*Changed values shown in bold.
Studies implemented in the next two decades evaluated the effects of gestational weight gain using these IOM 1990 categories and recommendations. This later work has supported the appropriateness of the primary recommendations for minimizing maternal complications and improving fetal outcomes (6, 7). In 2009, the IOM supported a re-evaluation of the 1990 guidelines (8). Because of the growing concern about obesity and the many studies done on this topic following the 1990 report, the reevaluation focused on the effect of pregnancy weight gain on maternal obesity development and particularly examined effects of weight gain in obese women. In the resulting publication, changes were made in the maternal BMI categories (to make them consistent with those used by the World Health Organization (WHO) and the National Heart, Lung and Blood Institute) and in the weight gain recommendations for these new BMI groups. These are also shown in Table 10-1.
Note that the changes in the BMI categories resulted in more women being classified as overweight, with fewer classified in the other three categories (9). As a result, the recommended gains of 13–18 kg for underweight and 5–9 kg for obese women applied to fewer women with more extreme weights than previously. In addition, as a result of the differences in the BMI categories and weight gain recommendations of the two reports, studies from before and after the 2009 report are not directly comparable. This chapter will indicate which publication’s categories were used when it is relevant to interpreting a study’s results.
To select the recommended weight gain range for a woman delivering at term, she must be placed in one of the four BMI groups (Table 10-1). Pre-pregnancy BMI should be determined at the first prenatal visit. When this is done, the mother’s measured height and reported pre-pregnancy weight are usually used, although a measured value for both is preferable. Studies show that only a minority (about one third) of women gain weight as recommended for their BMI group (10, 11), with more women gaining excessive weight than gaining too little (10, 12) even among obese women (13). Further, women generally overestimate height and underestimate weight (14, 15), resulting in underestimation of BMI and misclassification of some women into a lower BMI group, with its higher recommended gain. This is more likely to occur in women close to the lower cut-off of their true BMI group.
Underestimation of pre-pregnancy weight also contributes to overestimation of pregnancy weight gain, and of fat gain and weight retention, and falsely strengthens the relation between pregnancy weight gain and long-term weight (or fat) “retention.” Ohlin and Rossner (16) found that a large difference between reported pre-pregnancy weight and weight measured at about 10 weeks’ gestation—probably partly reflecting underestimation of pre-pregnancy weight—was linearly associated with what is often called “retained” weight postpartum. Although excessive fat gain early in pregnancy could also explain this relation in some women, most women do not appear to gain large amounts of fat early in pregnancy (see below). Thus, while high early pregnancy weight gain may contribute to postpartum weight retention, the effect of underestimation of pre-pregnancy weight will make many women falsely appear to have gained weight early in pregnancy and to have retained this weight after delivery. This may lead observers to believe that more women become obese because of pregnancy than actually do.
Variability of Pregnancy Weight Gain and Body Composition Changes
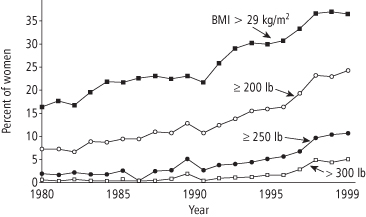
Pregnancy weight gain varies widely. Concern about pregnancy weight gain exceeding the recommended range is heightened by the increasing portion of women entering prenatal care already overweight or obese. These heavier women are those potentially most adversely affected by additional weight retained postpartum. The number of women entering prenatal care at a BMI >29 (obese) more than doubled in two decades, going from about 16% in 1980 to about 37% in 1999, as shown in Figure 10-1 (17). A more recent study of women in Massachusetts showed 49% overweight or obese by the 2009 guidelines (BMI > 25.0); 42% of these women would have been classified as overweight or obese by the 1990 guidelines (BMI > 26.0) (9). Considering that a significant portion of pregnant women exceed the weight gains recommended for them, with obese women more likely to exceed recommended gains (8), concern about obesity developing or worsening after a pregnancy is warranted.
Figure 10-1 Percentage of women with high weight or BMI above 29 at first prenatal visit, 1980 to 1999
Adapted from: Lu G, Rouse D, DuBard M, Cliver S, Kimberlin D, Hauth J. The effect of the increasing prevalence of maternal obesity on perinatal morbidity. Am J Obstet Gynecol 2001;185:845–9.
Pregnancy weight gain is due only partly to the products of conception. Increases occur in plasma volume and red cell mass, breast and uterine tissues, and body fat. Thus, differences in pregnancy weight gain do not reflect only differences in fat gain. To accurately identify factors raising the risk of postpartum obesity caused by pregnancy, the amount of fat gained must be estimated. Measurements of body composition using a multicompartment method (one of the best methods available) have demonstrated the dependence of fat gain on total weight gain (12).
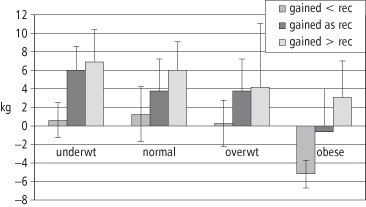
Figure 10-2 shows the increase in body fat between weeks 14 and 37 of pregnancy in 196 women delivering at term, classified by the 1990 IOM pre-pregnancy BMI categories and grouped according to weight gain within, less than, or more than the recommended range at that time (12) with 9 kg set as the upper limit for obese women. In 1990, the IOM had only set the lower limit (7 kg) for recommended weight gain for obese women. As shown, these weight gains were associated on average with some fat gain in all but obese women. In the study shown, obese women gaining 7–9 kg lost a small amount of fat on average. In all BMI categories, gains above the recommended range (or above 9 kg for obese women) were associated with greater mean fat gains, while gains below the recommended levels resulted in lower mean fat gains.
