Chapter 16 The Management of Epilepsy in Pregnancy
Introduction
Women with epilepsy have been reported to account for 0.3 to 0.4% of all pregnancies, although some population-based studies suggest a prevalence of epilepsy among pregnant women of up to 0.7%.1,2 The proportion of pregnancies with exposure to antiepileptic drugs (AEDs) is probably even higher, considering the increasing use of AEDs for other indications than epilepsy.3 The vast majority of these women will have uneventful pregnancies and give birth to perfectly normal children. However, medical management during pregnancy is a matter of special concern because maternal epilepsy and AED treatment are associated with an increased risk for an abnormal pregnancy outcome. The maternal and fetal risks associated with uncontrolled seizures generally necessitate continued drug treatment during pregnancy, but these seizure-related risks need to be weighed against the potential adverse outcomes in the offspring due to maternal use of AEDs. Further important issues to be considered are the effect of pregnancy on seizure control and gestation-induced alterations in the disposition of AEDs, as well as matters of relevance immediately after delivery such as breast-feeding.
Prepregnancy Counseling
To be effective, most actions that can be taken to optimize the management of epilepsy in pregnancy need to be completed or initiated before conception. Prepregnancy counseling is therefore essential. Issues that should be addressed are summarized in Table 16-1. Given that, in general, approximately half of all pregnancies are unplanned, information of relevance for future pregnancies should be the responsibility of all physicians treating young women with epilepsy. Such information should be given repeatedly and be brought up well before pregnancy is contemplated.
TABLE 16–1 Issues to be Addressed in Prepregnancy Counseling
| Genetic counseling concerning the risk of inheriting epilepsy |
| Interactions between hormonal contraceptives and antiepileptic drugs |
| The expected course of epilepsy during pregnancy and delivery |
| Fetal and maternal risks with uncontrolled seizures |
| Fetal risks associated with use of antiepileptic drugs during pregnancy |
| Possibilities and limitations of prenatal diagnostic screening tests |
| Principles of treatment of epilepsy during pregnancy |
| Folate supplementation |
Contraception
Considering the value of planned pregnancies in women with epilepsy, the importance of effective contraception cannot be overestimated. Enzyme-inducing AEDs may reduce the effectiveness of oral contraceptives by induction of the metabolism of estradiol and progesterone and possibly also by increasing the hepatic synthesis of sex-hormone-binding globulin (SHBG). The contraceptive failure rate has thus been estimated to increase several times among women on AEDs.4 AEDs with and without known inducing effects on oral contraceptives are listed in Table 16-2. These AEDs affect combined oral contraceptive pills, combined contraceptive patches, and progestogen-only pills, as well as progestogen implants.5 In women requiring enzyme-inducing AEDs, long-acting methods (e.g., medroxyprogesterone depot injection, hormone releasing or other intrauterine contraceptive devices, or barrier methods) should be considered, which in fact have been proposed as good options for all women with epilepsy. Should this not be acceptable, oral contraceptives containing at least 50 µg of estrogen could be considered.5
TABLE 16–2 Interactions Between Antiepileptic Drugs (AEDs) and Oral Contraceptives (OCs)
| AEDs Known to Induce the Metabolism of OCs | AEDs Known to be Induced by OCs |
|---|---|
| Phenobarbital | Lamotrigine |
| Primidone | Valproate* |
| Phenytoin | |
| Carbamazepine | |
| Oxcarbazepine | |
| Topiramate (at doses >200 mg/day) | |
| Lamotrigine (modest effect on norgestrel component) |
* Tentative, based only on one small study.9
The interaction between oral contraceptives and AEDs can be bidirectional. Estrogen-containing contraceptives reduce plasma concentrations of lamotrigine by at least 50%.6–8 This induction is rapidly reverted, and lamotrigine levels rise significantly during the pill-free week, when sequential pills are used.8 One small study suggests similar effects of combined contraceptives on valproate plasma concentrations.9 Concomitant use of valproate and lamotrigine, however, seems to block the effects of contraceptives on lamotrigine plasma concentrations.10
Maternal and Fetal Hazards with Seizures During Pregnancy
Epilepsy is a condition with potentially serious psychosocial and medical consequences for the patient. Seizures may cause physical injuries and occasionally even death and are thus good reasons to treat people with active epilepsy. These concerns related to the well-being of the patient with epilepsy in general are equally relevant during pregnancy. The importance of maintained seizure control during pregnancy is highlighted in a recent review of all maternal deaths in the U.K. during 1985–1999.11 Women with epilepsy were reported to account for 3.8% of all maternal deaths, considerably more than expected from the prevalence of epilepsy in pregnancy. The mortality was partly related to seizure occurrence after stopping AED treatment.11 Although the absolute risk is very low, the data underline the importance of seizure control for maternal health.
