Reconstructive techniques
Microsurgical clipping
Endosaccular coil embolisation
“Neck bridging devices”
Balloon-protected coil embolization
Stent-protected coil embolization
Flow-diverting devices
Deconstructive techniques
Parent artery occlusion [PAO]
PAO under bypass protection
Flow modification techniques
Reconstructive endovascular treatment of intracranial aneurysms [9] has become an established technique [12, 23–25] that can provide stable permanent occlusion in up to 85 % of the cases [2, 4, 5, 37, 38]. The angiomorphology of aneurysm recurrences after initial endovascular treatment is an important parameter to distinguish unstable aneurysm residuals that are potentially prone to hemorrhage from benign non-progressing lesions and thus to identify those patients who will need further treatment. A significant number of patients with minor non-progressing aneurysm remnants is followed angiographically, and there is growing evidence indicating that these are benign lesions. In those cases in which relevant aneurysm recurrences are documented upon angiographic follow-up, both endovascular [4, 25, 31, 37] and surgical techniques [4, 10, 17, 24, 28, 33, 39, 40] can be employed. In rare cases, combined treatment strategies including, for example, PAO under bypass protection [11, 13, 15, 16, 35, 41, 42], can be performed.
Patients and Methods
We recently described our experience in the combined endovascular and surgical management of patients suffering recurrences after both surgical and endovascular aneurysm treatment [6]. All patients were managed by a team of vascular neurosurgeons who are cross-experienced in both the microsurgical and endovascular treatment of cerebral aneurysms. Indications for surgical and endovascular treatment have been reported previously [32]. Endovascular treatment of aneurysm recurrences was performed via a transfemoral approach under biplane fluoroscopy and road mapping. Aneurysm remnants of the appropriate shape were selectively catheterized and the maximum number of coils possible was delivered under systemic heparinization. Remodeling balloons and self-expandable nitinol stents served as neck bridging devices in lesions with unfeasible neck morphology.