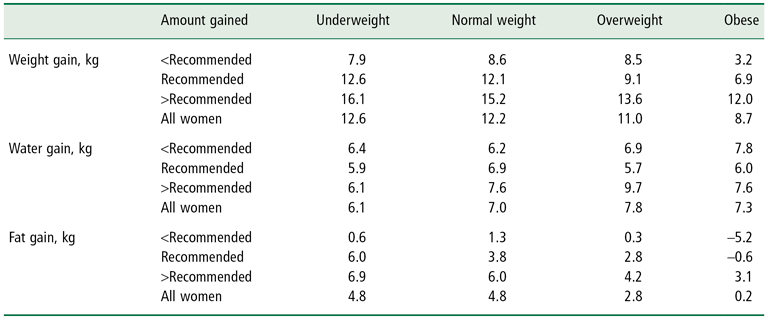
Table 10-2 shows how total weight gain related to fat gain and water gain between weeks 14 and 37 of pregnancy. Some increases are likely to have occurred before week 14. For example, a subsequent report (18) covered 63 women grouped as low, normal, or high BMI before pregnancy based on the 1990 IOM groupings. It showed that from before pregnancy through pregnancy week 9, average fat content increased by 0.4–2.1 kg fat in the three groups, with increases in average weight of 0.9–4.5 kg. The highest gains were in the high BMI group. Similarly, a study of 17 women in Sweden (19) showed a mean fat change of 0.8 kg with a weight change of 1.8 kg from pre-pregnancy to week 14. Thus, some fat gain occurs in the early pregnancy period which was not included in the study in Figure 10-2. Clearly, gains of a few kg of fat in early pregnancy could have an important effect on total fat gain.
Table 10-2 Body Weight, Fat, and Water Changes During Pregnancy in Women Classified by Pre-Pregnancy BMI Group and by Weight Gain Less than the IOM 1990 Recommended Gain, Equal to the Recommended Gain, or Greater than the Recommended Gain
Prospective studies have generally reported mean weight increases of no more than about 1 kg between pre-pregnancy and 6–12 months postpartum. A longitudinal study (20) of more than 2,000 women in the US examined five times between 1985–1986 and 1995–96 reported an average weight increase of 1 kg among normal weight women with pregnancy (whether completed or not) compared to similar women with no pregnancy. This increase occurred only after the first pregnancy and was greater (5–6 kg) in overweight women. A study of 1,423 Swedish women (16) observed that mean weight increased by 1.5 kg from pre-pregnancy to one year postpartum, an amount reduced to 0.5 kg when correction was made for the weight gain expected with aging and for estimated underestimation of pre-pregnancy weight. In this study, however, 14% of the women were heavier by 5 kg or more at one year postpartum than they were before pregnancy. In a review of weight changes across pregnancy, Gunderson and Abrams (21) estimated that, 6–18 months after delivery, up to 20% of women weigh an additional 5 kg or more than they did before pregnancy.
These data show that women may permanently add substantial weight, presumably fat, as a result of changes during pregnancy and the postpartum period, the amount partly depending on their pregnancy weight gain. What needs attention, however, is not simply pregnancy weight gain but excessive pregnancy weight gain and continued weight gain after delivery, as well as failure to lose weight postpartum. In many reports, more women gain excessively than within the recommended range. Lowering weight gain recommendations will not rectify adverse outcomes related to excessive pregnancy weight gain; further, lowering recommendations and even gestational weight gain will not prevent continued postpartum gain. To reduce the risk of maternal obesity, both excessive pregnancy weight gain and postpartum weight gain or failure to lose weight must be addressed, as they can be major contributors to developing obesity.
Effect of Maternal Obesity and Weight Gain on the Fetus
Relation of Maternal Weight Gain to Birth Weight; Birth Weight and Newborn Survival
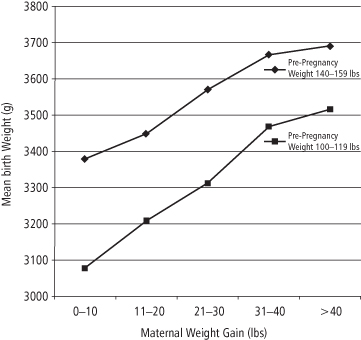
Normal fetal growth requires pregnancy weight gain. The consistently observed relation between pregnancy weight gain and birth weight in women of different pre-pregnancy weights was established decades ago (22) (see Figure 10-3). As shown, higher pre-pregnancy weight and weight gain are both associated with increased newborn weight.
Figure 10-3 Birth weight by maternal weight gain in women of high and low weight before pregnancy
(Adapted from reference 22)
Limiting pregnancy weight gain may result in lower maternal fat gain, as shown in Figure 10-2, but low weight gain (<0.27 kg/wk) is associated with an increased risk of low birth weight (<2500 g) and preterm delivery (RR = 1.22 and 1.11, respectively) (23), both of which are well-known risks for newborn mortality and suboptimal neonatal development. However, restriction of growth has effects across the whole birth weight spectrum, not just in low weight newborns. Improvement of fetal growth remains one of the goals of recommended pregnancy weight gain, and poor fetal outcome is a risk of inadequate gain.
Decades ago studies showed that infant survival improves as birth weight increases (24), with optimal early survival observed at what is typically considered high birth weight. For example, Rees and colleagues (25) classified birth weight in 500 g intervals using national data for 12.7 million births to adult White mothers delivering in 1983–87 and showed that minimum neonatal mortality occurred at birth weights of 3500–4499. Those with birth weights between 3000 g and 3499 g (which includes the US mean, 3316 g in 2004) (26) had a neonatal mortality rate 44% higher than these heavier infants. Seeds and Peng (27) reported similar results in a hospital population grouped into 250 g categories of birth weight. They reported a 50–300% greater mortality rate in birth weight categories near the US mean than in infants in the categories from 3500 to 4500 g.
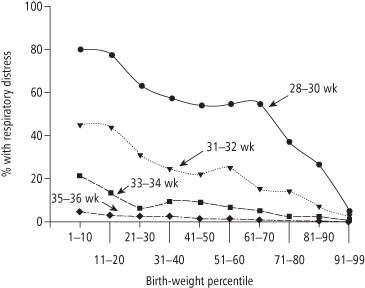
The survival advantage of high birth weight infants is not due solely to being born at term. Data show that, across the spectrum of gestational ages, those born larger for their gestational age have improved early survival even over those born at an appropriate weight for gestational age (AGA), with the poorest outcomes in those born both small for gestational age (SGA) and early (28). One large study (27) showed steady declines in neonatal mortality rates from the 10th to >95th percentiles of birth weight for gestational age. Furthermore, while data on respiratory distress syndrome (a major risk for the preterm newborn) show the expected decline in this condition with increasing gestational age in infants born between 31 and 36 weeks gestation (29), a dramatic decline was also observed at a given gestational age as weight increased from less than the 10th percentile through the 91st–99th percentile for gestational age (see Figure 10-4). Despite the extensive data demonstrating their lower risk, newborns weighing over 4000 g, or those above the 90th percentile of weight for age (who are often not actually large because they are born early), are often viewed as high risk and referred to as “macrosomic,” with numerous articles encouraging attempts to reduce such outcomes. In fact, US infant mortality would probably be substantially lower if the distribution of birth weights were raised and more infants had birth weights in the optimal high, or “macrosomic,” range.
Figure 10-4 Respiratory distress among preterm infants by birth weight percentiles and gestational age
(Adapted from reference 23)
Fetal Overgrowth and Diabetic Pregnancy
The common concern for newborns with a high birth weight probably has its roots in the historical association of serious neonatal problems with the high birth weight and high fat content of infants of diabetic mothers. When pregnancy weight gain is restricted, as was the case for much of the twentieth century, high birth weights are infrequent, but are particularly associated with uncontrolled diabetes during pregnancy. Maternal diabetes can increase passage of glucose to the fetus, inducing fetal insulin secretion. Insulin stimulates fetal growth and fat deposition. The fetus of a diabetic woman can grow both larger and fatter than if it had developed in a normal glucose environment. Even among well-controlled diabetic women, fetal fat content is increased (30).
A study of almost 280,000 births between 1991and 1993 (27) found a steady decrease in neonatal mortality through birth weights exceeding the 95th percentile of weight for gestational age compared to those in the 25th–75th percentiles. Fetal (intrauterine) mortality rates also declined steadily through birth weights of 4500 g for infants born to non-diabetic mothers, increasing slightly but not significantly at weights >4500 g. In contrast, a fetus weighing more than 4500 g had a more than six-fold greater risk of fetal mortality if its mother was diabetic than did a comparable weight fetus of a non-diabetic woman. Although infants of diabetic mothers constituted only 5.6% of infants weighing >4500 g, they contributed 27% of the deaths in that birth weight group, with a mortality rate of 14.5 per 1,000 births (27). Clearly, concern about infants exceeding 4500 g should be focused on those resulting from pregnancies complicated by diabetes, not on those resulting from hearty fetal growth in healthy mothers.
In recent years, the linking of diabetes and maternal obesity to fat content in the newborn, high birth weight frequency, cesarean section rates, and other adverse birth outcomes, has raised renewed concern, since diabetes prevalence during pregnancy has risen from 2.13 to 3.58 per 100 live births between 1990 and 2004 (26), and pre-pregnancy obesity has risen sharply, as shown in Figure 10-1. Both conditions are anticipated to continue to rise as the obesity epidemic continues. However, the risks associated with unrestricted fetal growth and resulting (healthy) high birth weight in the majority of pregnancies (which are still to non-diabetic, non-obese women) are not the same as the risks of high birth weight and excessive fetal fat deposition associated with diabetes or maternal obesity present before pregnancy. For example, maternal pre-pregnancy overweight and obesity result in a doubling of antepartum stillbirth, while weight gain in pregnancy is not related to stillbirth risk (31). The mechanisms by which diabetes and pre-pregnancy obesity reduce fetal and newborn survival are an area of intense research interest.
In summary, to identify the pregnancy weight gains that would optimize birth weight, it is important to distinguish between high birth weight resulting from normal fetal growth and high birth weight due to pregnancy pathology or pre-pregnancy maternal obesity. The recommendations for weight gain during normal pregnancy should not be determined by what occurs during a pathological pregnancy.
Relation of Birth Weight/Intrauterine Growth to Health and Development of the Offspring
Birth Weight and Developmental Health Outcomes
Across the spectrum, birth weights higher than the average have been positively associated with many beneficial outcomes in later childhood, in addition to the early survival advantages discussed above. Reported findings show increased birth weight associated with higher IQ at age 7 (about 2 pt/500 g between 2500 and 3999 g) (32); higher IQ at age 18, as well as higher earnings and educational attainment (33); improvement in a variety of cognitive measures at 8, 15, and 26 years of age, independent of many confounding social factors (34); and, independent of prematurity, decreased rates of postnatal hospitalizations for central nervous system viral infections, bacterial meningitis, respiratory infection, and viral or bacterial pneumonia, with a significant trend through age 10 years (35). Other studies show reduced susceptibility to stress in male conscripts, with increasingly greater protective effects up to 4200 g (36); a reduction in the odds ratio for depressive symptoms in women (37); and increased grip strength at age 53 (adjusted for earlier weights and heights) (38). Thus, the improved early survival observed in heavier infants is followed by an array of beneficial later outcomes. Nevertheless, these positive findings are not widely known or appreciated, and it is common for infants with weights in the healthy 3700–4500 g range to be viewed as high risk, rather than well-grown, while the poorer outcomes of “normal”, lower weight infants (those above 2500 g but below 3700 g) are rarely accorded concern.
Examples of how such misconceptions can misdirect our thinking are provided by two recent studies of weight gain during pregnancy. The first study (39) included pregnancies in both diabetic and non-diabetic women, and included birth weight above the 90th percentile for term births as one of the “adverse outcomes” in evaluating optimal pregnancy weight gain. The author concluded that weight gains lower than the IOM 1990 recommendations were optimal, in part because higher weight gain increased birth weight, which they assumed was undesirable. The second study, which included over 177,000 births to non-diabetic Bavarian women in 2004–6 (40), also treated SGA and LGA births (the 10th and 90th percentiles of birth weight in the reference population) as equally adverse. Ignoring the fact that SGA babies are at high risk while LGA babies are not, they examined weight gains associated with their joint predicted risk of ≤20%. They concluded that optimal weight gains for overweight and obese mothers were significantly lower than the IOM 2009 recommendations. They also suggested that weight loss during pregnancy might be beneficial for these heavier women because it could reduce the joint risk of LGA and SGA. No effort was made to distinguish the large infant of an obese mother from that of a normal weight mother, and longer range infant outcomes were not assessed. Such reports indicate why it is important that we examine carefully the many benefits to the newborn of being well-grown at birth, as well as the effects of pregnancy weight gain on maternal health.
Birth Weight and the Risk of Obesity and Chronic Disease
High Birth Weight
As shown above, newborn survival and later developmental outcomes of childhood are generally best in well-grown, heavy newborns. Nevertheless, questions continue to be raised as to whether longer-term health and survival are adversely affected by high birth weight. Although some studies report little or no association between birth weight and obesity in later life (41), some large cohort studies of female (42) or male (43) health professionals in the US, and of Danish (44) or Israeli (45) conscripts, have reported a relation between birth weight and later BMI (see 46 for a review). For example, Sorensen and colleagues (44) found a gradual increase in the mean BMI of young adults divided into eight birth weight groups ranging from ≤2500 g through ≥4501 g (Table 10-3). However, prevalence of adult obesity (BMI ≥ 30) in these birth weight groups was more variable, with similar low prevalence in those born at ≤2500 g and at 4001–4500 g. Nevertheless, the highest adult obesity rate was observed in those born ≥4501 g.
Table 10-3 Weight at Birth in Relation to Body Mass Index and Prevalence of Obesity at Around 20 Years of Age in 4,300 Subjects
Adapted from ref. (44).
| Birth weight (g) | Mean BMI | Prevalence of obesity, BMI ≥ 30 (%) |
| ≤2500 | 22.7 | 3.5 |
| 2501–3000 | 23.0 | 3.3 |
| 3001–3250 | 23.1 | 3.6 |
| 3251–3500 | 23.2 | 4.4 |
| 3501–3750 | 23.7 | 6.1 |
| 3751–4000 | 23.7 | 5.5 |
| 4001–4500 | 23.7 | 3.5 |
| ≥4501 | 24.8 | 11.4 |
One would need to distinguish children of obese women from children of women with diabetes, and examine differences in adult lean mass as BMI increased, to be certain how to interpret these data. Often BMI is treated as a perfect index of obesity or of body fat. Although variations in BMI may correlate with body fat in adults, that does not ensure that observed differences in BMI in selected groups are due to body fat differences (47). This may be especially true during the period of child growth. To determine the relation of birth weight to subsequent obesity, direct measures of adult body composition, both fat and lean, are necessary. But only a few studies make such measurements.
Elia and colleagues (47), using a four-compartment model to assess body composition at 6.5–9.1 years of age, showed a decrease in percentage of fat and in fat mass and an increase in lean mass, as birth weight increased. Similarly, Chomtho and colleagues (48) used a four-compartment model at a mean age of 11.7 years and found that in boys, but not girls, birth weight was positively related to later BMI and to the fat-free-mass index (fat-free-mass/ht2, analogous to body mass index, which is mass/ht2). However, birth weight was not associated with either the fat mass index (fat-mass/ht2) or with percentage fat in either boys or girls. Sayer and colleagues (49), using quintiles of birth weight ranging from <3062 g to >3969 g, have shown that at a mean age of 64 years those born heavier have a higher lean mass, not a higher fat mass, than those born at a lower weight. Thus, one cannot assume that high adult BMI reflects the same composition in those of different birth weights. Similarly, De Lucia Rolfe and colleagues (50) used DEXA and ultrasound measurements to determine fat content, and compared nine groups defined by their tertiles of birth weight and tertiles of adult BMI at ages 30–55 years. They found that in both the lowest and highest tertiles of birth weight, 34% of the infants were in the highest tertile for BMI as adults. Moreover, the higher birth weight infants who became high BMI adults had lower visceral fat as adults than lower birth weight infants with high adult BMI. It is worth noting that visceral fat is believed to be the fat store most determinative of adult chronic diseases such as coronary heart disease (CHD), glucose intolerance/diabetes, and the metabolic syndrome.
Other studies have examined the relation of birth weight to later lean mass. Phillips (51) reported that muscle mass, as determined from urinary creatinine excretion, was positively associated with birth weight in men and women. Differences were substantial, with adult muscle mass being only 18.8% (11.9 kg) of body weight in women weighing ≤2500 g at birth and rising to 24.7% (16.1 kg) in those with birth weights >3400 g, with similar trends in men. Non-muscle mass (which includes fat mass) was not associated with birth weight. Similarly, in a study of men and women in their 70s (52), which used dual-energy X-ray absorptiometry (DEXA) to determine adult body composition, birth weight was associated with later lean mass but not fat mass, with sex, age, and height controlled. When adult weight was controlled, the positive association of birth weight with lean mass was still observed and total fat mass was significantly negatively associated with birth weight, showing that lower birth weight resulted in higher adult fat mass at a given weight. About 25% of the variation in lean mass in both sexes was explained by birth weight and this relation remained significant with adult height, weight, and age controlled.
Interesting results also have been reported from a study in Iceland (53), where mean birth weight is high (3.8 kg in males and 3.7 kg in females). Quartiles of birth weight (lowest quartile, ≤3.45; highest quartile >4.0 kg) were significantly positively related to adult BMI. However, birth weight was not related to the odds of adult obesity (BMI ≥ 30 kg/m2) and was inversely related to truncal fat determined as the ratio of subscapular to triceps skinfold thickness.
Although higher adult BMI is associated with increased fat, and higher birth weight is associated with higher adult BMI, increasing birth weight is demonstrated not to be associated with increased adult fat content. Nevertheless, one might still ask whether increased birth weight per se increases later disease risk, even if not by way of increased adult fatness. Hales and colleagues (41) examined the relation of birth weight to several disease markers in men age 59–70. In this study, mean adult BMI showed no clear overall trend with increasing birth weight. Mean BMI varied between 26.4 and 26.8 in five of six birth weight groups which ranged up to 4309 g birth weight, and rose to 29.1 in those born heavier than 4309 g. Nonetheless, they adjusted (for BMI) their findings for various disease markers and found that the odds ratio for impaired glucose tolerance decreased steadily and significantly as birth weight increased, from an OR = 6.6 in those weighing ≤2495 g at birth to an OR = 1.0 in those weighing >4309 g. Systolic blood pressure also declined steadily and significantly from 173 to 161 mm of mercury across the same birth weight groupings. The decrease in fasting insulin levels with increasing birth weight was at the margin of significance (p = 0.06), but 2-hour insulin levels during a glucose tolerance test were significantly lower in those born with the highest birth weights, ranging from 224 pmol/L in the lowest birth weight group to 124 and 143 pmol/L in the two highest birth weight groups. Thus, birth weights above 4300 g were associated with lower blood pressure and the best results for markers of diabetes risk late in life compared to all the lower birth weight groups. Similarly, research from Iceland has shown that high birth weight in a population with low levels of maternal diabetes is not related to coronary artery disease (54) and is protective against adult high blood pressure and dysglycemia (53, 55–56).
Outcomes may differ in pregnancies characterized by diabetes, however. Boney and colleagues (57) report on the rate of development of the metabolic syndrome (obesity, dyslipidemia, hypertension, and glucose intolerance) in four groups of children, born either large or appropriate for gestational age (LGA or AGA), to mothers with or without gestational diabetes mellitus. At age 11 years, rates of obesity, defined for this study as BMI >85th percentile, did not differ in children born LGA of a non-diabetic mother compared to those born AGA. However, 15% of those born LGA to a mother with gestational diabetes manifested three or more components of the metabolic syndrome; rates were 3.0% to 5.3% for the three other groups, showing no elevation in LGA infants of women without diabetes. The risk of developing the metabolic syndrome between 6 and 11 years was not significantly different between LGA and AGA offspring in the non-diabetic group, but at age 11 was significantly different between LGA and AGA offspring of mothers with gestational diabetes, with a 3.6-fold greater risk among children born LGA. In Icelanders, maternal diabetes and glucose intolerance rates are low, mean birth weight is high (>3700 g), and the offspring’s risk of later diabetes is low (53). In contrast, among Pima Indians, maternal type 2 diabetes is common and high birth weight is associated with increased risk of diabetes in later life (56). These findings show that large size at birth is not adverse in itself, but it can be when it results from maternal pathology. The higher BMI of adults who were born at higher birth weight decades ago is largely due to increased lean mass. It remains to be seen whether this will remain true as an increasing proportion of high-weight newborns are those born to obese and diabetic mothers.
Low Birth Weight
An ever-growing body of literature has demonstrated that restricted intrauterine growth, whether manifested as low birth weight (<2500 g) or small size for gestational age, is associated with an increase in the proportion of body fat and associated chronic diseases in adulthood (58–60). These diseases include ischemic heart disease (61); coronary heart disease, particularly in those with low birth weight and high adult BMI (62); higher blood pressure in high-weight Philippine adolescents born low weight (63) in adults from five developing country cohorts with lower birth weight (64); and an increased systolic blood pressure at age 50 in Swedish men (65), where men in the top third of BMI at age 50 showed a 9 mm decrease in systolic blood pressure for each kg increase in birth weight. These observations are consistent with increased rates of hypertension, as well as stroke, type 2 diabetes, and coronary heart disease, in the Nurses’ Health Study (I and II) in women who were low weight at birth (42, 66, 67), as well as from data on men in the Health Professionals Follow-up Study (43). It is worth noting that 97% of the obese women followed in the Nurses’ Health Study I were not high birth weight babies, showing that high birth weight is not a major cause of obesity in the US (42).
Interestingly, postnatal weight gain early in life may matter, irrespective of birth weight. In a multivariable analysis of about 12,000 children, Stettler and colleagues (68) reported an odds ratio of 1.17 for risk of overweight at age 7 years with increased rate of weight gain in the first four months of life, which was independent of birth weight. Similarly, in a study of term infants born with appropriate weight for gestational age, accelerated growth in the first two years of life was associated with increased BMI and increased body fat percentage at age 7 years (69). Low birth weight or small size for gestational age appears to particularly raise the risk of later obesity (70), especially when it is followed by rapid postnatal growth, which seems to contribute to increased central or truncal fat mass with lower muscle mass (59, 71). Similar findings have been reported for infants born very preterm (before 32 weeks’ gestation), where increased birth weight (although birth weights would still be very low) was not associated with percentage body fat determined from four skinfold thicknesses at age 19 years, although it was associated with adult height, weight, fat mass, and BMI, indicating that increased birth weight was related to increased overall body size at age 19 years (72). However, in this study rapid weight gains early or late in infancy were associated with a higher percentage body fat and with an abdominal pattern of fat distribution, in agreement with the other studies mentioned.
The relationship of chronic diseases to the combination of low birth weight followed by rapid postnatal growth supports the importance of avoiding growth restriction in utero and raises questions, as yet unanswered, about the effects of promoting “catch-up” growth in early childhood in those born low weight. Although at least part of the association between low birth weight and adult overweight may be due to social class (since both are more common in groups with low socioeconomic status), recent studies that include measures of social class suggest that this is not a decisive factor (58).
The overall conclusions that can be drawn from these combined studies are that the well-grown newborn has both short- and long-term health and survival advantages. Infants whose growth is pathologically accelerated by maternal diabetes and possibly by obesity can have different outcomes. They should be considered separately when assessing risks or developing weight gain recommendations.
Lactation
The Effect of Maternal Obesity on Lactation
It has long been thought that production of breast milk should assist in the postpartum loss of weight gained during pregnancy. Full, exclusive breastfeeding is estimated to require 2.62 MJ/day (626 kcal/day) (73), part of which is expected to come from the mobilization of maternal fat (0.72 MJ/day, or 172 kcal/day), particularly in well-nourished women. Therefore, long and full lactation should be most desirable in overweight and obese women, who are likely to be those most adversely affected by retaining fat gained during pregnancy and who might be least able otherwise to regulate their weight. Thus, it is particularly interesting that, compared to normal-weight women, obese women have been shown to be less likely to successfully initiate breastfeeding and to breastfeed for shorter durations if they do initiate (74–75). Higher maternal BMI is associated with a delay of lactogenesis II, the time of onset of copious milk production (74). Subsequent work has demonstrated that, during suckling at 48 hours postpartum, women with a BMI > 26 kg/m2 secreted less prolactin (a hormone essential for successful lactation) than lower-weight women. Further, in multivariate analyses adjusting for time since delivery and the duration of the nursing episode, only maternal overweight or obesity remained significant predictors of lower prolactin secretion during suckling at day 7 (75). This is the key period for the establishment of lactation and these altered hormonal responses in overweight women are likely to play a role in their increased failure to sustain breastfeeding. In addition, excessive (or inadequate) pregnancy weight gain, as defined by the IOM guidelines, in women of any weight status has also been shown to be associated with a shorter duration of breastfeeding (76).
The Effect of Lactation on Postpartum Weight Changes
The effect of lactation on maternal body weight has been examined in many studies comparing lactating and non-lactating postpartum women. Early in the postpartum period, a loss of body weight appears to be physiological and natural among lactating women, even if they are not consciously dieting, but the usual rate of weight loss is slow. On average, lactating women lose 0.2–0.8 kg/month during the first six months, excluding the immediate postpartum period (77–80). During the next six months, weight loss is slower, 0.1–0.2 kg/month (78, 81–82).
However, well-nourished lactating women have been consistently reported to consume 200–800 kcal per day more than comparable formula-feeding women (77, 83–85). Formula feeders who successfully lose weight postpartum consume significantly fewer calories than do breast-feeders who lose weight. In some studies, the formula feeders actually lose weight more quickly than breast-feeders during the early months after delivery (77, 83, 86). Dewey and colleagues (78) compared weight changes in lactating and non-lactating postpartum women selected because they were not dieting. Non-dieting breast-feeders lost weight more quickly than non-dieting formula feeders, starting at four months postpartum. By six months postpartum, breast-feeders had lost about 2.8 kg more than the formula feeders and had returned to their pre-pregnancy weight by one year postpartum. Formula feeders stopped losing weight by nine months postpartum and on average remained about 4% above their pre-pregnant weight at two years postpartum. These combined results indicate that breast-feeders lose more weight more quickly than formula feeders, unless the formula feeders are significantly more calorie restricted.
