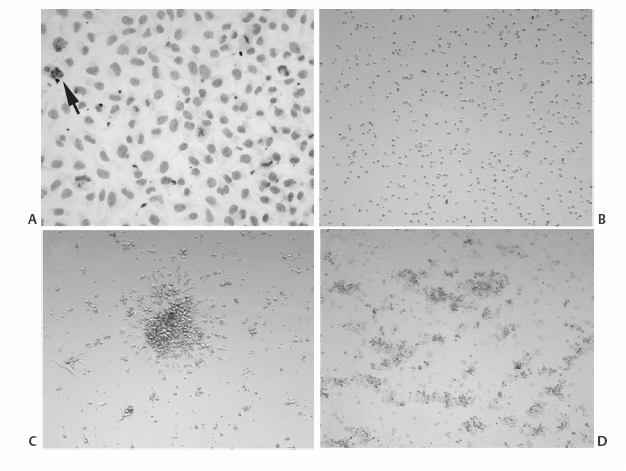
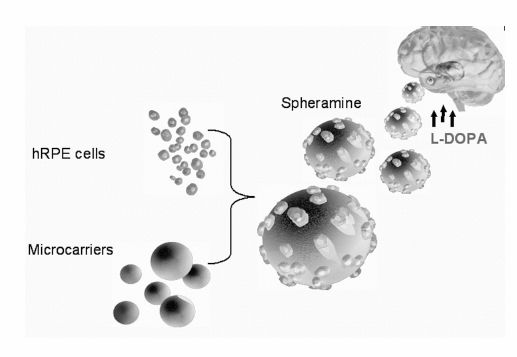
17 The Future of Treatment for Advanced Parkinson Disease Shivanand P. Lad, Eleonora M. Lad, Roy A. E. Bakay, and Jeffrey H. Kordower This chapter provides an overview of our present understanding and some of the future potential for various surgical and/or restorative treatments for advanced Parkinson disease (PD) that have been performed to date and are currently being investigated. The continued success of deep brain stimulation (DBS) requires device optimization, advances in lead/stimulation technology, and effective combination of DBS with adjunct techniques. First, leads must be more durable and less inflammatory and must easily provide stimulation while resisting electromagnetic interference. Further understanding of electrode properties on brain parenchyma, through clinicopathological studies and the Medtronic-sponsored market surveillance “Brain Autopsy Research Program” (www.medtronic.com), will aid in these adaptations. Next there must be customized lead technology to more precisely deliver targeted stimulation, potentially through alteration of electrode geometries. To date, the use of neural engineering design tools and finite element models have led to a better understanding of how varying the dimensions of electrode contacts affects the volume of tissue activated at stimulation parameters used in clinical settings. One study demonstrated that when the heights and radii of contacts were increased, as compared with standard 3387/3389 electrodes, the volume of tissue activated increased and decreased, respectively.1 In addition to altering contact dimensions, varying spacing and number of contacts can offer additional customization of stimulation. Another potential modification involves altering the current direction. Potentially, by bisecting or quartering the lead, current could be focally spread toward areas for optimal effect and away from areas that induce side effects. Development of more sophisticated stimulation technologies, such as closed-loop systems, is also a potential area of growth. Currently, DBS is an open-loop system (i.e., no feedback is present to alter stimulation parameters as needed). The development of sensor technology for the DBS system could improve the efficacy of DBS in the treatment of diseases, such as epilepsy, where seizures occur intermittently and electroencephalographic changes precede ictal events. On-demand use of DBS for essential tremor has been tried with patients performing their own turning on and off of stimulation so that the unit was on only 22% of the time, thus improving battery life.2 The first true attempt at using DBS in a closed-loop system for the treatment of epilepsy through stimulation of the anterior nucleus of the thalamus resulted in a 40% seizure reduction in a small number of treated patients.3 Thus the technology is available, though the details and practical aspects will need to be refined. Further in the future, DBS technology may augment brain—computer interface systems (BCIs), which use invasive electrode techniques to obtain recordings from populations of individual neurons.4–10 Electrodes that simultaneously stimulate and record may facilitate learning and rehabilitation because preliminary studies have shown that electrical stimulation leads to improved motor outcome in impaired individuals.11–13 Though much work is needed to optimize microelectrodes and three-dimensional control for effective BCI, combining BCI technology with high-frequency stimulation may lead to further neurorehabilitation advances.14 Competing with the mechanical devices will be the biological therapies. Since its discovery in the 1960s, treatment of Parkinson disease (PD) has focused on dopamine (DA) replacement via the prodrug levodopa (L-dopa), which is converted to dopamine centrally by aromatic amino acid decarboxylase (AADC). To reduce the peripheral metabolism of L-dopa, and side effects such as nausea and vomiting, L-dopa is administered in combination with a peripheral AADC inhibitor such as carbidopa (Sinemet, Merck & Co., Inc., Whitehouse Station, NJ) or benserazide (Madopar, Roche, Switzerland). This is true even though it is now becoming widely appreciated that the nigrostriatal degeneration seen in PD may occur relatively late in the disease process15 and that PD is a disease of both the central and peripheral nervous system.16 Still most of the therapeutic interventions for PD focus on modifying cardinal symptoms that are mediated by nigrostriatal degeneration. In this regard L-dopa remains the most widely used and most effective antiparkinsonian agent to date, providing substantial clinical benefit initially for almost all PD patients. However, several limitations prevent its long-term clinical effectiveness.17 First, motor complications such as dyskinesias and motor fluctuations are common, developing in up to 80% of PD patients treated with L-dopa for > 5 years. Second, PD patients develop symptoms unresponsive to L-dopa therapy, including freezing episodes, postural instability, falls, autonomic dysfunction, constipation, depression, and dementia. Third, despite the early symptomatic value of L-dopa, the underlying neurodegenerative process in PD continues unabated, with most patients experiencing permanent disability. Altogether, chronic treatment with L-dopa coupled with advancing disease eventually results in severe disability for patients. Over time, it becomes increasingly difficult to deliver a dose of L-dopa that both controls parkinsonian motor features and avoids dyskinesias. Patients with advanced PD may cycle between disabling “on” and “off” states for the majority of each day. These problems limit the long-term utility of L-dopa and have resulted in a search for more effective treatment strategies. Current medical approaches to the treatment of L-dopa—induced motor complications include manipulation of the dose and frequency, addition of dopamine agonists, long-acting formulations of levodopa, catechol-O-methyl transferase (COMT) inhibitors, monoamine oxidase-B (MAOB) inhibitors, and N-methyl-D-aspartic acid (NMDA) receptor antagonists.18 These adjuvant treatments can provide benefit to individual patients, particularly when motor complications are mild, but are ineffective for patients with established motor fluctuations and dyskinesias. Over the past decade, insight into basal ganglia circuitry in the normal and disease states has allowed for the development of novel neuroprotective and restorative treatment approaches that have the potential to greatly improve the quality of life for PD patients. One of the many surgical approaches being investigated for the treatment of PD is neurotransplantation, a therapeutic approach with the aim of replacing the neurons that are lost in PD. The advantages of this method are targeted delivery of neurons, which attempts replacement of neurotransmitters and neurotrophic factors at the desired site, regulation of implanted cells by the surrounding brain cells, and long-term function due to incorporation of the implanted cells in the brain.19 Neural transplantation was first tested in animal models. As early as 1890, neural grafting started to be performed. These early experiments characterized the embryonic neurons in terms of phenotype and ability to create normal connections within the adult host brain. In the host studies employing transplantation as a treatment for neurological disorders, the types of cells transplanted were either fetal neural tissue or chromaffin cells of the adrenal gland. Optimal neural cells useful for transplantation are cells that have finished their migration in the developing brain, no longer divide, and are at a level of maturity at which they express the neuronal phenotype desired, but have not yet extended neurites that would be damaged during tissue collection and transplantation.20 Neural tissue collected for grafting by microdissection is transplanted either as a cell suspension or as solid grafts of small pieces of tissue via stereotactic methods. During tissue processing and transplantation within the host brain, fetal neurons are faced with numerous challenges that negatively impact their survival, such as oxidative stress, ischemia, lack of trophic factors, and host immune reaction against the transplanted cells. As a result, only 5 to 20% of transplanted cells survive the grafting procedure, with most of the cell death occurring in the first 4 days after transplantation.21 Grafts of neural origin can be (1) autografts, in which tissue is harvested from the host; (2) allografts in which cells are taken from an individual of the same species; or (3) xenografts in which tissue is obtained from a different species. During transplantation, the blood—brain barrier is temporarily opened by the surgical procedure, allowing the immune cells to have access to the brain. Allografts and xenografts present molecules on the cell surface that may be recognized by the immune system as foreign during this time period, causing the rejection of the grafts. Graft rejection can be prevented by the use of immunosuppressive therapy during the few days in which the blood—brain barrier is open. However, in clinical trials employing allogeneic fetal neural tissue, the duration of immunosuppression has varied extensively, from no immunosuppression use22 to up to 6 months.23 Transplantation immunology in the central nervous system (CNS) is complicated and rarely studied in primates. The published work suggests that allografts in primates and humans are unlikely to reject.24 PD is one of the CNS disorders most amenable to transplantation due to the strong relationship between cardinal symptoms and striatal dopamine insufficiency. Thus the hope is that transplantation of dopamine-secreting cells into the striatum may help reestablish striatal function and restore movement in PD patients. Neural transplantation as a clinical therapy for PD started in 1985 with autologous transplants of chromaffin cells of the adrenal gland.25 Transplanted chromaffin cells produce dopamine, norepinephrine, and epinephrine. The first clinical trials of chromaffin cell transplants were performed in Sweden and Mexico and were soon followed by trials by U.S. investigators. The results of the surgery were disappointing. The