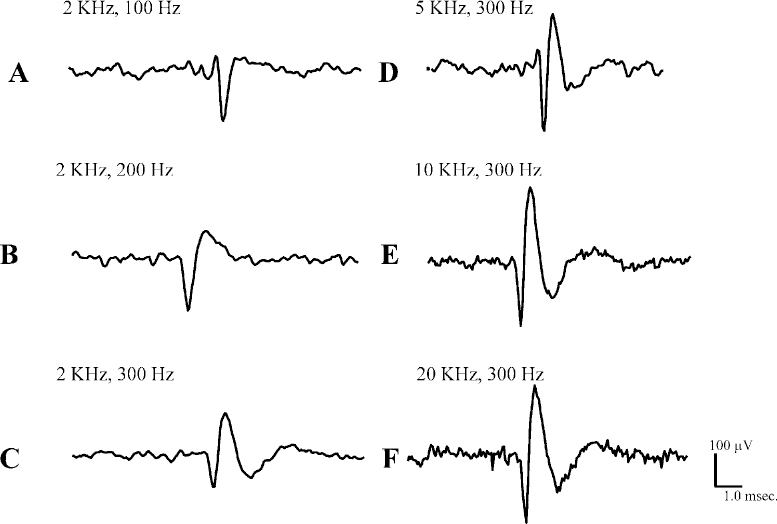
12 With the resurgence of pallidotomy and, more recently, with the advent of deep brain stimulation of the thalamus and the subthalamic nucleus in patients with refractory Parkinson’s disease and other movement disorders, performing microelectrode recording to localize the globus pallidus, the thalamus, and the subthalamic nucleus has become an integral part of the neurosurgical procedure. The electrophysiological properties of pallidal and subthalamic nucleus neurons can be used to localize these areas, allowing the accurate placement of lesions or stimulating electrodes within the target areas. Electrophysiological data collected during the neurosurgical procedure can be analyzed off-line and used to support models of corticobasal ganglia circuitry in PD and other movement disorders. These electrophysiological data, in combination with anatomical, histochemical, pharmacological, positron, and functional MRI studies have led to a better understanding of neural circuits involved in movement disorders. The purpose of this chapter is to give an overview of electrophysiological data analysis: its acquisition, its reduction, and the information that can be extracted from these analyses. The basic technique of extracellular recording of neuronal discharge has remained unchanged over the years,1 but with the emergence of integrated circuits and microcomputer technology, the electrophysiological equipment has become more compact and more user-friendly Several companies offer intraoperative microelectrode guidance systems for data acquisition. In the data acquisition process, certain electrical and biological factors will interfere with the recorded signal. These factors will always be present to varying degrees and will influence the quality of neuronal recordings and hence subsequent data analysis. It is important to recognize and identify these interfering factors and to endeavor to minimize them. At the same time, it is important to attempt to increase the magnitude of the biological signal (i.e., to achieve a high signal-to-noise ratio). The electrical properties of the microelectrode, such as tip size, tip configuration, and electrode impedance, can determine the magnitude and stability of the neuronal signal. The material used in the manufacture of the electrode, such as platinum,2 tungsten,3 and stainless steel, can also influence recording characteristics. Generally, a small-tip diameter (1–2 μm) microelectrode with high impedance (1.0 mΩ) can isolate the firing of single neurons, but electrical noise, as discussed below, may be substantial. Reducing the impedance of the microelectrode by platinizing it to increase the tip area, or by passing a small current through it to remove additional insulation, will reduce the impedance and will also reduce noise and 60 Hz interference. Good recordings from single neurons with commercially available electrodes are often obtained with impedances of 0.4 to 0.6 mΩ. Large-tip-diameter microelectrodes (> 20 μm) with low impedance (0.2–0.4 mΩ) will give rise to recordings with good stability and reduced electrical noise, but at the price of recording multiple units. Therefore, attempts are made to obtain a balance between these microelectrode properties such that stable recordings can be made from well-isolated single neurons for sufficient periods of time. One of the most common artifacts that can mask neuronal signals is the presence of 60 Hz and its second and third harmonics (240 Hz and 480 Hz, respectively) arising in the AC power supply to the lights and to the equipment in the operating room. The presence of this interference or “hum” will impede the visual and auditory recognition of spikes, making it difficult to collect data and to conduct subsequent off-line data analysis. Extracellularly recorded action potentials are in the μV or low mV range, whereas stray electrical signals are often in the tens of mV range. Much of the 60 Hz artifact can be eliminated by turning off nonessential electrical equipment. Proper grounding of the patient, the operating room equipment, and the recording equipment is essential. It is important to avoid “ground loops” in which there are multiple connections of different resistance between the patient, ground, and various pieces of equipment. In some cases, grounding the electrically powered operating room table will reduce 60 Hz. In other cases, grounding the table will increase 60 Hz. It is worthwhile to try a variety of grounding configurations of the equipment and to try removing equipment connected to the patient, such as EKG leads, which may be producing an “antenna effect.” An ohmmeter can be used to determine which equipment is grounded and to examine the electrical resistance pathway from the patient to ground, before and after the ground lead is attached to the