Other work shows that increased energy expenditure per se does not accelerate postpartum weight loss in lactating women unless dietary intake is controlled (87). An integrative review of such studies (88) concludes that, early in lactation, women who are not consciously limiting food intake will increase calorie intake rather than more quickly mobilizing the fat they stored during pregnancy, and will also compensate with increased food intake for the additional energy expenditure of increased exercise. Later in lactation, these biological controls seem to be less strong, so that weight and fat loss continue even though body fat has declined. It appears that to be most effective for body fat and weight reduction, breastfeeding must be of high intensity or exclusive, continued through most of the first year after delivery, and combined with some dietary control.
However, prolonged full lactation is practiced by relatively few women in the US. For example, only 46% of US mothers report exclusively breastfeeding at hospital discharge, with 17% continuing do so at six months (89), as recommended for all women. In a study of National Immunization Survey data from 2002 (90), maternal reports indicated that 71.4% of the children had ever been breastfed. At three months, 51.5% were reported as being breastfed to some extent, with 42.5% exclusively breastfed. At six months, these rates had dropped to 35% and 13%, respectively. At one year, only 16% of infants were receiving any breast milk. Similar results were obtained from retrospective data from the National Survey of Children’s Health for 2003, with higher rates among immigrant women than among “native” women (91). Thus, full lactation is practiced by a minority of women, even early in the first postpartum year. In the second half of the first year postpartum, it is even less common. Thus, prolonged, full lactation does not appear to be playing a major role in postpartum weight reduction for US women; this is probably even truer for overweight or obese women who would most benefit from increased energy demands of full and prolonged lactation, but who are least likely to breastfeed exclusively or for a long period, as discussed above. Despite these limitations, if more women were to breastfeed their infants according to current recommendations, lactation would make a valuable contribution to reducing the risk of maternal obesity development associated with reproduction.
Breastfeeding and Obesity Development in the Offspring
Several studies in the last decade suggest that breastfeeding reduces the risk of later obesity development in the child. Grummer-Strawn and Mei (92) showed that non-Hispanic White infants breastfed for 6–12 months had an adjusted odds ratio of 0.70 for overweight at age 4 years, with an even lower odds ratio (0.49) among those breastfed for more than 12 months, compared to those never breastfed—a reduction in overweight of more than 50%. Breastfeeding for any duration was also protective against underweight (BMI for age <5th percentile).
Bergmann and colleagues (93) showed that formula-fed children had higher BMIs and greater skinfold thickness (SFT) by three months of age. By six months, a higher percentage of the formula-fed infants exceeded the 90th and 97th percentiles for both skinfold thickness and (age- and sex-specific) BMI. In multivariate analyses adjusted for maternal overweight, smoking in pregnancy, and social status, formula feeding remained a significant predictor of overweight and obesity (by BMI percentiles) in the child at age 6 years. In this study, breastfeeding was found to be protective, with an odds ratio of 0.53 for overweight and 0.46 for obesity in those who were breastfed to any extent for three months or more. Gillman and colleagues (94) studied healthy children at age 9–14 years and examined the relation to initiation and duration of breastfeeding of numerous economic, social, biological, and behavioral factors, all based on data reported by the child or the mother. After controlling for many factors predictive of breastfeeding or overweight (>95th percentile for age and sex), they found that, compared to those mostly formula fed, children who had been mostly breastfed in the first six months had an odds ratio of 0.78 for being overweight later. Similarly, with respect to duration of breastfeeding, the odds ratio for overweight was 0.80 when comparing those breastfed six months or more to those mostly or only formula fed.
In a meta-analysis of 17 studies, Harder and colleagues (95) found a dose-response relation between duration of breastfeeding and reduced risk of overweight (defined by BMI in most studies, with follow-up of various lengths, mostly into later childhood) with odds ratios of 1.0 (no effect) for <1 month of breastfeeding, 0.81 for 1–3 months, 0.76 for 4–7 months, and 0.67 and 0.68 for 7–9 and >9 months, respectively. The odds ratio for overweight was 0.96 per month of breastfeeding, indicating a 4% reduction in risk for each month of breastfeeding. Demonstration of an increased benefit with longer breastfeeding duration supports the interpretation that it is the feeding method that is responsible for the effect and not other factors, such as social class, associated with both obesity and breastfeeding rates.
There are a few common criticisms of these works. Specifically, factors known to be associated with both increased breastfeeding and decreased obesity rates (such as higher maternal education and SES and lower BMI) may not be completely controlled for; data on breastfeeding may not be accurate when recalled years later; and, most importantly, changes in BMI may not represent differences in body fat. A recent study (96) largely avoided these problems. Over 12,000 non-pregnant women in Southampton, England, aged 20–34 years, were followed, with home interviews and measurements taken. Additional data were collected during and after pregnancy from those who became pregnant (1,195 births), with breastfeeding duration determined at home visits at 6 and 12 months postpartum. DEXA measurement of the child’s body composition at age 4 years was completed in 539 study children. Almost 88% of the study women initiated breastfeeding, reducing the effects of selection into the breastfeeding groups. At aged 4 years, fat mass and fat mass index (fat mass/ht2) showed significant decreases (p = 0.002, 0.001, respectively) with increased duration of breastfeeding through age 1 year. Lean mass and lean mass index (lean mass/ht2) were not associated with duration of breastfeeding. BMI showed a non-significant (p = 0.059) trend downward with increased duration of breastfeeding. The analyses for fat mass, fat mass index, and BMI controlled for the mother’s age, her measured BMI and height, education, social class, and smoking in late pregnancy, as well as infant birth weight and age at introduction of solid foods, variables often thought to be associated with both obesity development and breastfeeding duration. These results indicate that breastfeeding results in a lower fat mass, without corresponding reductions in lean mass or BMI. This conclusion is supported by work examining growth rates at different intervals in the first six years (97). Infants breastfed for at least four months showed slower increases in weight and BMI, but no difference in growth in length across this time period.