patients experienced a very modest and short-lived (1 to 2 years) improvement in symptoms, and 40% of the 126 patients that underwent surgery in the United States suffered from complications of the procedure.20 Data from patients that came to autopsy revealed that no DA-producing chromaffin cells had survived. Thus the improvement in symptoms in some patients was likely due to a neurotrophic effect on the host DA neurons.26,27 Due to the poor success of this surgical approach, chromaffin cell transplantation was halted in the United States in 1991. In 1988, human fetal ventral mesencephalon (VM) transplants replaced chromaffin cell transplants. During the following decade, more than 200 patients received fetal DA neuron grafts worldwide.19 The results of these transplantation trials were highly variable in terms of transplantation method, improvement in symptoms, and development of side effects. Although many grafted patients have shown motor improvement, others have experienced significant worsening of levodopa-induced dyskinesias (LIDs). LIDs in nongrafted individuals are involuntary, excessive movements that are due to long-term use of L—dopa, the mainstay pharmacological treatment for PD.28 Initial transplantation studies performed in the 1990s were unblinded and employed a small number of patients. Nevertheless, they suggested that fetal DA neurons survived and could generate moderate clinical improvements over 3 to 4 years.29–39 An important clinical grafting study was the large, double-blinded, placebo-controlled clinical trial performed by Freed and colleagues.22 Forty patients with severe PD, between the ages of 34 and 75, were randomly assigned to a neuron cell transplant group or to a sham transplant group. Before surgery, the Unified Parkinson’s Disease Rating Scale (UPDRS) scores of all 40 patients off medication were an average of 58 to 71, and the UPDRS scores on medication were 12 to 30. Each patient received solid tissue grafts derived from a total of four fetal VMs. The tissue was in the shape of strands that had been cultured for up to 4 weeks and then implanted in the putamen using a frontal approach. In the sham-transplanted patients, holes were drilled through the skull but transplant needles never penetrated the dura. The primary outcome of the study was a subjective global rating of change in disease severity at 1 year after surgery, ranging from -3.0 (maximal aggravation) to 3.0 (maximal improvement). At 1 year after surgical procedure, the transplantation group and sham-treated group did not differ in their mean scores on the global rating scale (0.0 ± 2.1 for the transplant group, and –0.4 ± 1.7 in the sham group, respectively). Fifteen percent of the patients experienced dystonia and dyskinesia during off phases, defined as periods when L—dopa was reduced or even withdrawn. Although these results were disappointing, transplantation was effective in ameliorating parkinsonian symptoms in the younger patients (under 60 years of age), as measured by UPDRS (p = .01), and Schwab and England scales (p = .006). A second double-blind, controlled transplantation trial was conducted by Olanow and collaborators.23 Thirty-four patients with severe PD between the ages of 30 and 75 were randomized to receive bilateral grafts with one or four donors VMs per side in the postcommissural putamen or a sham surgery. Transplanted patients had two surgical procedures separated by 1 week. Sham-treated patients received burr holes that only partially penetrated the skull. Unlike the method used by Freed et al,22 tissue used for transplantation was stored for a maximum of 2 days before transplantation. Patients were treated with cyclosporine for 2 weeks before the surgical procedure and for 6 months thereafter. Transplanted patients exhibited increased striatal fluorodopa uptake, suggestive of surviving DA neurons. The primary end point of the study was a change in the UPDRS scores during off phases. Unfortunately, results of the study revealed no difference in the primary end point measure between placebo and transplant groups. It was found that patients in the four-donor group had a tendency, albeit not statistically significant, to have a greater symptomatic improvement than patients in the one-donor group. Perhaps in a manner analogous to the Freed study, patients with milder disease (although not necessarily younger) had a significant treatment effect compared with sham-treated patients (p = 0.006). After the surgery, a large proportion of grafted patients (56%) developed dyskinesias during practically defined off periods despite amelioration of dyskinesia during on periods in many patients. The mechanisms mediating the postoperative worsening of some aspects of dyskinesias are unclear. There is reason to suspect that several factors are causative: graft-derived patterns of reinnervation, immune response to grafted cells, L—dopa priming, and others. The role of these factors on the development of levodopa and graft-induced dyskinesias is being elucidated in primate and rodent models.40,41 Clinical trials have suggested that fetal nigral transplantation is beneficial for a certain population of