The fetal effects of maternal seizures depend on the type of seizures. Although other seizure types have negligible effects, generalized tonic-clonic seizures increase the pressure in the pregnant uterus and may lead to a trauma if the patient falls. Generalized tonic-clonic seizures also induce lactic acidosis,12 and this has been shown to transfer to the fetus.13 Convulsive seizures may also cause fetal bradycardia,14 and status epilepticus can result in intrauterine death.15,16 However, the large prospective EURAP antiepileptic drugs and pregnancy registry reported only one case of intrauterine death16 and no maternal mortality among 36 cases with status epilepticus. Furthermore, recent data suggest that the number of stillbirths is not increased among women that are adequately treated for their epilepsy during pregnancy.17,18 Occurrence of seizures during the first trimester does not seem to increase the risk of malformations in the offspring.2,18–33
Seizure Control in Pregnancy
So far, the largest prospective study on seizure control in pregnant women with epilepsy concluded that the majority, 58%, remained seizure free throughout pregnancy, and that 18% had convulsive seizures on some occasion during pregnancy (EURAP).16 Whether pregnancy in itself affects the course of epilepsy remains a partly controversial issue, with variable observations in the literature. Pooling data (altogether 2249 pregnancies) from studies published after 1980, seizure control was the same during pregnancy as before in 62.5%, improved in 11.4%, and deteriorated in 24.6%.21,25,34–51 These comparisons are, however, hampered by several methodological problems, including a different type of management during pregnancy compared to before and prospective follow-up during pregnancy compared to a retrospective prepregnancy baseline. Nevertheless, these data demonstrate that seizure control will be unaffected by pregnancy in most cases and that the majority of women with epilepsy will remain seizure free throughout. Some of the reported changes in seizure control may also be explained by the normal random fluctuations in seizure occurrence in epilepsy and thus be unrelated to pregnancy. However, women with localization-related epilepsy,16,37,46,49 epilepsy of long duration,34,39,43,46,47,49,50 and in particular poor seizure control before pregnancy46,50 are more likely to deteriorate. The effect of pregnancy may vary between patients with similar types of seizure disorders, but also in different pregnancies of the same patient,50,53 and is thus difficult to predict.
Pharmacokinetic, metabolic, hormonal, physiological, and psychological factors have all been suggested as contributing causes to gestational changes in seizure control. In some cases, changes in seizure frequency may be related to lack of compliance, not seldom due to maternal fear of teratogenic effects of AEDs,36,37 or for other reasons of decreased plasma AED levels.36,37,54,55
Recent reports from prospective pregnancy registers indicate that poor seizure control, increased seizure frequency, or need for increased dosage may be more common with lamotrigine and oxcarbazepine than with other AEDs.16,56 This might be related to pharmacokinetic changes, which will be discussed separately in the following section.