A standard pterional craniotomy was used in the majority of aneurysms treated surgically. A unilateral anterior interhemispheric approach was used with pericallosal artery aneurysms, and supraorbital craniotomies allowing subfrontal clipping under endoscopic assistance were used in selected cases. Intraoperative angiography was routinely used, and indocyanine green (ICG) angiography was additionally performed during the last few years [27].
Unclippable aneurysm recurrences that are unfeasible for additional embolization were managed by PAO under bypass protection. In these cases, double-barrel STA-MCA low-flow bypasses were performed 48 h before endovascular balloon test occlusion [BTO] and subsequent PAO in the awake patient. In two cases of previously embolized, thrombosed, large basilar apex aneurysms, deliberate surgical basilar artery trunk occlusion was performed.
Results
As outlined in Table 2, several scenarios of insufficient aneurysm treatment are possible. Illustrative cases for these scenarios are given in Fig. 1, showing insufficient aneurysm treatment as an acute complication during the surgical or endovascular procedure [21], delayed aneurysmal hemorrhage from undetected recurrences [22], and progressive aneurysm repermeation detected upon angiographic follow-up [4, 14, 29].
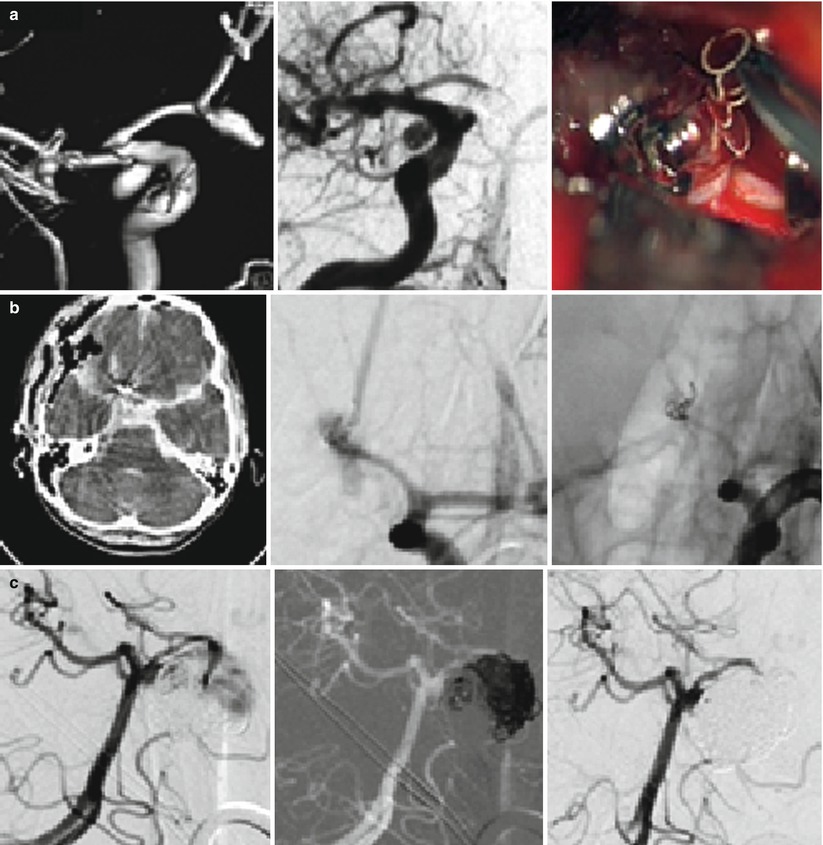
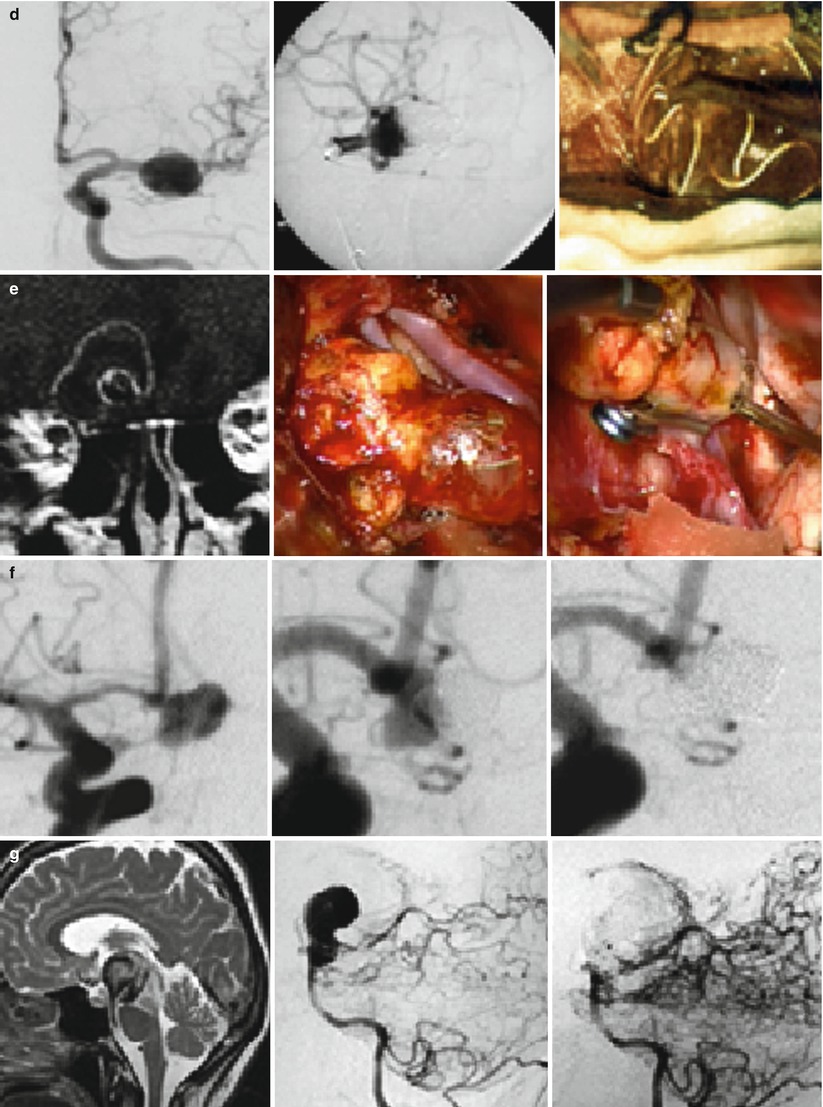
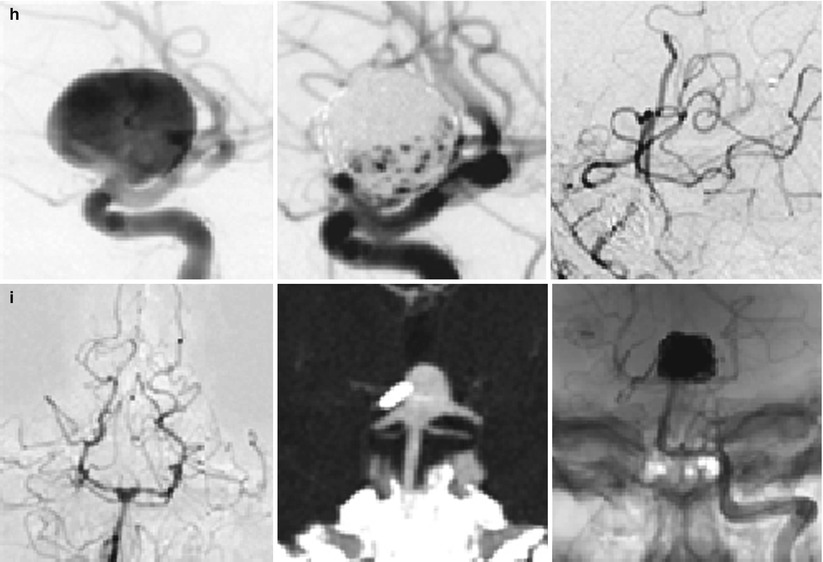
Table 2
Scenarios of insufficient aneurysm therapy requiring additional treatment
Acute clinical complications | |||
1. EVT complication | 2. Vessel occlusion | 3. Surgery [coil removal] | Fig. 1a |
1. EVT complication | 2. Acute repeat SAH | 3. Surgery [clipping] | Fig. 1b |
Delayed clinical complications | |||
1. Uneventful EVT | 2. Repeat SAH | 3. 2nd EVT [coiling] | Fig. 1c |
1. Uneventful EVT | 2. Repeat SAH | 3. Surgery [clipping] | Fig. 1d |
Delayed technical complications | |||
1. Uneventful EVT | 2. AN recurrence | 3. Surgery [clipping] | Fig. 1e |
1. Uneventful EVT | 2. AN recurrence | 3. 2nd EVT [coiling] | Fig. 1f |
1. Uneventful EVT | 2. AN recurrence | 3. 2nd EVT [stenting] | Fig. 1g |
1. Uneventful EVT | 2. AN recurrence | 3. Surgery [PAO + EC/IC BP] | Fig. 1h |
1. Uneventful Surgery | 2. AN recurrence | 3. EVT | Fig. 1i |



Fig. 1
Scenarios of insufficient aneurysm therapy requiring additional treatment as encountered in our population. As outlined in Table 2, (a, b) demonstrates endovascular complications managed by acute surgery, (c, d) depict delayed complications from aneurysm recurrence and repeat SAH managed by both surgery and endovascular re-intervention, and (e–i) illustrate the management of delayed aneurysm recurrences after both endovascular and surgical treatment. ACom anterior communicating artery, ICA internal carotid artery, PAO parent artery occlusion, SAH subarachnoid hemorrhage
Discussion
Aneurysm size [4, 23, 31], width of the neck [29, 31, 43], treatment during the acute phase after aneurysm rupture [4, 29], initially suboptimal angiographic aneurysm occlusion [2, 4, 14, 23, 29, 31], and length of follow-up [2, 29] have been correlated with an increased risk of aneurysm recurrence. The reported rates of aneurysm recurrences after initial endovascular treatment range from 6.1 to 33.6 % [4, 14, 29], indicating the lack of consensus on the definition of relevant aneurysm re-opening. This high incidence of aneurysm residuals contrasts with the low re-intervention rate of 4.7–17.4 % [4, 5, 25, 30] and demonstrates the role of individual judgment in deciding which aneurysm residual is of sufficient concern to warrant re-treatment.
As demonstrated in Fig. 1, the problem of incomplete or unstable aneurysm occlusion is found after both surgical and endovascular treatment.
Among surgically treated patients, Feuerberg et al. [7] assessed the natural history of surgical aneurysm rests and concluded that the rebleeding risk was between 0.38 and 0.79 % per year. Advanced techniques of intraoperative monitoring, including micro-Doppler ultrasound, neuroendoscopy, ICG videoangiography [27], and intraoperative angiography have been implemented to detect incorrect clip positions and in turn reduce the misclipping rates.
In the endovascular field, Byrne et al. [2] reported a rebleeding rate of 0.4 % (1/221) for stable non-progressing aneurysmal remnants after endovascular treatment. In marked contrast, he found a 7.9 % (3/38) rebleeding rate for angiographically unstable aneurysm residuals [3]. The degree of aneurysm occlusion after treatment was strongly associated with the risk of re-rupture [14], i.e., 24.5 % (12/90) of aneurysms subtotally occluded, but only 0.8 % (2/254) of aneurysms with complete or almost complete occlusion rebled. The annual re-rupture rates after endovascular treatment reported in the literature range from 0.11 to 0.32 % [5, 23, 24, 34], with the ISAT trial reporting a 0.2 % re-hemorrhage rate for the endovascular population in the first year.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








