For a diagnosis of Apathy the patient should fulfil the criteria (A), (B), (C) and (D)
(A) Loss of or diminished motivation in comparison to the patient’s previous level of functioning and which is not consistent with his age or culture. These changes in motivation may be reported by the patient himself or by the observations of others
(B) Presence of at least one symptom in at least two of the three following domains for a period of at least four weeks and present most of the time
Domain B1: Loss of, or diminished, goal-directed behaviour as evidenced by at least one of the following:
Loss of self-initiated behaviour (e.g., starting conversation, doing basic tasks of day-to-day living, seeking social activities, communicating choices)
Loss of environment-stimulated behaviour (e.g., responding to conversation, participating in social activities)
Domain B2: Loss of, or diminished, goal-directed cognitive activity as evidenced by at least one of the following:
Loss of spontaneous ideas and curiosity for routine and new events (i.e., challenging tasks, recent news, social opportunities, personal/family and social affairs)
Loss of environment-stimulated ideas and curiosity for routine and new events (i.e., in the person’s residence, neighbourhood or community)
Domain B3: Loss of, or diminished, emotion as evidenced by at least one of the following:
Loss of spontaneous emotion, observed or self-reported (e.g., subjective feeling of weak or absent emotions, or observation by others of a blunted affect)
Loss of emotional responsiveness to positive or negative stimuli or events (e.g., observer-reports of unchanging affect, or of little emotional reaction to exciting events, personal loss, serious illness, emotion-laden news)
(C) These symptoms (A–B) cause clinically significant impairment in personal, social, occupational or other important areas of functioning
(D) The symptoms (A–B) are not exclusively explained or due to physical disabilities (e.g., blindness and loss of hearing), to motor disabilities, to diminished level of consciousness or to the direct physiological effects of a substance (e.g., drug of abuse, a medication)
In AD, apathy is the most frequently encountered symptom at all stages of the disease (Robert et al. 2005). In the REAL-FR cohort study, the prevalence of apathy and hyperactivity symptoms increased significantly during the 4-year follow-up period, whereas the prevalence of affective and psychotic symptoms did not (Gonfrier et al. 2012). Overall, the prevalence is estimated to be in the order of 60 % of AD outpatients (van Reekum et al. 2005). Several studies have also indicated that apathy explained at least partially the loss of autonomy in daily life activities (Lechowski et al. 2009).
Furthermore, several studies have shown that in addition to the impairment of cognitive performances, neuropsychiatric symptoms are present very early in the AD process. Apathy and depressive symptoms are the most frequent neuropsychiatric symptoms in mild cognitive impairment (MCI) (Feldman et al. 2004; Gabryelewicz et al. 2004; Lopez et al. 2005). In a prospective study on predictive factors for AD, Robert et al. (2006) reported that the presence of mild signs of apathy in MCI patients was cross-sectionally associated with a higher degree of memory impairment. The same study also examined the influence of apathy dimensions on the risk of developing AD in 214 patients with MCI during a 3-year follow-up. Anxiety, depression and apathy assessment were included in the battery of neuropsychiatric tests. After 3 years, the risk of conversion to AD was significantly higher for patients with lack of interest, which is one of the core apathetic symptoms (Gonfrier et al. 2012). It was demonstrated that apathetic but not depressive symptoms are a major risk factor for conversion to dementia in MCI subjects (Vicini Chilovi et al. 2009). In a 2-year follow-up study, the rates of conversion to dementia were 24 % for MCI without depression or apathy, 7.9 % for depressed MCI, 19 % for depressed-apathetic MCI and 60 % for apathetic MCI. It is important to recall that, although apathy and depression have substantial overlap in key symptoms, there are different syndromes with different neurobiological correlates (Tagariello et al. 2009).
Apathy can be also diagnosed in other neurological diseases. For example, a multicenter study aimed at validating the apathy diagnostic criteria in daily clinical practice showed that apathy was present in 55 % of the AD patients, 70 % of the mixed dementia patients, 43 % of the MCI patients, 27 % of the Parkinson’s disease patients, 53 % of the schizophrenic patients and 94 % of the major depressive episode patients (Mulin et al. 2011). Interestingly, apathy is seen with a general prevalence of roughly 60 % in diseases involving directly the cortex areas and, in average, in 40 % of diseases affecting subcortical structures like in most post-stroke situations, for example (van Reekum et al. 2005).