Although the data on general changes in seizure control during pregnancy are conflicting, the findings are consistent concerning an increased risk of seizures during labor and delivery. Seizures occur at labor and during delivery in approximately 2.5% of the cases,16,18,37,39,44,46 an almost 10-fold greater risk than otherwise during pregnancy. The risk is higher for patients with seizures earlier during pregnancy.16
Status epilepticus occurs in about 1% of the cases.16,34–37,39,40,43,44,46,49,50,57 The incidence of status epilepticus is probably not higher in pregnancy than otherwise. It requires prompt attention and should be treated according to the same principles as otherwise. Refractory status epilepticus in the third trimester of pregnancy could also be an indication for a caesarean.58
Effects of Pregnancy on Pharmacokinetics of Antiepileptic Drugs
Pregnancy is associated with several physiological changes that may affect drug disposition and thus maternal plasma concentrations and fetal exposure to AEDs.59,60 Decreased protein binding is relevant for highly bound drugs. It will result in reduced total plasma concentrations, but unaffected unbound drug levels. Because the unbound concentrations determine the pharmacological effects in the mother and exposure to the fetus, alterations in protein binding will not have any clinical consequences. However, total plasma concentrations can be misleading in pregnancy for highly protein-bound AEDs, such as phenytoin and valproic acid.61,62 Enhanced drug elimination due to induction of metabolizing enzymes is clinically the most important mechanism for gestation-induced alterations in AED kinetics. This occurs with drugs metabolized through the cytochrome P450 system (e.g., phenytoin and phenobarbital), but is even more pronounced for lamotrigine and possibly oxcarbazepine, drugs eliminated through glucuronidation.63–65
Effects of pregnancy on plasma concentrations of AEDs are summarized in Table 16-3. The decline in AED concentrations generally begins during the first trimester. In late pregnancy, the decrease is on average 55 to 61% for phenytoin total and 18 to 31% for unbound concentrations;61,66–69 0 to 42% for carbamazepine total and 0 to 28% for unbound concentrations; 50 to 55% for phenobarbital,62,73 55% for primidone; 50% for valproic acid total and 0 to 29% for unbound concentrations.61,74
TABLE 16–3 Effects of Pregnancy on Plasma Concentrations of Antiepileptic Drugs
| AED | Approximate Average Total Plasma Concentration in Late Pregnancy Compared to Before | Approximate Average Unbound Plasma Concentration in Late Pregnancy Compared to Before |
|---|---|---|
| Phenobarbital | 50% | Probably same as total concentration* |
| Phenytoin | 40–45% | 70–80% |
| Carbamazepine | 60–100% | 75–100% |
| Valproate | 50% | 75–100% |
| Lamotrigine | 25–40% | 30–40% |
| Oxcarbazepine | 40–50% | Probably same as total concentration* |
| Levetiracetam | 40–50% | Probably same as total concentration* |
| Topiramate | No data available | No data available |
| Tiagabine | No data available | No data available |
| Gabapentin | No data available | No data available |
| Pregabalin | No data available | No data available |
| Zonisamide | No data available | No data available |
* Data on unbound concentrations during pregnancy are lacking, but decline can be assumed to be the same as for total concentrations based on the comparatively low-binding to plasma proteins for this drug in general.
Lamotrigine plasma concentrations decrease by an average of 68% during pregnancy, with a wide interindividual variability and sometimes with deterioration of seizure control.75–79 The decrease of lamotrigine plasma levels is markedly reduced10 when lamotrigine is taken in combination with valproic acid.
More limited data indicate a decrease of a similar magnitude for the active moiety of oxcarbazepine, its monohydroxy derivative.80,81 A 50% decrease in levetiracetam concentrations has been reported in late pregnancy.82–84 Very limited or no information is available on possible changes in disposition of the other newer-generation AEDs.63
Most studies involving older-generation AEDs have failed to demonstrate a relationship between seizure control and alterations in AED plasma concentrations.35,45,49 However, with lamotrigine monotherapy breakthrough, seizures have been linked to the marked decline in plasma concentrations in a relatively high proportion of patients.78,79 Prospective pregnancy register data also indicate that patients on lamotrigine more often need either dosage adjustments during pregnancy16,56 or additional AEDs.16
Oxcarbazepine has been reported to be associated with a poorer seizure control during pregnancy compared to other monotherapies.16 This could also be explained by pharmacokinetic alterations because the pronounced decline in plasma concentrations of the active moiety of oxcarbazepine was associated with breakthrough seizures in a small case series.80
Developmental Toxicity
The most important among the effects that have been attributed to developmental toxicity of AEDs are intrauterine growth retardation, increased prevalence of minor anomalies and major congenital malformations, impaired postnatal cognitive development, and more or less specific fetal AED syndromes. Some of these effects are well documented, whereas the occurrence of others is more controversial.
