Fig. 20.1
(a) Temporal changes in the lower limb motor function in model mice with spinal cord injury. Significant improvement of lower limb motor function was observed by using the HINT treatment method. (b) Tracer experiment results of pyramidal tract primary neurons with biotinylated dextran amine. Re-elongation of severed axons was not observed despite the significant lower limb motor function improvement by the HINT treatment method. (c) With the administration of valproate, the differentiation of transplanted GFP-positive neural stem cells into MAP2-positive neurons was promoted, whereas differentiation into GFAP-positive astrocytes was inhibited
Because the transplanted neural stem cells are involved in improving motor function, we first examined the differentiation trends. On the basis of immunohistochemistry analyses, we observed that with the HINT method, neuronal differentiation of transplanted cells was greatly enhanced in the treatment group (Fig. 20.1c). Many of the neuronal projections derived from transplanted cells extended to the cranial side and the caudal dorsal funiculus (pyramidal tract area) as well as to cells near the anterior cornu of the spinal cord. Furthermore, using electron microscopy, we observed neurons derived from transplanted cells had received axons from the host neurons, then projected to cells on the anterior cornu of the spinal cord, and finally formed synapses.
Next to examine the route the damaged neuron takes, we used adenoviral injection to forcibly express wheat germ agglutinin (WGA) tracer that is passed from the primary to the tertiary neuron through the synapse into the motor cortex area. Surprisingly, in the HINT treatment group, the WGA had travelled from the cerebral cortex to the primary motor neurons and transported to transplanted cell-derived neurons. We also observed that WGA had travelled from the lesion to the peripheral side of cells on the anterior cornu of the spinal cord (Fig. 20.2a, b).
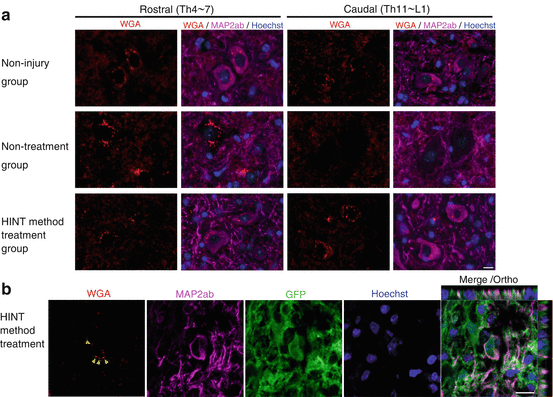

Fig. 20.2
(a) Evaluation of transsynaptic neural network regeneration using WGA. In the nontreatment group, WGA-positive neurons were almost nonexistent on the caudal side of the central injury location; however, in the HINT treatment method group, WGA-positive neurons were observed on the caudal side. (b) WGA-positive cells were observed in the neurons derived from transplanted cells in the vicinity of the central injury location
Because host neurons were largely unobserved in the region around the lesion, it was strongly suggested that neurons derived from transplanted cells regenerate the damaged neural network in a relay fashion [14]. Moreover, to specifically remove the transplanted cells after treatment, we transplanted neural stem cells expressing the diphtheria toxin receptor. Once a treatment effect was observed, we administered diphtheria toxin to the mice (Fig. 20.3). We used an in vivo imaging system to verify that only transplanted cells were removed. By following the survival of transplanted cells over time, we observed that lower limb motor function again deteriorated to the same extent as in the nontreatment group. These data indicate that transplanted cells that undergo directed differentiation with valproate are directly involved in the improvement of lower limb motor function [14].
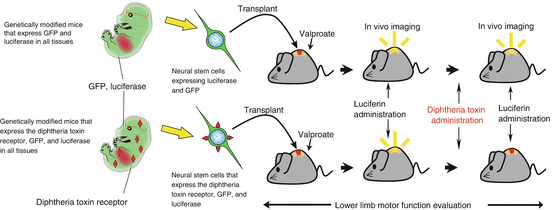

Fig. 20.3
Specific removal of the transplanted cells and observation of transplanted cell survival using in vivo imaging system. By the evaluation of specific removal of luciferase containing transplanted cells and observation of transplanted cell survival using in vivo imaging systems, it is possible to evaluate the amount of transplanted cells surviving in the same host. At the same time it is possible to specifically remove transplanted cells using the diphtheria toxin from a live host through the expression of the diphtheria toxin receptor on stem cells. Transplanted cells were evaluated by analyzing lower limb motor function whether or not they were directly involved in lower limb motor function improvement
20.3 Discussion
Recently, we have begun to understand the importance of molecular epigenetic mechanisms in the maintenance of cell differentiation and differentiation traits, including cell reprogramming. In this study, through in vivo differentiation of transplanted neurons, we have succeeded in regenerating the neural network of the host that was damaged. We directed in vivo differentiation of transplanted stem cells to neuronal lineage by changing the chromatin structure to facilitate histone acetylation.
Following our results, Bonner et al. also succeeded in regeneration by forcing relay connections using fetal neural progenitor cell-derived neurons of the sensory neural network, which were destroyed by spinal cord injury [15]. The use of neurotrophic factors in axon guidance is very interesting. In addition to regenerating the damaged circuits by neurons derived from transplanted neural progenitor cells rather than host-derived cells, the development of complex therapies such as this will continue in future. Lu et al. transplanted a fibrin matrix containing neurotrophic factors and rat fetal-derived neural stem cells to the damaged portion of the rat spinal cord and dramatically improved lower limb function [16]. Similar to our results, transplanted cells derived from neurons extended many long projections and formed synapses with the neurons of the host. Taken together, these data showed that the proposed neural network reconstruction mechanism of the relay connection by neurons derived from transplanted cells, in fact, exists.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








