a large multicenter study. However, these deficits were often transient and resolved with removal of the traction weights, and the retrospective design did not allow for variables such as sedation. The topic of early surgical decompression for SCI was reviewed by carrying out an OVID-Medline search from 1950 to June 2009 using medical subject headings (MeSH) of “spinal cord injury” and “decompression, surgical” and “trauma” as keywords. All languages were included. In addition, references from these articles and chapters in spine textbooks were reviewed. This review included all surgical intervention that occurred < 72 hours following injury. Review articles were excluded. Evidence from clinical trials was defined as class I (well-designed and well-conducted randomized, controlled trials), class II (prospective cohort studies or controlled studies with well-defined comparison groups), or class III (case series, retrospective reviews, and expert opinion).22 Based on this search strategy we identified 19 animal and 31 human studies. To focus on the most current evidence, we reviewed and analyzed those articles published after 1995, which consisted of four animal studies and 11 human studies. We found that there is no class I evidence to support either the presence or the duration of a therapeutic window to provide the benefit of preserving neurological function. There are, however, both experimental and clinical data that can help guide clinicians in the management of traumatic SCI. A summary of animal studies published after 1995 is presented in Table 11.1 (studies published prior to 1995 are not discussed due to space constraints). We identified 20 animal studies that addressed the question of prolonged compression of the spinal cord and its effect on outcome. Although a detailed review of each of these studies is beyond the scope of this text we aim to highlight three of the most recent studies and demonstrate that the duration of spinal cord compression is proportional to the neurological outcome. Carlson et al23 in 2003 studied spinal cord compression in a dog model with a piston acting to apply force to the spinal cord. They compared outcomes when removing the piston at 30 minutes versus 3 hours and found the latter group performed worse neurologically. Similarly Dimar et al24 used a rat model and with a range of timed extradural compression up to 72 hours and demonstrated that animals with shorter compression times fared better neurologically. In each of these studies the concept of prolonged exposure to secondary injury mechanisms was hypothesized to account for worse neurological outcome, and with removal of the stimulus for these secondary mechanisms came better neurological outcomes. Carlson’s group attempted to quantify this by correlating lesion volume with exposure time and demonstrated a positive relationship. Most recently, Rabinowitz et al25 conducted a randomized prospective study in dogs comparing early surgical decompression (6 hours) with or without methylprednisolone compared with methylprednisolone alone. Using a model originally described by Delamarter et al26 a single surgeon performed a laminectomy at the L4–L5 level (equivalent to the thoracolumbar junction in humans) and circumferentially compressed the dura by 60% with a nylon band. The surgical wound was then closed and the animals lifted from anesthesia (the nylon band was left in situ). The animals were then randomized to one of three groups (methylprednisolone + early decompressive surgery, saline + early decompressive surgery, or methylprednisolone only). Medical therapy with methylprednisolone or saline was initiated 1 hour after the lesion-inducing surgery. Decompressive surgery was performed 6 hours following the initial insult by taking the animals back to the operating room and removing the nylon band. The animals randomized to not receive decompressive surgery had the band in place for the duration of the experiment. The animals were followed clinically and electrophysiologically for 2 weeks at which point they were sacrificed and examined histologically. The authors demonstrated that surgical decompression, with or without methylprednisolone administration, offers greater neurological improvement than the use of methylprednisolone alone. This is an important study that compared two therapies at the forefront of human treatment that have not yet been compared head to head. The authors rightfully comment on the value of such a trial. Pearls • Animal models provide a controlled environment for studying the effects of spinal cord compression on the potential for neurological recovery, • This controlled environment differs from human spinal cord trauma. • Studies show that a shorter duration of spinal cord compression correlates with improved neurological outcome.
Role of Early Surgical Decompression for Spinal Cord Injury
 Pathophysiology
Pathophysiology
 Definition of Timing
Definition of Timing
 Nonoperative Treatment
Nonoperative Treatment
 Quality of Evidence for Nonoperative Treatment
Quality of Evidence for Nonoperative Treatment
 Summary of Literature Review
Summary of Literature Review
 Discussion of Level I, II, and III Evidence
Discussion of Level I, II, and III Evidence
Animal Studies
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree