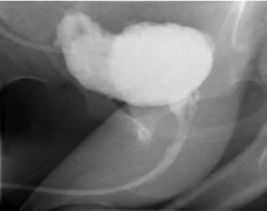
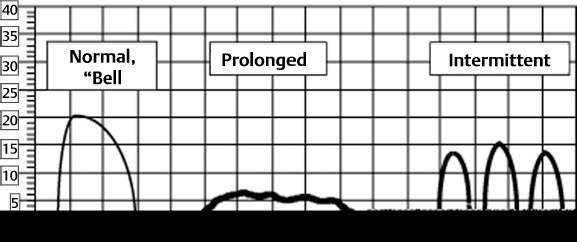
8 Urological Aspect of Tethered Cord Syndrome II: Clinical Experience in Urological Involvement with Tethered Cord Syndrome Antoine E. Khoury With the acceptance of tethered cord syndrome (TCS) as a true and important medical problem a multidisciplinary approach has allowed us to gain a better understanding of its pathophysiology and the complex ramifications of this anomaly on other organs and systems. Along with other manifestations such as lower extremity sensory and motor disturbances, the lower urinary tract (LUT) shares the potentially affected nervous system pathways and is commonly involved. The abnormal interplay between bladder and urethra, along with abnormalities in urine storage and elimination, leads to changes that may have significant effects in patients’ quality of life and that could also lead to irreversible kidney damage. The impact on the urinary tract ranges widely depending on the timing of the diagnosis, the severity of the process, and the other associated neurological problems besides the tethered cord. The common physical feature among these patients is the abnormal anchoring of the spinal cord at its end by an inelastic structure.1 As shown in experimental models, the lumbosacral cord is markedly susceptible to hypoxic stress generated by constant or intermittent traction between the attachments of the lowest pair of dentate ligaments and caudal fixation.2,3 These metabolic and electrophysiological changes are at least in part due to stretch-mediated distortion of vascular structures, with progressive impairment of oxidative metabolism,2 and are prevented or improved by surgical release of the cord, which halts the noxious injury and possibly facilitates neuronal reparative mechanisms. The nerve cells located in the gray matter of the tethered spinal cord have a higher metabolic demand than surrounding axons.3 This and the fact that the conus medullaris is most vulnerable to traction because of its vicinity to the caudal fixation point may explain the sometimes early and isolated appearance of bladder dysfunction as the initial manifestation of a tethered spinal cord. It also raises the possibility that a minimal degree of traction applied to the cord can result in the aforementioned impaired oxidative metabolism and electrophysiological changes without actual displacement of the cord caudally. The progressive deterioration of symptoms and signs in patients presenting with primary incontinence or the appearance of secondary incontinence may presumably be the result of repeated and/or longstanding metabolic insults. These include further intermittent cord stretching by flexion-extension of the spine, relative systemic hypoxia during strenuous exercises, local hypoxia secondary to venous congestion caused by Valsalva maneuver or abdominal straining, or during periods of growth spurts, which may aggravate the traction on the cord because of disproportionate elongation of the vertebral column. TCS presents to the urologist as (1) patients with a history of open spinal dysraphism, (2) patients with urinary tract dysfunction as part of a constellation of other neurological or orthopedic abnormalities, (3) children with isolated refractory LUT symptoms, (4) asymptomatic children with lumbosacral cutaneous abnormalities, or (5) patients with other abnormalities associated with an increased risk of TCS. Even though there may be some overlap, following are highlights of the clinical characteristics of these groups: The clinical findings and symptomatology that patients present with is secondary to the stretch-induced functional disorder in the lumbosacral cord. Therefore, the examination should be directed toward motor and sensory deficits in the lower limbs, urinary or fecal elimination problems, and musculoskeletal deformities.12 Some of these symptoms may be difficult or even impossible to evaluate because urinary and fecal incontinence is part of normal development up to a certain age. Urinary dribbling or incontinence, sensation of incomplete emptying, urinary frequency, recurrent urinary tract infections, difficulty initiating or sustaining the urinary stream, fecal incontinence, constipation, unexplained lower extremity pain or pain that is triggered/aggravated by flexion-extension of the spine or prolonged sitting, and propensity to stumble or fall should all be investigated. Intractable or severe constipation must not be overlooked because it has been determined that up to 9% of these patients may have spinal cord abnormalities, most commonly TCS.13 From a urological perspective, it is important to develop a high index of suspicion in patients presenting with