PD patients. However, many patients do not show symptomatic improvements after surgery, and many develop side-effects such as debilitating dyskinesias. In addition, the survival of implanted neurons on a percentage basis is relatively poor, making it necessary to transplant large amounts of tissue per patient. Fetal dopaminergic tissue is difficult to obtain due to a lack of availability and ethical considerations. Given these problems, it is not likely that fetal nigral grafting is feasible for generalized therapy for PD. However, all research performed in the field of nigral grafting was and continues to be an important learning tool in laying the foundation for future transplantation strategies using other cell-based therapies. Another exciting alternative source of cells are stem cells for which two principally different ways of grafting in PD are being evaluated.42–44 In the first method, cells are predifferentiated in vitro to dopaminergic neurons prior to transplantation (Fig. 17.1). Thus stem cells could become an almost unlimited source for the generation of DA neurons. The cell preparations could be standardized and quality controlled with respect to viability and purity. The second alternative is that the stem cells or progenitor cells differentiate in vivo to dopaminergic neurons after implantation into the striatum or substantia nigra. These neurons may integrate better as compared with primary fetal DA neurons and, in the ideal scenario, reconstruct the nigrostriatal pathway. However, whether this will be possible remains to be seen. It will require understanding the mechanism(s) needed to instruct the immature stem cells to differentiate into the missing DA neurons and function in the PD brain. Hypothetically, DA neurons could be made from stem cells of four different sources: embryonic stem cells from the fertilized egg, embryonic neural cells from the fetal or those of adult brain, or stem cells in other tissues (e.g., the hematopoietic system). Another still unresolved issue is whether nondopaminergic neurons and glial cells, normally present in the mesencephalic grafts used so far in PD patients, are important for the differentiation and function of the DA neurons. If this is the case, an enriched population of predifferentiated DA neurons may not be the optimal preparation. Fig. 17.1 Potential sources of dopaminergic cells are shown. (A) Human NT2 progenitor cells in culture for 3 days. Cell lines from tumor are a concern for tumor regression (arrow points to multinucleated cell). (B) Rat neural SVZ cells in isolation (division 1) within proliferation media. (C) Human neurosphere (conglomeration of neuroblasts) after replating 1 week in differentiation media. (D) Rat neural SVZ cells clumping together after three divisions (~21 days) and now 2 days in proliferation media. In general, cell replacement strategies based on stem cells or progenitor cells offer several advantages over fetal VM cells. First, human fetal material may not be needed, reducing both the practical and ethical issues associated with the use of fetal tissue. Second, unlimited numbers of dopaminergic neurons can be produced in vitro, allowing for a detailed characterization and standardization of their properties and quality. Third, these types of cells allow for the generation of stem/progenitor cell banks because the cells can be cryopreserved and expanded according to needs. Thus DA neurons derived from stem/progenitor cells constitute one of the most promising tools for cell replacement therapy in PD. The functional benefits observed in younger22 or less severe23 PD patients, even though these effects were observed with secondary end points, suggest that cell-based replacement therapies may be effective with proper patient selection. Thus there is still a future for dopaminergic cell—based therapies in PD if a reliable source of cells can be identified. Stem cells can be that source. Potentially, stem cells can be propagated in vitro to produce large numbers, if not a limitless number, of cells for grafting and studies are under way to create an environment to drive these cells to both neuronal and dopaminergic phenotypes. It appears that fibroblast growth factor and sonic hedgehog are necessary for the induction of a dopaminergic phenotype. Stem cells can be derived from embryonic, fetal, and adult sources although at the present time, the embryonic stem cells appear the most promising in preclinical studies. Initial attempts to study embryonic stem cells in models of PD used cells derived from mice. Following grafting into 6-OHDA lesioned rats, robust survival of implanted stem cells, the presence of a neuronal phenotype with dopaminergic properties, and functional recovery on standard tests relevant to this model have been demonstrated.45–47 As an intermediary step, investigators have grafted stem cells derived from monkeys into cynomolgus monkeys and rodents.47,49 Modest survival was reported for several months, but, clearly, optimization of the procedure is still required. Recently, Goldman and colleagues50 reported benefit following grafting of human embryonic stem cells in rats receiving intraventricular 