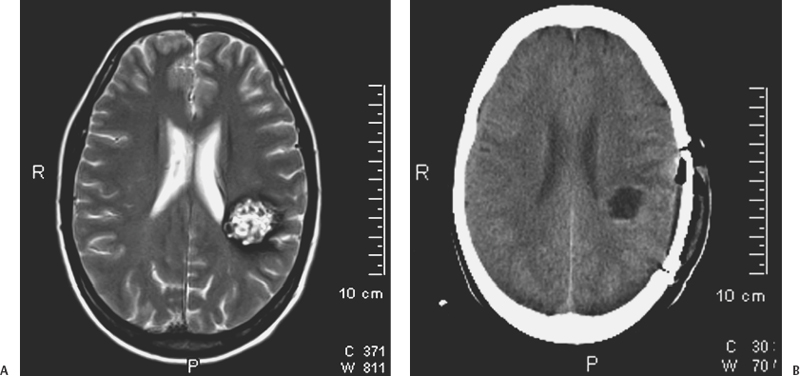
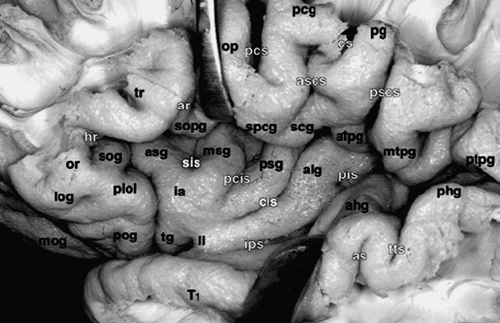
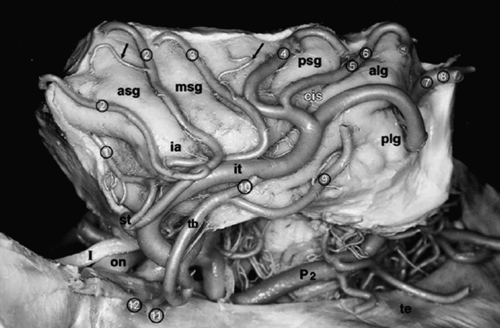
9 Carlo Bortolotti, Beniamino Nannavecchia, Giuseppe Lanzino, Paolo Perrini and Alvaro Andreoli Cerebral cavernous malformations (CMs) have a prevalence of 0.5%,1 and their distribution is proportional to the volume of the various portions of the central nervous system. Approximately 80% are distributed above and 20% below the tentorium. The majority of the supratentorial lesions occur in the cortical or subcortical regions.2 Thus, supratentorial CMs are frequently encountered in clinical practice. In this chapter, we will focus our attention on supratentorial lobar and insular CMs because deep-seated supratentorial CMs and CMs in atypical locations (i.e., cranial nerves) are treated in detail elsewhere. Cavernous malformations located in the supratentorial compartment are often located under the cortical mantle and adjacent to eloquent areas; therefore, they still represent a challenge for the neurosurgeon in terms of operative morbidity. The introduction and further development of image-guided neurosurgical systems has greatly facilitated some of the challenges encountered in the surgical removal of these lesions.3–9 As a result, several recent surgical series have reported excellent results after excision.10–14 Clinical manifestations of supratentorial CMs include seizures, hemorrhage, or progressive neurologic deficits with or without frank hemorrhage. With the widespread use of magnetic resonance imaging (MRI) a significant number of supratentorial CMs are discovered incidentally.15 Symptoms often present in the third or fourth decade of life, although supratentorial CMs can occur in childhood and older adults. Supratentorial CMs are relatively benign lesions with a small annual incidence of bleeding.16–25 The strategy in the management of cerebral CMs is to balance the natural history of these lesions (annual bleeding rate estimated to be between 0.25 and 0.7% per person per year in absence of prior history of hemorrhage) with the patient’s condition and the risk of surgery.26 The most frequent clinical presentation is seizure.14,27 Seizures secondary to supratentorial CM can often be controlled with medical treatment only, and the risk of hemorrhage is low in patients who present with seizure. For these reasons, some authors question the need to consider surgery in such cases unless the seizures cannot be medically controlled. However, numerous studies have reported improved seizure control after lesionectomy, and an inverse relationship exists between presentation-to-surgery interval and seizure control after surgical excision. Successful control of seizure is greatest in patients who undergo resection soon after onset of symptoms. The indications to consider surgery in patients with seizures are discussed in more detail elsewhere in this book. Overall, in the case of a patient with a single seizure at presentation or seizures of recent onset, surgery should be considered whenever safe because a short latency between the onset of seizures and surgical resection seems to correlate with better outcome. Unlike cerebral arteriovenous malformations, CMs rarely cause large intracerebral hemorrhage, and the incidence of symptomatic recurrent hemorrhage is relatively low.19 Nevertheless, surgery should be considered in patients with symptomatic hemorrhage as supratentorial CMs can be resected with a very low morbidity in the majority of cases.10–14 More challenging from a diagnostic point of view are those patients presenting with headache in absence of acute hemorrhage. Although CMs can cause headache, it is our policy to consider for surgical resection only those patients with refractory headache that clearly localizes to the area of the CM. Recently a “dynamic nature” of CMs in terms of “growing lesions” has been recognized.28–37 Surgical excision should be recommended in the case of lesions that have shown progressive enlargement on serial neuroradiologic studies even in the absence of symptoms. Observation is usually indicated in patients with incidental, asymptomatic lesions that remain “quiescent” and do not show any growth on serial MRI studies. The role of radiosurgery is limited for supratentorial non-basal ganglia CMs as currently these lesions can be simply resected and localized with very low morbidity and mortality.38,39 Cavernous malformations are frequently located adjacent to primary functional areas and can affect brain functions by mass effect, irritative effect, and consequence of hemorrhage. With recent refinements of minimally invasive surgery supported by neuronavigation systems, it is possible to surgically resect the majority of supratentorial CMs with low surgical morbidity3,6–8 (Fig. 9-1). Moreover, intraoperative recognition of eloquent cortex areas by