Nevertheless, not all studies support these effects of breastfeeding. A study of children born in Belarusian hospitals (98) examined the effects on the child of a hospital intervention that dramatically increased breastfeeding rates (exclusive breastfeeding at age three months was 43.3% for the intervention group and 6.4% for the control group). It showed no reduction in BMI, stature, skinfold thicknesses, or waist or hip circumference at age 6.5 years in those exposed to the intervention compared to those born in control hospitals. However, these body measures are not as specific as the DEXA measurement of body fat and may have missed important fat differences. In addition, overall, the observed rates of childhood obesity in Belarus were much lower than in the US. Thus, it is possible that breastfeeding acts on pathways of importance for determining child fat content in the US, where obesity rates are high, while such pathways may not have been important in Belarus.
Underestimation of Effects of Breastfeeding
In assessing the relationships between breastfeeding and maternal weight changes or health outcomes of the child, the potential influence of measurement problems should be considered. Breastfeeding data are often obtained from reports given years after the breastfeeding period. Duration estimates are subject to rounding to 3-month intervals and estimates of breastfeeding often fail to differentiate among practices that range from exclusive breastfeeding to occasional breastfeeding with substantial use of formula, juice, water, cereal, etc., all of which can reduce the benefits of breastfeeding. As a result, most studies do not observe the true and full effects of breastfeeding and thus underestimate important associations. Considering the difficulty of getting accurate retrospective assessments of breastfeeding practices as well as the inaccuracy of reported weights and heights of the offspring, the consistency of the studies is impressive. If overweight in the young adolescent could be decreased by as much as 20–25% nationwide by increased breastfeeding rates in infancy, breastfeeding would probably be the single factor with the greatest potential for prevention of obesity development later in life. One can only wonder whether the nadir in breastfeeding rates that was observed in the late 1960s and early 1970s (99) was a factor in the rise in obesity rates two decades later.
Summary
The literature on obesity and reproduction indicates that pregnancy is followed by unwanted, long-lasting increases in body weight in some women. Excessive weight gain during pregnancy (more than recommended) raises the likelihood that a woman will maintain an increased weight and BMI at one year postpartum compared to before pregnancy. Overweight and obese women are more likely to gain excessive weight during pregnancy than lower-weight women. While this partly reflects the low weight gains recommended to them, they have a greater need than normal-weight women to avoid further weight gain. More needs to be done to assist women to gain weight as recommended. In addition, more information is needed on the optimal pregnancy weight gains for the mother and baby, when the mother is severely obese.
Adequate pregnancy weight gain as defined by the IOM is important for the optimal growth and postnatal development of the infant. Infants born small for gestational age are more likely to develop a central fat distribution pattern in adulthood, a pattern associated with adult obesity and increased rates of several associated chronic diseases. High-weight newborns have higher lean mass in adulthood than infants born at low weights who attain the same adult BMI. The risk of adult obesity, and its associated chronic diseases, is lower in infants who are large for gestational age if they are not the product of a diabetic pregnancy. We do not yet know if this is also true for the large infant of a very obese mother.
The recommended pattern of infant feeding, which involves exclusive breastfeeding for the first six months postpartum and continued breastfeeding with provision of complementary foods for the remainder of the first postpartum year (and beyond, if desired), appears to help women lose fat gained during pregnancy. Longer durations of breastfeeding also are associated with reductions in the child’s risk of development of obesity in adult life. Obese women are less likely to successfully initiate breastfeeding, and those who do breastfeed terminate breastfeeding earlier than non-obese breastfeeders, suggesting a need for special interventions to assist breastfeeding in obese women.
In conclusion, to optimize her own postpartum body weight and the weight of her offspring, a woman should make efforts to avoid obesity before reproduction, avoid gaining more than recommended during pregnancy, focus on reaching an appropriate body weight by 6–12 months after delivery, and aim to breastfeed exclusively for as much of the first postpartum year as is possible.
Summary: Key Points
- Obese women have poorer pregnancy outcomes from the time of implantation through delivery and lactation, and their children are at higher risk of long-term suboptimal health.
- In the last few decades, an ever-increasing number of women have been entering pregnancy already overweight or obese, and more manifest diabetes during pregnancy.
- Weight gain recommendations for pregnancy have been established by the IOM for underweight (12.5–18 kg), normal weight (11.5–16 kg), overweight (7–11.5 kg) and obese women (5–9 kg), based on their pre-pregnancy BMI. These recommend gains limit fat deposition in obese women and normalize body fat in underweight women, but most women do not gain as recommended.
- Pregnancy can raise obesity risk if women gain more weight than is recommended during pregnancy, fail to lose sufficient weight postpartum, or gain weight postpartum due to lifestyle changes.
- Higher pre-pregnancy weight and greater pregnancy weight gain both contribute to increasing infant birth weight.
- High birth weights, from 3500 to 4500 g, are associated with lower body fat in adulthood, and many other good short- and long-term outcomes for the infant, unless the mother was diabetic. High weight infants of diabetic mothers have higher body fat, mortality, and other poorer short- and long-term outcomes than high weight infants of non-diabetic women.
- Increased breastfeeding intensity and duration are associated with lower rates of subsequent obesity in the infant; breastfeeding also helps weight loss in the lactating woman.
- Obese women have a lower rate of initiation of successful breastfeeding and shorter breastfeeding durations.