secondary incontinence (i.e., incontinence that presents in a child previously toilet trained for > 6 months) and refractory primary incontinence. The early diagnosis and detection of an underlying tethered cord as the responsible etiological factor for incontinence could potentially result in complete reversal of the bladder dysfunction, especially when corrected promptly. When a neurogenic cause for the incontinence is suspected, a careful physical evaluation should be performed. The lumbosacral area should be examined for deformities and for any of the stigmata of spina bifida occulta that are associated with a tethered spinal cord. The lower extremities should also be evaluated for discrepancy in size, presence of muscle wasting, foot deformities, or foot drop. Some findings on physical exam, such as lower-extremity hyperreflexia or spasticity,3 should raise the possibility of associated higher cord lesions (i.e., syringomyelia, tumors, multiple sclerosis). As part of the initial evaluation a urinalysis and urine culture are obtained to rule out any other underlying abnormalities, such as a urinary tract infection, that may be responsible for the patient’s symptoms. Patients suspected of having an underlying neurogenic cause for their urinary incontinence are usually screened with a lumbosacral spine x-ray. It has been suggested that a carefully evaluated plain x-ray of the lumbosacral spine may help detect patients with complicated enuresis who deserve a more extensive evaluation.14 This test may also help establish the possibility of neurogenic bowel by disclosing a significant amount of fecal material in the colon. Radiological evaluation of the LUT is typically obtained by voiding cystourethrography. Even though invasive, a great deal of information may be gained from this relatively inexpensive study. A voiding cystourethrogram (VCUG) is ordered to study the posterior urethra in males (for evidence of posterior urethral valves or stricture), the bladder neck (for elevation and hypertrophy), the presence of vesicoureteral reflux (VUR), the thickness of the bladder wall, the presence of trabeculation or diverticulae, and changes in the shape of the bladder, such as elongation as seen classically in the “Christmas tree” form that signifies a neurogenic bladder (Fig. 8.1). Under the age of 3 months, ultrasonography of the spinal canal is the screening investigation of choice for the evaluation of infants suspected of having TCS. Abnormal findings include a low or blunt conus medullaris, a thickened or fatty filum terminale, fixed dorsal position of the cord in the thecal sac, and lack of pulsatile movement of the cord.9 As the child’s spine undergoes normal skeletal maturation ultrasonography quickly looses its usefulness for this purpose.15 If ultrasonographic examination is abnormal or technically inadequate, magnetic resonance imaging (MRI) is indicated as an accurate secondary examination to clearly define the malformation. Fig. 8.1 Voiding cystourethrogram (VCUG) on a patient with neurogenic dysfunction. Note abnormal bladder contour and shape. The diagnosis of TCS is primarily based on neurological and musculoskeletal signs and symptoms. Imaging features are in general obtained to support rather than make the diagnosis.3 MRI has revolutionized the noninvasive evaluation of spinal cord lesions. The major obstacle to obtaining an MRI in children is the need for sedation or general anesthesia. Patients with tethered cord have displacement of the conus and elongation of the cord, with a sometimes thickened or fatty density in the filum. Proper imaging not only helps establish the diagnosis but also allows for identification of associated anomalies before and after treatment (such as scar formation, tumors, syrinx, or bony abnormalities within the spinal canal). Urodynamic testing allows for a functional evaluation of the LUT. A full urodynamic evaluation is obtained in those patients with a known tethered cord who are referred for urological assessment or in an incontinent patient when an underlying neurological etiology is suspected. To understand and apply the results of this test, a comprehensive understanding of the LUT physiology is required. The LUT is composed of the bladder (including the ureterovesical junction area), the urethra, and the pelvic floor/urinary sphincter mechanism. The detrusor muscle is responsible for storage and voiding, whereas the urethra is responsible for control and conveyance of urine for elimination. The normal bladder should allow for low-pressure storage of urine delivered from the kidneys via the ureters. Normal innervation of the LUT allows for this phenomenon to occur. The efferent pathways consist of parasympathetic fibers from