integration of functional data (fMRI) with stereotactic or intraoperative MRI further facilitates safe and radical resection of CMs even in the primary motor, sensory, or speech areas.4,5,9,40 Schlosser and coworkers41 performed fMRI studies in 24 patients harboring vascular malformations (22 AVMs [arteriovenous malformations] and 2 CMs) adjacent to primary somatosensory, motor, or visual cortex. They analyzed functional MRI (fMRI) imaging before and after therapeutic intervention and correlated the results of post-therapeutic fMRI studies with the patients’ clinical status. Functional activation did not lie within the nidus of the lesion in any of the 24 baseline studies. Jannin et al.5 reported their results using neuronavigation during surgery of supratentorial CMs through a transsulcal approach. They used 3D MRI reconstructions to select and transfer the sulci close to the CM to the neuronavigation system and superimposed the graphics into the right ocular of the microscope. This application of the neuronavigation system resulted in shorter skin incisions as well as less brain trauma by using the course of a sulcus as trajectory. More recently, Gralla and coworkers4 retrospectively analyzed the postoperative results of 26 patients with deep-seated CMs or CMs adjacent to critical areas operated with the aid of neuronavigation and intraoperative fMRI. They concluded that the intraoperative visualization of eloquent cortical areas by integration of functional information with neuronavigation allowed fast recognition of critical brain areas improving the safety of surgical resection. Despite significant advances in the surgical resection of symptomatic CMs in critical areas, insular CMs still pose significant challenges. Only a few reports are available to detail surgical resection of CMs in this area, and a mere 24 cases of surgical resection of insular CM were found in the literature (only six located in the left insula). Because of the particular surgical pitfalls of operating lesions located in the “fifth lobe,” a brief review on the anatomy and vascularity of this area is appropriate. The insular cortex or isle of Reil is a very complex structure that histologically represents several areas differing in their cortical organization: allocortex, located in the antero-inferior part, mesocortex surrounding the first one, and isocortex in the most superior and posterior portion.42–44 Of the cerebral lobes, the insula is the only one that cannot be seen on the surface of the brain because it is entirely covered by the frontal, parietal, and temporal opercula. This anatomic configuration, the complex vascularization provided by middle cerebral artery (MCA) branches, and the proximity of the basal ganglia/internal capsule complex makes operating in this region a technical challenge. This complexity also explains why many of the insula neurophysiologic functions have only recently been discovered.43 Figure 9-1 (A) Axial T2-weighted image demonstrates a typical supratentorial CM with the characteristic heterogeneously hyperintense reticuled central matrix surrounded by a continuous circumferential rim of T2 hypointense hemosiderin. (B) Postoperative CT scan after total removal of the lesion. Figure 9-2 Surface anatomy of the insula. alg, anterior long insular gyrus; as, acoustic sulcus; ascs, anterior subcentral sulcus; asg, anterior short insular gyrus; atpg, anterior transverse parietal gyrus; cis, central insular sulcus; cs, central sulcus of Rolando; hr, horizontal ramus of Sylvian fissure; ia, insular apex; ips, inferior peri-insular sulcus; li, limen insula; log, lateral orbital gyrus; mog, medial orbital gyrus; msg, middle short insular gyrus; mtpg, middle transverse parietal gyrus; op, pars opercularis of F3; or, pars orbitalis of F3; pcg, precentral gyrus; pcis, pre-central insular sulcus; pcs, precentral sulcus; pg, postcentral gyrus; phg, posterior Heschl gyrus; pis, postcentral insular sulcus; pog, posterior orbital gyrus; plol, posterolateral orbital lobule; pscs, posterior subcentral sulcus; psg, posterior short insular gyrus; ptpg, posterior transverse parietal gyrus; sis, short insular sulcus; sog, suborbital gyrus; sopg, subopercular gyrus; spcg, subprecentral gyrus; tg, transverse insular gyrus; tr, Pars triangularis of F3; tts, transverse temporal sulcus; T1, superior temporal gyrus. (From Ture U, Yasargil DC, Al-Mefty O, Yasargil MG. Topographic anatomy of the insular region. J Neurosurg 1999;90:720–733. Reprinted with permission.) Morphologically, the insular lobe has a pyramidal shape. It is separated from the rest of the cortex by anterior, superior, and inferior peri-insular sulci (Fig. 9-2). Laterally it is bounded by the frontal, parietal, and temporal opercula and medially (from lateral to medial) by the extreme capsule, claustrum, external capsule, lenticular nucleus, and internal