INTRAUTERINE GROWTH RETARDATION
Reductions in body dimensions, in particular head circumference, have been reported in several cohorts of children exposed to AEDs.23,32,85–90 This has been associated with polytherapy,85–87,89,90 and some investigators have also found associations to monotherapy with phenobarbital, primidone, or carbamazepine. Wide et al.89 studied body dimensions in infants exposed to AEDs in utero in a Swedish population over a period of 25 years, comparing data to the general population. There was a clear trend toward normalization of the head circumference over the time period in parallel with a shift from polytherapy to monotherapy, despite an increasing use of carbamazepine. Other more recent studies also suggest that, with present treatment strategies where monotherapy prevails, microcephaly may no longer be more common among infants of mothers treated for epilepsy during pregnancy.91,92
MINOR ANOMALIES AND FETAL AED SYNDROMES
Minor anomalies are structural variations without medical, surgical, or cosmetic importance. These frequently occur in normal infants, but combinations of several anomalies can form a dysmorphic syndrome, which may indicate a more severe underlying dysfunction.93 Minor anomalies and dysmorphic syndromes have been reported more frequently in infants of mothers treated for epilepsy during pregnancy. Facial features such as hypertelorism, depressed nasal bridge, low-set ears, micrognathia, and distal digital hypoplasia, sometimes in combination with growth retardation and developmental delay, were first reported in association with exposure to phenytoin.94 Subsequently, however, similar patterns have been associated with exposure to carbamazepine.95 Valproate exposure has been claimed to cause a somewhat different dysmorphic syndrome characterized by thin arched eyebrows with medial deficiency, broad nasal bridge, short anteverted nose, and a smooth long filtrum with thin upper lip.93 However, there is a considerable overlap in the various dysmorphisms, and their drug specificity has been questioned. A more general term, fetal or prenatal AED syndrome, has therefore been suggested.96 In addition, the pathogenesis is still somewhat controversial, and some investigators have attributed most of the minor anomalies to genetic factors rather than drug exposure.97 It should, however, be underlined that minor anomalies are much more difficult to assess objectively than major malformations, and that the incidence of minor anomalies in exposed infants varies markedly between studies.98
MAJOR CONGENITAL MALFORMATIONS
The rate of major congenital malformations is two to three times higher among children of mothers with epilepsy compared to the general population. The reason for this risk increase is probably multifactorial, including effects of seizures and epilepsy, genetic and socioeconomic factors, and teratogenic effects of AEDs. However, the available evidence strongly suggests that exposure to AEDs is the major cause. Pooling data from 26 controlled studies,2,19,21,22,24,26,30,32,50,91,97,99–113 the malformation rate was 6.1% in offspring that had been exposed to AEDs (n = 4630), compared to 2.8% in children of untreated women with epilepsy (n = 1292) and 2.2% in offspring of mothers without epilepsy (n = 40, 221). Nevertheless, a genetic influence is shown by the increased risk of malformations when there is a family history of malformations.19,25,27,31,107,114–134 The importance of genetic susceptibility to teratogenic effects of AEDs is further supported by case-control studies reporting a higher proportion of relatives with epilepsy in patients with cleft palate or lip135–139 or neural tube defects.140,141
The fact that most studies report higher malformation rates in association with polytherapy than monotherapy with AEDs, and a dose-effect relationship for teratogenic risks with some AEDs, lends further support to the importance of AEDs for adverse pregnancy outcome among women with epilepsy. The rate of major malformations was 6.8% among children exposed to AED polytherapy (n = 4253), compared to 4.0% after monotherapy (n = 8339) when data from 74 studies were pooled.2,18,19,21–24,26,28,31,32,42,46,50,56,95,97,100–102,104–106,108–111,119,122,142–183
Several studies have observed a dose-related risk of birth defects in association with valproate exposure.25,152,153,158,160,184–186 Although some studies have failed to demonstrate this relationship,24,28,187 it appears that the risk is greater with dosages above 800 to 1000 mg/day. Recently a dose-related effect was reported also for lamotrigine,150 although this was not confirmed in another study.188 The severity of epilepsy may of course confound the association between doses and the risk of malformations, as well as the association between polytherapy and the risk of malformations, but it has shown beyond any reasonable doubt that exposure to AEDs is the major cause of increased birth defect rates among children of women with epilepsy. It is furthermore reasonable to assume that, in general, risks are lower with monotherapy than polytherapy and with lower compared to higher dosages.
