Fig. 21.1
Diagram of the GFP, MNTS1, and MNTS1/p75− lentivirus vectors
We monitored the locomotor functional recovery in all four groups: control, MSC, MSC-MNTS1, and MSC-MNTS1/p75−, respectively, using the BBB scoring scale and BBB sub-score [44, 45] (Fig. 21.2a, b). There was no significant difference in the BBB scores and BBB sub-score between the cell transplant and control groups. To evaluate the sensitivity of hindlimb cutaneous hypersensitivity, heat and mechanical stimuli were examined at 4 and 6 weeks after injury (Fig. 21.2c, d). Prior to contusion injury, the mean latency in second(s) to withdraw from a noxious heat stimulus was 17.7 ± 0.4 s (Intact group, n = 10). Following contusion injury, withdrawal latencies to noxious heat in the control and MSC groups were significantly decreased at 4 or 6 weeks following injury (4 weeks; 12.5 ± 0.8 and 13.4 ± 0.9 s or 6 weeks; 11.7 ± 0.8 and 11.8 ± 0.7 s, respectively). On the other hand, the MSC-MNTS1 and MSC-MNTS1/p75− group demonstrated significantly improved withdrawal thresholds compared to the control and MSC groups at 6 weeks after contusion, such that thresholds (6 weeks; 14.6 ± 0.7 and 16.2 ± 0.6 s, respectively) were no different from that of pre-contusion thresholds. Prior to contusion injury, the mean withdrawal threshold to innocuous mechanical stimuli was 14.8 ± 0.2 grams (Intact group, n = 10). Following contusion injury, withdrawal latency to mechanical stimuli in the control and MSC groups was significantly decreased at 4 or 6 weeks following injury (4 weeks; 8.0 ± 1.2 and 10.2 ± 1.0 s or 6 weeks; 9.1 ± 1.0 and 11.4 ± 0.9 s, respectively). In contrast, the MSC-MNTS1 group demonstrated improved withdrawal thresholds compared to the control group at 4 or 6 weeks after contusion, such that thresholds (4 weeks; 12.1 ± 1.0 s, 6 weeks; 12.9 ± 0.6 s, respectively) were no different from that of pre-contusion thresholds. The MSC-MNTS1/p75− group also demonstrated improved withdrawal thresholds compared to the control and MSC group at 6 weeks after contusion, such that thresholds (14.3 ± 0.5 s) were no different from that of pre-contusion thresholds.
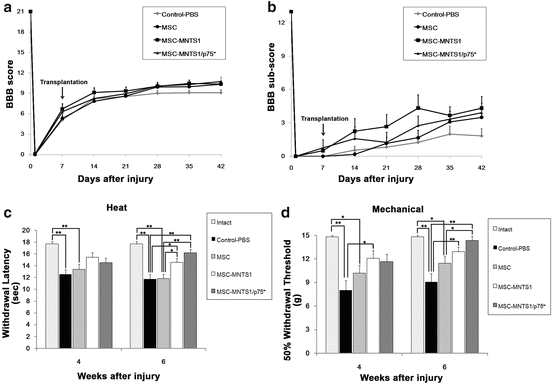

Fig. 21.2
Transplanted MSC-MNTS1 and MSC-MNTS1/p75−, but not MSCs, reduce the sensitivity of cutaneous hypersensitivity. (a and b) Open-field walking as determined by the BBB scores and BBB sub-score. Downward arrow indicates time of transplantation. Hindlimb function was assigned scores from 0 to 21 (flaccid paralysis to normal gait). Values are means ± SEM (n = 12, each). (c and d) Rats were tested at 4 and 6 weeks following contusion and hind paw responses to noxious heat (heat) and innocuous mechanical (mechanical) stimuli were recorded. Values are means ± SEM (n = 12, each). *: P < 0.05. **: P < 0.01
Histopathological analysis of tissue by hematoxylin and eosin and luxol fast blue staining was based on area and volume values of contusion calculated by computer extrapolation using stereological methods from contour/analysis of single sections. Cavity, lesion, and preserved myelin volumes and area were obtained in the control and transplant groups. Cavity volume revealed significantly greater preservation in the MSC-MNTS1/p75− group compared with the control group (P = 0.036). Cavity area at 1,000 μm rostral to the epicenter revealed significantly greater preservation in MSC, MSC-MNTS1, and MSC-MNTS1/p75− compared with the control group (P = 0.014, P = 0.042, and P = 0.011, respectively). There was no significant difference in the lesion volume and area between the cell transplant and control groups. There was also no significant difference in preserved myelin volume and area between the cell transplant and control groups.
To assess the effect of increased neurotrophin on axonal growth by comparing the MSC, MSC-MNTS1, and MSC-MNTS1/p75− groups, we examined neurofilament subunit (NF-M)-positive axons using stereology at the epicenter and at 4 mm rostral and caudal to the epicenter in axial sections (Fig. 21.3). While few NF-M-positive neuronal axons were observed at the rim of the lesion epicenter in both the control and MSC groups, there were significantly more NF-M-positive neuronal axonal counts in the MSC-MNTS1/p75− group at the 4 mm site rostral to the lesion epicenter compared with the control group (P = 0.011) and at the lesion epicenter compared with the control and MSC groups (P = 0.020 and P = 0.009, respectively). There were significantly more NF-M-positive axonal counts in the MSC-MNTS1 group at the 4 mm caudal to the lesion epicenter site compared with the MSC group (P = 0.043).
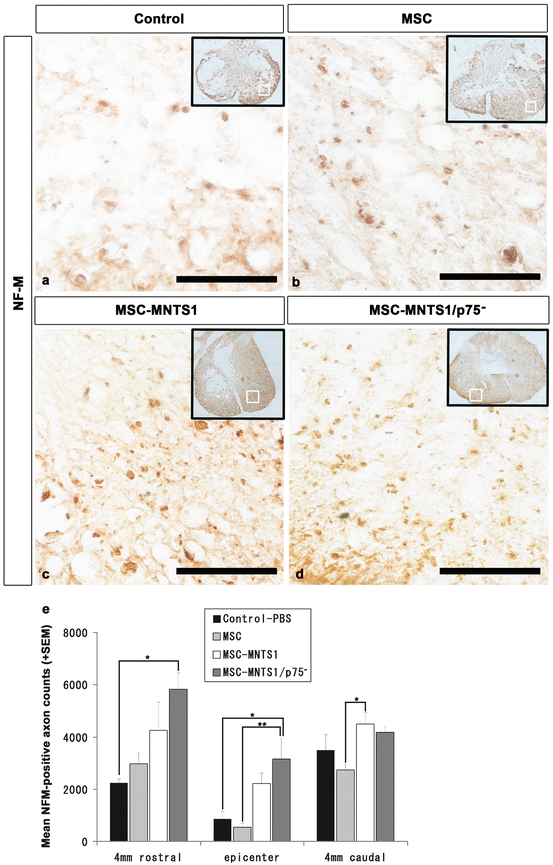

Fig. 21.3
Transplanted MSC-MNTS1 and -MNTS1/p75–, but not MSCs, promote axonal growth. (a–d) Representative images of axial sections stained for NF-M at the lesion epicenter in all four groups. Many more NF-M-positive axons were present in axial sections of the MSC-MNTS1 Fig. 21.3 (continued) (c) and MSC-MNTS1/p75− (d) groups, when compared to control (a) or MSC (b) groups. Scale bar: 50 μm. (e) Quantitative analysis of the NF-M-positive axon counts at the epicenter and 4 mm rostral and caudal to the epicenter. While few NF-M-positive neuronal axons were observed at the rim of the lesion epicenter in the control group, there were significantly more NF-M-positive neuronal axon counts in the MSC-MNTS1/p75− group at 4 mm rostral to the lesion epicenter site compared with the control group and at the lesion epicenter compared with the control and MSC groups. There were significantly more NF-M-positive neuronal axon counts in the MSC-MNTS1 group at 4 mm caudal to the lesion epicenter site compared with the MSC group. Values are means ± SEM (n = 4, each). *: P < 0.05, control versus MSC-MNTS1 or MSC-MNTS1/p75−. **: P < 0.01, MSC versus MSC-MNTS1/p75−
To assess the effects of transplantation on the extent of angiogenesis after SCI, axial sections of the injured spinal cord were examined immunohistochemically using stereology for RECA-1 (Fig. 21.4, a marker for endothelial cells). While a few RECA-1-positive blood vessels were observed at the lesion site in axial sections of the control and MSC groups, significantly more RECA-1-positive blood vessels were found in the MSC-MNTS1/p75− group compared with the control and MSC groups (P < 0.01 and P = 0.024, respectively).