the sacral spinal cord (S2, S3, and S4), sympathetic fibers from the thoracolumbar cord (mainly T12, L1, and L2), and somatic efferents, including the pudendal nerve, which innervates the striated sphincter muscle. Together they promote detrusor muscle contraction or relaxation with coordinated sphincter activity, a process that involves different neurotransmitters, including acetylcholine, norepinephrine, and nitric oxide. The afferent sensory nerves from the bladder transmit information via the lumbosacral spinal cord through axons in the pelvic, hypogastric, and pudendal nerves. Their proper function senses bladder fullness, responds to changes in bladder wall stretch, and triggers nociception to overdistention. The fiber types involved (A and C fibers) have recently sparked interest in new concepts for managing neurogenic states, yet to be described here. The complex innervation of the bladder and urethral control mechanism aims at achieving low-pressure storage and periodic voluntary complete expulsion of urine. As proposed by Wein,16 this can be better understood by dividing the LUT function into two phases: filling/storage and emptying/voiding. Normal function occurs by accommodating increasing volumes of urine at low pressures, with appropriate sensation, in the absence of involuntary contractions, and with a closed bladder outlet (i.e., the bladder should be compliant, capacious, and stable). Storage pressures higher than the net glomerular filtration pressures are associated with a detrimental effect on renal function, deterioration of upper tract morphology, interference with ureteral transport of urine to the bladder, and VUR. Bladder emptying, on the other hand, must involve a coordinated contraction of the detrusor muscle that is of adequate magnitude and duration to effectively empty the bladder, with an appropriate decrease in resistance at the bladder outlet level without distal functional or anatomical obstruction. The urethral control mechanism should be coordinated with the detrusor muscle. Normally, sphincteric relaxation slightly precedes detrusor muscle contraction, which in turn causes continued sphincteric relaxation. This is termed by some as the detrusor-sphincter reflex and is sacral cord mediated. The urodynamic evaluation of the LUT consists of three basic studies: (1) urinary flow rate and estimation of PVR urine; (2) cystometrography; and (3) outlet resistance and coordination.17 In conjunction, this functional assessment aims at evaluating urine storage (by addressing the “3 Cs”: capacity, compliance, and contractility), voiding function, and bladder outlet activity. In selected cases the addition of fluoroscopic realtime imaging along with the use of hydrosoluble contrast media allows for evaluation of anatomical features of the LUT and may aid in better understanding of the functions of the LUT, specifically the interaction between the bladder and its outlet. A urinary flow rate (or uroflow) is the initial phase of urodynamic testing. It consists of a voiding study that plots the volume of urine expelled from the urethra per unit time and is expressed in milliliters per second.17 The main parameters evaluated are the maximum flow rate (Qmax), the voided volume, the flow time, and the shape of the curve or flow pattern. Peak flow rates are graphically displayed against the voided volume and then plotted on a nomogram, allowing for estimation of normal values based on the patient’s age and sex. The PVR urine volume can then be measured by bladder ultrasonography or catheterization. The Qmax should be attained in the first one third of the voided volume, and the uroflow curve should be uninterrupted, “bell-shaped,” leading to complete bladder emptying (see Fig. 8.2). A normal uroflow curve with an adequate voided volume and with minimal or no PVR urine indicates that the bladder capacity is satisfactory and that the coordination between the detrusor muscle and the urethral control mechanism is likely normal. It also excludes any significant functional or mechanical outflow obstruction. Intermittent, fractionated, or “staccato” flow patterns, prolonged “plateau” curves, and persistently elevated PVR volumes (i.e., > 10% of the estimated bladder capacity), should be considered abnormal in most clinical settings.17
Clinical Presentation of Urological Dysfunction Secondary to Tethered Cord Syndrome
Patient Evaluation History and Physical Exam
Lumbosacral X-ray and Voiding Cystourethrogram
Ultrasonography
Magnetic Resonance Imaging
Role of Urodynamics in Evaluating Lower Urinary Tract Function
Evaluation of Voiding Function
Urinary Flow Rate and Estimation of Postvoid Residual Urine
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