The types of malformations reported at higher rates in the offspring of epileptic mothers are mainly heart defects, neural tube defects, facial clefts, hypospadias, and limb reduction defects. A specific association has been reported between neural tube defects and valproate114,140,153,189–194 and, to a lesser extent, between barbiturates and heart defects.114,140,153,189–194 Some studies also reported a higher risk of limb reduction defects associated with valproate exposure190,196 or hypospadias153,190 and a higher risk of oral cleft with barbiturates.27,190,192 A recent study also suggests an increased risk for oral clefts associated with lamotrigine.197
A major issue with profound implications for the management of epilepsy in pregnancy is whether AEDs differ in their overall teratogenic potential. Earlier studies have largely failed to address this because of methodological shortcomings, in particular insufficient numbers of pregnancies and low statistical power.198 For this reason, large prospective epilepsy and pregnancy registries have been established in recent years, each enrolling thousands of pregnancies with AED exposure. Some of these registries have published pregnancy outcome data in association with individual types of AED exposures. A summary of recent studies is presented in Table 16-4.
TABLE 16–4 Malformation Rates with Exposure to Different Antiepileptic Drugs in Monotherapy (N = Offspring with Malformation)
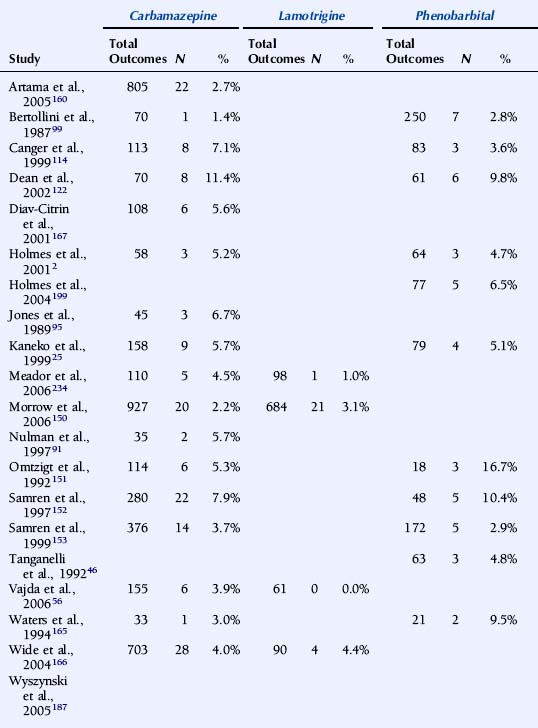
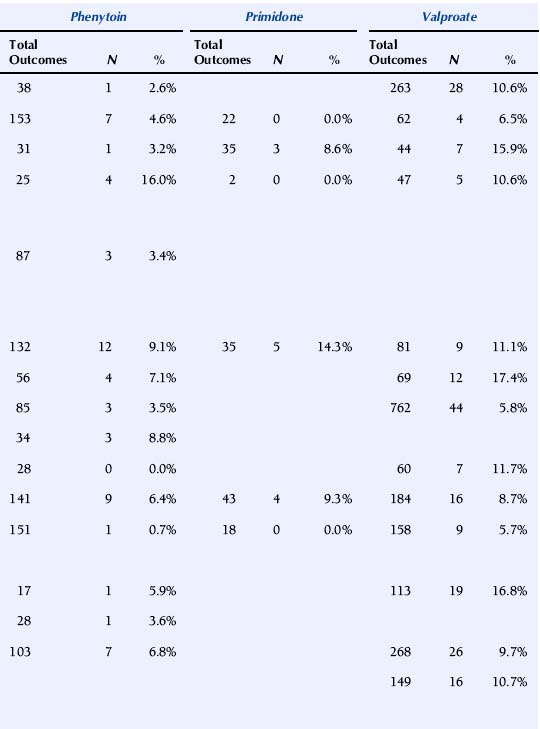
The U.K. Epilepsy and Pregnancy Register reported malformation rates based on 3607 prospective pregnancies.150 The overall prevalence of major congenital malformations for all cases exposed to AEDs was 4.2% (95% CI: 3.6% to 5.0%), slightly higher, but interestingly not significantly so, compared with 3.5% (1.8% to 6.8%) for infants of women with epilepsy who had not taken AEDs during pregnancy. The rate of birth defects was greater for pregnancies exposed to valproate in monotherapy, 6.2% (4.6% to 8.2%; n = 715) than to carbamazepine monotherapy, 2.2% (1.4% to 3.4%; n = 900). The malformation rate for those exposed to lamotrigine monotherapy was 3.2% (2.1% to 4.9%; n = 647). A correlation between major congenital malformations and drug dosage was found for lamotrigine.150
The North American AED Pregnancy Registry has released outcome data for four AEDs.187,197,199,200 With exposure to phenobarbital monotherapy 6.5% (95% CI: 2.1 to 14.5%; n = 77) had major malformations. The relative risk (RR) compared with the external background rate (infants of mothers without epilepsy from a Boston Hospital with a malformation rate of 1.62%) was 4.2 (95% CI: 1.5% to 9.4%), and compared with three other AEDs combined in monotherapy from the same registry 2.0 (95% CI: 0.9 to 4.5%).199