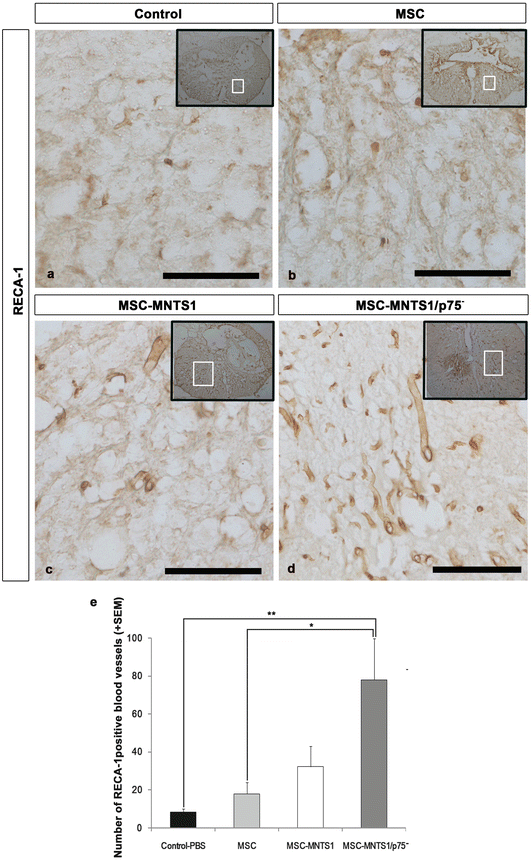

Fig. 21.4
Transplanted MSC-MNTS1, but not MSCs and MSC-MNTS1/p75−, promotes angiogenesis after SCI. (a–d) Representative images of axial sections stained for RECA-1 at the lesion epicenter in all groups. Many more RECA-1-positive blood vessels were present in axial sections Fig. 21.4 (continued) of the MSC-MNTS1/p75− groups (d), when compared to control (a), MSC (b), and MSC-MNTS1 (c) groups. Scale bar: 100 μm. (e) Quantitative analysis of blood vessels at the lesion epicenter. RECA-1 immunostaining revealed that significantly more RECA-1-positive vessels were observed at the lesion site in the MSC-MNTS1/p75− group compared with the control and MSC groups. Values are means ± SEM (n = 4, each). *: P < 0.05, control versus MSC-MNTS1/p75−. **: P < 0.01, MSC versus MSC-MNTS1/p75−
Contusion injury to the spinal cord results in the chronic inflammatory events including activated microglia and macrophages. To assess the effects on the recruitment of these cells in the injury site, we examined ED-1 immuno-positive cells which are characterized as activated microglia/macrophages using stereology at the epicenter in axial sections. Quantitative analysis revealed that there were significantly more ED1-positive cells at the epicenter site compared with the MSC, -MNTS1, and -MNTS1/p75− groups (P < 0.01, each). To assess the effects of transplantation on astrocyte reactivity after SCI, axial sections of the injured spinal cord were also examined immunohistochemically using stereology for GFAP. While GFAP immunoreactivities of the control and MSC groups were found in the white matter, those of the MSC-MNTS1 and -MNTS1/p75− groups were reduced in that area. There were also significantly more GFAP-positive cells in the control group at the epicenter site compared with the MSC-MNTS1/p75− groups (P = 0.037).
To assess the cell survival of the MSC, -MNTS1, and -MNTS1/p75− grafted onto the injured spinal cord, coronal sections of the injured spinal cord were examined immunohistochemically for GFP. At three posttransplant periods, large numbers of transplanted cells were identified at 1 week after transplantation, followed by a gradual decrease around 4 weeks. In contrast, few grafted cells were found at 6 weeks after transplantation.
21.4 Discussion
Together our results indicate that cellular treatments after contusive SCI with MSC-MNTS1 or MSC-MNTS1/p75− reduce cutaneous hypersensitivity but do not improve recovery of motor function. Although our studies showed that in all transplanted groups the early survival of transplanted cells, attenuation of acute inflammation and a reduced cystic cavity volume, the transplantation of the MSC-MNTS1 and MNTS1/p75−, but not the naïve MSCs, promoted axonal growth and prevented hypersensitivity after SCI. Moreover, transplantation of MSC-MNTS1/p75− promoted angiogenesis and modified glial scar formation. Finally, we provide for the first time evidence that treatment with transduced MSCs alleviated SCI-induced mechanical allodynia and thermal hyperalgesia of the hindlimb significantly better than documented with naïve MSCs.
Recently, transplantation of MSCs after SCI has been widely tested because they can be used as autografts, can be readily collected within a short time, and can be genetically modified to overexpress neurotrophic factors. Published studies indicate that the transplantation of MSCs promote overall functional recovery after SCI [17, 19–22] and do not induce allodynia in response to mechanical or thermal stimulation [23]. The mechanisms by which MSCs exert their effects on the injured spinal cord have also begun to be elucidated [6, 25, 46]. There is increasing evidence that MSCs may be immunosuppressive, synthesize a number of neurotrophic cytokines that stimulate nerve growth, including BDNF, NGF, and vascular endothelial growth factor (VEGF). These transplanted cells may also act as guides for regenerating axons across the lesion site of the injured spinal cord [25].