32.2 What Is Known About Its Anatomical Bases?
Alexander (Cummings 1997) first described a series of parallel frontal-subcortical circuits that link the frontal cortex to the striatum, globus pallidus and thalamus. Cummings further developed this concept which greatly helped in understanding the physiology of behaviour and neuropsychiatric changes occurring with lesions involving these circuits.
Five circuits are described and share a common structure. They originate in the prefrontal cortex, project to the striatum, connect to the globus pallidus and substantia nigra and reach the thalamus. The complete circuit is a closed loop with projections from the thalamus back to the prefrontal cortex (Tekin and Cummings 2002).
Motivated behaviour is supported by the anterior cingulate-subcortical circuit which originates from the anterior cingulate cortex in Brodmann’s areas (BA) 24 and 32. The corresponding neurons project to the ventral striatum called “limbic striatum”, then to the ventral pallidum and the substantia nigra before reaching the anterior thalamus. A projection from the anterior thalamus back to the cingulated cortex closes this circuit (Fig. 32.1).
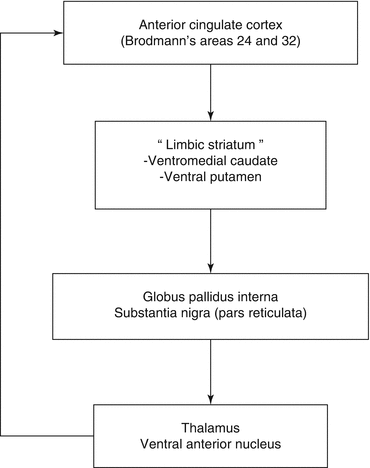

Fig. 32.1
The anterior cingulated-subcortical circuit
Lesions affecting this circuit produce a decrease in motivation also called anterior cingulated syndrome. Infarcts, tumours, hydrocephalus, haemorrhage, encephalitis, degeneration or trauma which involved part of this circuit have been reported in patients producing syndromes called akinetic mutism or abulia which can be considered as extreme forms of apathy. Post-stroke apathy in particular is observed roughly in 35 % of the patients (van Reekum et al. 2005) and is difficult to separate from depression (Hama et al. 2007). Except in rare cases of specific anterior cingulate infarction (Anderson et al. 2003), apathy seems to result more often from subcortical damages such as thalamic or striatal involvement than from cortical lesions (Starkstein et al. 1993; Murakami et al. 2013). In general, the prevalence of apathy in diseases involving the basal ganglia is reported to be around 40 %.
In summary, apathy can be observed in various neurological diseases as long as they affect the anterior cingulated-subcortical circuit. In AD, this is due to cortical involvement, while after stroke it is more likely to be related to subcortical lesions.
From the neurobiological point of view, the projections from the frontal cortex to the striatum are mediated by glutaminergic neurotransmission as well as the projections from the thalamus to the cortex. The connections between the striatum and the globus pallidus externa-substantia nigra are mediated by γ-aminobutyric acid (GABA) as well as the connections between the latter and the thalamus (Tekin and Cummings 2002). However, these circuits are modulated by other neurotransmitter systems. The dopaminergic neurons projecting from the substantia nigra pars condensa to the striatum can affect the cingulate-subcortical circuit. Apathy is frequently observed in Parkinson’s disease (Aarsland et al. 2007). Murakami et al. (2013) studying 149 post-stroke patients found that apathetic patients had lesions mostly located in the brain stem and in the striatum on MRI and suggested that apathy symptoms were associated with serotoninergic and dopaminergic dysfunction. A significant correlation between dopamine striatal activity measured using 11C-raclopride and anterior cingulated metabolism was demonstrated in the context of age-related decline (Volkow et al. 2000). David et al. (2008) found a significant correlation between lack of initiative and striatal dopamine transporter levels, measured with SPECT independently of motor activity, in a small group of patients presenting either AD or Lewy body dementia. The neurotransmission systems implicated by the results of the specific apathy treatment effects include dopamine, acetylcholine, serotonin and norepinephrine (van Reekum et al. 2005).