capsula.44–46 The triangular surface of the insula is divided into an anterior and posterior part by the central sulcus of the insula, which extends obliquely from the superior peri-insular sulcus to the limen insulae. In about two thirds of cases, this sulcus represents the prolongation of the central sulcus. The anterior part of the insula is normally composed of three main gyri: the anterior, middle, and posterior insular gyri; whereas the posterior portion consists of an anterior and a posterior long insular gyri. In planning the surgical approach to an insular lesion, a key point is the knowledge of the arteries supplying this region. The insula is supplied mainly by the M2 segment of the MCA (Fig. 9-3). After dividing into the superior and inferior trunks at the level of the limen insulae, the M2 segments of the MCA fan out over the insular cortex and give rise to several arterial branches (Fig. 9-3). In their microanatomical study of the insular arterial supply, Ture et al.45 described an average of 96 insular arteries. Eighty-five to 90% of these arteries are classified as short insular arteries supplying the insular cortex and the extreme capsule (Fig. 9-4). Ten percent are medium-sized arteries and supply the claustrum and external capsule. The remaining 3 to 5% are long arteries reaching the corona radiata. Another contribution to the vascularization of the insula may come from the lateral lenticulostriate arteries (LLAs). The LLAs usually arise from the inferomedial aspect of the M1 segment, penetrate the anterior perforated substance, and supply the head and body of the caudate nucleus, the lenticular nucleus, the internal capsula, and the corona radiata.45 In some cases,47,48 the LLAs may originate from M1 as well as from the M2 segment and supply more lateral structures such as the claustrum and the external capsula. Ture and colleagues45 reported an M1 origin of the LLAs in 78% of their specimens. In the same study, the LLAs originated from the frontal or temporal branch in 18% of cases and from the superior or inferior trunk of the M2 near to the main bifurcation of the MCA in 4% of cases. During surgery for insular lesions, it is very important to be aware of these anatomic variations. Occlusion of one of these vessels while trying to reach a deep-seated insular CM may result in postoperative hemiplegia. The M3 segment of the MCA begins at the anterior, superior, and inferior peri-insular sulci where the arteries angle abruptly and supply the hidden medial surface of the frontoparietal and temporal opercula. They then continue their course on the surface of the Sylvian fissure to supply the cortical surface of the convexity becoming the M4 segments. In a microanatomical study of the insular vessels, Varnavas and Grand49 found that in 90.6% of the specimens, the same artery supplying the central insular sulcus continued on to become the rolandic artery. During surgery in this region, it is imperative to avoid damaging this vessel. Because of its location under the perisylvian opercula and its complex vascular supply, the insula presents numerous surgical pitfalls. Refinements of neuronavigation systems and improved understanding of the anatomy and physiology of this region have increased the safety and accuracy of microsurgery of the insular region. In planning surgery to an insular lesion, the nature of the lesion itself is very important in allowing safe resection. Unlike primary glial tumors, CMs are very well demarcated lesions that displace rather than infiltrate normal brain parenchyma. Thus, they lend themselves to be removed through a limited exposure. Outcome in surgery of insular CMs depends basically on four steps: (1) meticulous and wide opening of the Sylvian fissure while preserving the integrity of the perisylvian opercula and the Sylvian vessels; (2) microsurgical dissection of the M2-M3 branches and of the lenticulostriate vessels; (3) choice of a convenient trajectory to reach and remove the lesion that does not appear on the cortical surface; (4) respect of the internal capsula especially in deep-seated insular CMs. In our opinion, neuronavigation is an important aid in points (3) and (4) because precise localization of the lesion and avoiding damage of the internal capsule in deep-seated insular CMs still represent a challenge even for the most experienced and skilled neurosurgeon.
Supratentorial Cavernous Malformations
 Clinical Presentation and Indications for Treatment
Clinical Presentation and Indications for Treatment
 Surgical Treatment
Surgical Treatment
Cavernous Malformations in Eloquent Areas
Cavernous Malformations of the Insula
Microsurgical Anatomy and Vascularization of the Insula
Surgical Considerations and Technique
Supratentorial Cavernous Malformations
Only gold members can continue reading. Log In or Register to continue

Full access? Get Clinical Tree