patient. Fluorescent lights generate a considerable amount of electromagnetic noise and should be turned off if possible. In some circumstances, it is feasible to construct a Faraday cage around the operative field, which can reduce 60 Hz and other electrical noise. In general, removing 60 Hz in the operating room requires as much art as it does science. If grounding and shielding cannot eliminate 60 Hz artifacts, a 60 Hz notch filter may be incorporated into the recording system to eliminate unwanted frequencies. Such filters may be passive or active. The active filters may be thought of as “learning” the pattern of interference, then introducing a signal to counteract the interference. One drawback of active filters is that they may reintroduce unwanted interference for a brief period of time if the recording conditions are changed, until the new situation is “learned.” In contrast, passive filters may not be as efficient, but they may be depended on to greatly attenuate 60 Hz interference. Most oscilloscopes are designed to display a negative potential recorded at an electrode and led to the active input, with respect to ground, as a downward deflection on the screen. It is useful to calibrate the recording system with signals of known polarity and amplitude to be certain of the amplification of the system and the polarity display conventions that are being used. In extracellular recordings, low-pass (high-frequency cutoff) and high-pass (low-frequency cutoff) filters are employed to remove high-frequency and low-frequency interference, respectively. Filters for microelectrode recording are usually set to attenuate all signals below ~300 Hz and all signals above ~2 kHz. Although these filter settings will remove unwanted potentials and noise, there may also be some distortion of the morphology of the spikes. It is important to recognize these effects with respect to the electrical signal of interest. For the typical monophasic action potential of ~1 ms, the effect of increasing filtering of the high frequencies by reducing the low-pass filter setting from 20 to 2 kHz can be seen in Figure 12–1. The neuronal action potential, represented by the downward deflection (which represents a negative potential at the microelectrode), is reduced in amplitude and is made rounder by the filtering (compare Fig. 12–1F with Fig. 12–1A, B, and C). It is also apparent that decreased high-frequency filtering increases high-frequency baseline noise (Fig. 12–1F). A good compromise that preserves action potential morphology and minimizes baseline noise is the 10 kHz filter (Fig. 1E). The effect of reducing the amount of low-frequency activity allowed to pass by means of increasing the high-pass filter setting from 100 Hz to 300 Hz is shown in Figures 12–1A and 1C. The action potential is made smaller, and immediately following the action potential an artifactual potential of opposite polarity and longer duration is introduced into the recording. These effects are due to the properties of the resistance-capacitance (R-C) networks that are used in electronic recording equipment. They can be illustrated and understood by considering the effects of R-C networks on sine waves and on square waves of various durations that are passed through such networks. Therefore, the most faithful recording of the morphology of neuronal potentials will be made by using the filter settings as “wide open” as is compatible with stable recording. The brain normally moves with each respiration and each pulse wave. Brain movement (pulsation) moves neurons to and fro with respect to the recording electrode, resulting in changes in spike amplitude and often changes in frequency of discharge. It is important to recognize such changes, which can be accomplished by cross-correlation of the discharge with the suspected biological cause. Movement of a limb may be associated with changes in venous outflow from the brain, which may cause a change in intracranial pressure and produce brain movement and a change in discharge rate. As a microelectrode is advanced through the brain, there may be a sudden increase in pulsation artifact; presumably the electrode tip has come close to a blood vessel. The pulsation decreases as the electrode is advanced. Distortion or disruption of neuronal membrane results in an injury potential characterized by a marked increase in the firing rate of the neuron, often with decreasing amplitude of discharge. That this is an artifact and not a true reflection of neuronal activity can be seen by the lack of stability in the recording.
Techniques in Data Analysis: General Principles
MANJIT SANGHERA AND ROBERT GROSSMAN
Factors Influencing the Quality of Microelectrode Recording
Properties of the Microelectrode
Ambient Electrical Signals
Recording Conventions
Filters
Biological Artifacts
Neupsy Key
Fastest Neupsy Insight Engine