32.3 How Does Apathy Reflect on Molecular Imaging?
The first imaging results which strengthened the anterior cingulated circuit concept were obtained using regional cerebral blood flow (rCBF) SPECT studies. They were confirmed later on using 18-fluorodeoxyglucose PET metabolic imaging.
32.3.1 SPECT Studies
The first study was published by Craig et al. (1996). They studied 31 AD patients using the Neuropsychiatric Inventory (NPI) (Cummings 1997) and perfusion SPECT. Perfusion SPECT data were obtained using a 133Xe calibrated 99mTc-HMPAO method which combined the spatial resolution of HMPAO with the absolute quantification of regional cerebral blood flow provided by 133Xe. With this method, rCBF could be expressed in millilitres per hundred grams and per minute (Darcourt et al. 1993). Regional measurements were performed using regions of interest (ROI) automatically derived from an MRI template coregistered to SPECT images. They found a significant correlation between the NPI apathy score and the reduced regional cerebral blood flow in the anterior frontal regions – including the anterior cingulate area – and in the thalami. These correlations survived taking into account the covariance of MMSE and dysphoria. Although the results did not specifically involve the anterior cingulated gyrus but several anterior areas, this work was the first in vivo imaging study demonstrating the relationship between anterior cingulated cerebral blood flow and apathy using region of interest analysis (ROI) and absolute quantification.
Benoit et al. (1999) studying the “behavioural and psychological signs and symptoms of dementia” in a population of 20 AD patients observed that apathy was the most frequent behavioural symptom. Using ROI analysis on 99m-ECD SPECT images, they demonstrated a negative correlation between the NPI apathy score and the perfusion of the right anterior cingulate.
The first work addressing this issue using voxel by voxel analysis was performed by Migneco et al. (2001) who confirmed the specific involvement of the anterior cingulate gyrus in apathy. They included 41 patients; 21 were considered apathetic according to the NPI using a threshold of 2 on the apathy score. Twenty other non-apathetic patients were also included. SPECT studies were performed using 99mTc-ECD and a three-headed gamma camera (Prism 3000 XP, Philips) equipped with a LEUHR fan beam collimator. SPM96 was used to compare apathetic versus non-apathetic patients and showed a significantly decreased perfusion in apathetic patients in a very clearly defined zone corresponding to the anterior cingulated BA 24 (Fig. 32.2). The study which included 28 AD patients and 13 non-demented patients also showed that the results remained independent from the aetiology.
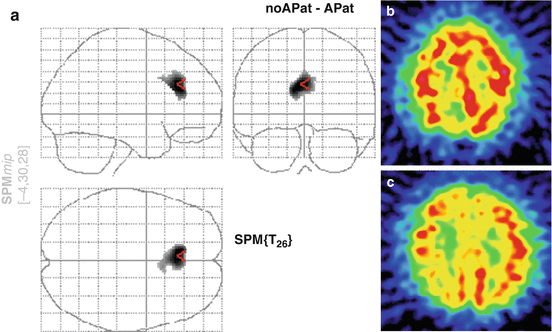

Fig. 32.2
99mTc-SPECT demonstration of anterior cingulate involvement in apathy (Adapted from Migneco et al. (2001)). (a) SPM analysis comparing apathetic and non-apathetic patients showing a very specific difference in anterior cingulated area (BA 24). (b) Transaxial cut involving the anterior cingulated cortex in a normal subject. (c) Same level of transaxial cut in an apathetic patient showing hypoperfusion in the anterior cingulate region
The same group (Benoit et al. 2004) using a dedicated Apathy Inventory scale (AI) (Robert et al. 2002) and SPM analysis of 99mTc-ECD SPECT data was able to refine the correlation analysis. In 30 AD patients they correlated the behavioural and emotional dimensions of apathy measured using the AI with rCBF. Total AI score was negatively correlated with the left and right superior orbitofrontal gyrus and with the left middle frontal gyrus (BA 10) perfusion. Concerning AI clinical dimensions, the lack of initiative score correlated negatively with anterior cingulated rCBF (BA 24 and 25). Lack of interest had an inverse correlation with orbitofrontal gyrus rCBF (BA 10) and emotional blunting with left superior dorsolateral prefrontal cortex (BA8). According to this work, the different dimensions of apathy are related to different orbitofrontal areas, and anterior cingulated involvement is more specifically related to the lack of initiative.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