In contrast to reported positive findings, other studies indicate no significant improvements on BBB motor score after transplantation of MSCs into a contused or transected spinal cord [15, 47–49]. Here we report that naïve MSC also failed to promote functional recovery and reduce hypersensitivity after contusive SCI [10]. MSCs have been shown to attenuate chronic inflammation and injury-induced sensitivity to mechanical stimuli after experimental SCI [23]. Our results show that although there were significantly less ED1-positive cells in the naïve MSC-transplanted group compared with control, no significant prevention of hypersensitivity after SCI was observed with this cell treatment [10].
The generation of neuropathic pain after SCI is a major quality of life issue for many people living with SCI [50, 51]. Regarding the positive results of preventing hypersensitivity in the MSC-MNTS1 and -MNTS1/p75− groups, several mechanisms may underlie the improvement of sensory function [52]. Neurotrophins affect essentially all biological aspects of neuronal function, including survival, differentiation, growth, and apoptosis by using a two-receptor system, consisting of Trk tyrosine kinases and the p75NTR neurotrophin receptor [26]. Three types of Trk receptors, namely, TrkA, TrkB, and TrkC, mediate the biological activity of neurotrophins. Trk receptor tyrosine kinases undergo rapid transphosphorylation following ligand binding, leading to a cascade of protein phosphorylation in the cell. The distinctive neuronal deficiencies reported in mice with null mutations of the TrkA, TrkB, and TrkC genes are similar to those observed in mice with null mutations in the NGF, BDNF, and NGF genes, respectively. Therefore, the Trk receptor appears to mediate the survival-promoting action of neurotrophins on developing neurons.
Multineurotrophin NT3/D15A is capable of mimicking the downstream effects of BDNF and NT-3 through binding to both TrkB and TrkC receptors, respectively [33]. Transplantation of NT3/D15A-expressing glial-restricted precursor cells in the subacute phase of SCI led to an increase of myelin formation and recovery of hindlimb locomotion [34]. Transplantation of NT3/D15A-expressing Schwann cells in the subacute phase of SCI also promoted the survival of transplants and increased myelination as well as 5-HT-, DβH-, and CGRP-positive fibers in the implants [35]. MNTS1, which contains only seven amino acid changes, binds all receptors of the Trk family. This molecule efficiently induces autophosphorylation of TrkA, TrkB, and TrkC and supports the survival of more sensory neurons from DRG and NG as well as sympathetic neurons than any of the neurotrophins individually [33].
One of the most prominent biological functions of p75NTR is that it induces cell death through the death-domain sequence which is distantly related to the intracellular domains of Fas and TNF receptors. This effect is mediated by the proneurotrophin form [26]. Neurotrophins and their receptors are essential growth factors in the formation of the heart and vascular system, and the complete ablation of the p75NTR causes severe developmental defects [53]. Also, p75NTR has been implicated in endothelial cell apoptosis [54]. Consistent with these findings, our study shows that animals receiving MSCs secreting the MNTS1/p75− had more RECA-1-positive blood vessels within the injured spinal cord [10]. This result may be related to the Trk receptor activation which has been shown to promote angiogenesis. Although we observed reduced cavity volume and extensive axonal growth in the transplanted MSC-MNTS1 and -MNTS1/p75− groups, this morphological finding did not result in improved locomotor functional recovery [10]. The failure to observe motor recovery is most likely due to the fact that the cell transplants may not have survived chronically in the present contusive SCI model. Thus, longer-term survival may be necessary for the cell transplants to influence the preservation of myelin and vital white matter tracts critical in the return of locomotor function after thoracic SCI. Also, additional studies are required to determine whether multiple MSC injections at possibly different pericontusional sites may be necessary to promote walking in this SCI model.
21.5 Conclusion
MSCs have a great potential as therapeutic agents against various human diseases including SCI. These cells can be readily obtained through established clinical procedures and are easy to isolate and expand for autotransplantation with limited risk of rejection. In this study, we have succeeded in preventing cutaneous hypersensitivity after SCI by the transplantation of MSCs secreting a multineurotrophin without p75 activity. Further studies are required to ensure the long-term safety and efficacy of manipulated MSCs to improve locomotor function and other quality-of-life issues after SCI.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