The rate of major malformations with exposure to valproate monotherapy was 10.7% (95% CI: 6.3 to 16.9%; n = 149). RR compared with background rate was 7.3 (95% CI: 4.4 to 12.2%), and OR compared with an internal comparison group (infants exposed to all other AEDs as monotherapy in the same registry) was 4.0 (95% CI: 2.1 to 7.4%).187 The prevalence of major malformations among infants born to women taking lamotrigine monotherapy was 2.7% (95% CI: 1.5 to 4.3%; n = 564). RR compared with the background rate was 1.7 (95% CI: 1.0 to 2.7%). An increased risk was found for orofacial clefts with exposure to lamotrigine, RR being 32.8 (95% CI: 10.6 to 101.3%) compared with background rate.197 Among infants exposed to carbamazepine as monotherapy in the first trimester, 2.6% (95% CI: 1.5 to 4.3%; n = 873) had major malformations. RR compared to the background rate was 1.6 (95% CI: 0.9 to 2.8%).200
The Australian AED and Pregnancy Register recently released outcome data based on 555 pregnancies, of which 485 were prospective.56 Valproate at doses above 1100 mg/day was associated with significantly higher risk of fetal malformations than other AEDs.
Comparatively high malformation rates with valproate have been reported in two additional national register studies. The nationwide population-based Swedish Medical Birth Registry was published based on 1398 pregnancies with exposure to AEDs.166 The OR for severe malformation after exposure to valproate monotherapy (n = 268) compared with carbamazepine monotherapy (n = 703) was 2.59 (95% CI: 1.43 to 4.68%). In a study based on the Finnish drug prescription database and the National Medical Birth Registry, (pregnancies with AED exposure (n = 1411), the risk of malformations was higher in fetuses exposed to valproate monotherapy (malformation rate 10.7%; OR = 4.18%; 2.31% to 7.57%) than of untreated patients. In contrast, the risk of malformations was not elevated in association with exposure to carbamazepine, oxcarbazepine, or phenytoin monotherapy.160
It is difficult to compare rates of birth defects between registries because of significant differences in methodology, including methods for enrollment and inclusion, exclusion criteria, and duration of follow-up, as well as criteria for teratogenic outcome. Nevertheless, a higher malformation rate in association with valproate compared with some other AEDs, in particular carbamazepine, has been a consistent finding. Recent studies indicate that the prevalence of birth defects with lamotrigine is similar to that with carbamazepine. The manufacturer’s International Lamotrigine Pregnancy Registry thus reported major birth defects in 22 of 802 first trimester monotherapy exposures (2.7%; 1.8% to 4.2%).188 Unfortunately, prospective data on teratogenic outcome in association with exposure to other newer generation AEDs is very scarce, and no firm conclusions can be drawn about their relative safety in pregnancy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