6-OHDA. These cells were grown in vitro on mouse feeder layers and would not be practical for clinical trials. Still the authors found robust survival of grafted cells for up to 8 weeks following transplantation. In vivo, the cells maintained the dopaminergic phenotype that had been demonstrated previously in vitro. Additionally, they reported benefit on a variety of functional tests. Thus stem cells show promise for future therapeutic trials. However, significant challenges still remain. Prior to initiating clinical trials with stem cells, growth control in vivo needs to be demonstrated, long-term survival and maintenance of dopaminergic phenotype needs to be established, and, critically, the origin and mechanism of the off-medication dyskinesias, seen in the fetal transplant trials, need to be eliminated so this severe graft-induced side effect does not recur in future stem cell trials. A new option for cell transplantation has been explored. Human retinal epithelial pigment cells (hRPE) have long been known to produce a precursor to dopamine, L-dopa, as a step in the formation of neuromelanin.51 The hRPE cells are placed on a microcarrier consisting of 100 μm crosslinked porcine gelatin particles, and the product is called Spheramine (Titan Pharmaceuticals, Inc., South San Francisco, CA) (Fig. 17.2). These particles may enhance survival of transplanted cells in the CNS by providing a physical support structure for epithelial cell growth. These cells release several different trophic factors in addition to L-dopa. Spheramine grafts in rats improved motor function.52 A sham-controlledstudy in monkeys rendered hemiparkinsonian with intracarotid 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) demonstrated significant improvement in the experimental group with respect to monkey UPDRS motor subscore.53 The Spheramine-grafted monkeys demonstrated lasting dopaminergic activity, verified by positron emission tomography (PET) scan. Based on these favorable results, a pilot study was performed in six patients. All patients in the pilot study showed dramatic initial improvements of nearly 34% from baseline in UPDRS scores when off medication. Total UPDRS scores and multiple other behavioral tests demonstrated improvements at 6 months.54 At 12 months, three of six patients still showed improvement compared with baseline.55 This approach is in a pivotal phase II clinical trial (Bayer Scheria, Berlin). PD is a chronic neurodegenerative disease with symptoms typically appearing after ~50% of substantia nigra dopamine neurons have degenerated and ~70% of striatal DA is lost.51 Disease progression results in continued loss of nigral DA neurons and striatal DA at a rate of ~5 to 10% per year.57 The slow, progressive nature of this disease provides opportunities for therapeutic intervention aimed at blocking nigral cell loss, and possibly promoting regeneration and axonal sprouting of surviving dopaminergic neurons. Neurotrophic factors represent a family of proteins that play a critical role in controlling neuronal survival, differentiation, growth, and apoptosis and have therefore received considerable interest as therapeutic agents in neurodegenerative disorders. Since the initial discovery of the prototypic neurotrophin, nerve growth factor (NGF), 50 years ago by Levi-Montalcini, numerous additional trophic factors have been discovered.58–60 Attempts to supply trophic factors clinically have thus far been disappointing due to poor delivery methods. In initial clinical trials, trophic factors were delivered either systemically or into the cerebrospinal fluid (intraventricularly or intrathecally) in patients suffering from amyotrophic lateral sclerosis, peripheral neuropathy, and Alzheimer and PD. In each of these trials, the trophic factor did not reach its target neurons due to either poor penetration across the blood—brain barrier or limited passage of protein from the cerebrospinal fluid into the brain parenchyma. Furthermore, neurotrophic factors, whose receptors are expressed widely throughout the CNS, produce unacceptable side effects when delivered by non-site-specific routes. From past clinical trials, it is clear that, in addition to identifying a promising trophic factor, it is essential to have a viable delivery mechanism that ensures delivery specifically to those vulnerable neurons in that particular disease state. Dopaminergic nigrostriatal neurons are sensitive to a variety of trophic factors,59,60 including members of the fibroblast growth factor family (FGF-1, FGF-2), the glial cell line—derived neurotrophic factor family (GDNF, neurturin, persephin), platelet-derived growth factor (PDGF), insulin growth factors (IGF-1, IGF-2), brain-derived neurotrophic factor (BDNF), and neurotrophin-4 (NT-4). Of these proteins, GDNF seems to be the most potent trophic factor for nigrostriatal dopamine neurons that are affected in PD.
Advances in Deep Brain Stimulation
Biological Therapies for Parkinson Disease
Cellular Therapies for Parkinson Disease
Stem Cell Transplantation
Alternative Cell-Based Therapies
Neurotrophic Factor for Parkinson Disease
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








