Intraluminal placed atherosclerotic plaque during open carotid endarterectomy (a); Carotid endarterectomy with temporary indwelling shunt (arrow) (b)
Recently, in addition to open surgery for brachiocephalic artery stenosis, endovascular treatment is being actively introduced. Endovascular surgery includes intravascular access to the stenosis area via puncture of the femoral artery with subsequent expansion of the stenosis area (angioplasty) and stent placement. The comparison of open and endovascular surgeries in multicenter clinical studies indicates no significant differences in outcome of both procedures in the general patient population [8–10]. However, the carotid endarterectomy is preferable procedure for patients of advanced age (>70 years) and stenosis, caused by calcified plaques, especially, in severely deformed carotid arteries [8, 11, 12]. The advantages of the endovascular procedure for patients with increased risk of CEA (post-radiation stenosis, restenosis, and high stenosis at the C2 vertebra level) are demonstrated [8, 10, 13].
The temporarily clamping of the carotid artery is a common feature of both reconstructive surgeries causing the alteration of cerebral hemodynamics through collateral redistribution of blood supply to the brain tissue [14–16]. The inadequacy of the collateral circulation leads to development of acute cerebral blood flow (CBF) insufficiency that could result in the cerebral infarction [15]. It was shown that at the time of carotid artery clamping, hypoperfusion of the brain tissue registered in 5–7% cases causing an intraoperative ischemic stroke with severe disability or death [15, 17]. This accounts for up to 25% of all specific complications of carotid arteries’ surgery [18].
Currently, there is no consensus in utilizing a particular neuroprotection during carotid revascularization. The feasibility of routine [14, 19, 20] or no shunts [21–23] for all interventions and the selective use of temporary intraluminal shunt (TIS) during carotid arteries reconstruction depending on the indications [13, 24–26] are under the consideration.
For patients with uncertain tolerance for carotid clamping the use of a TIS is a safe method, that allows for minimizing the risk of possible hypoperfusion complications (Fig. 1b) [19, 27, 28]. Additionally, it does not limit the time of arteries reconstruction allowing meticulous atherosclerotic plaque removal and performing more delicate vascular suture, particularly with a shunt inserted into the lumen of the artery [14, 19, 28].
- 1.
More complex technical conditions to work in the area of ASP that could be associated with limited mobility and poor visualization of the arterial lumen in the area of the distal and proximal margin of the arteriotomy incision. That makes it difficult to separate the ASP and to control the residues of intact intima that may cause dissection of intima residues leading to arterial thrombosis.
- 2.
Failure of the TIS due to its thrombosis, torsion, incorrect position in the vessel lumen, and its clamping with tourniquets.
- 3.
Increased risk of cerebral thromboembolism.
- 4.
The risk of vascular intima damage by a shunt distally to the arteriotomy incision that may cause the intima dissection and early postoperative thrombosis of the reconstructed artery.
However, opponents of the routine use of a TIS suggest that the incidence of CBF decompensation is comparable and sometimes even less than the incidence of complications associated with the use of a shunt. Thus, performing the main stage of reconstruction without the use of TIS allows achieving a lower number of intraoperative ischemic complications compared to routine use of shunts [22].
At the same time, it is obvious, that for a certain group of patients, a complete denial of TIS leads to a higher rate of ischemic complications. For patients with occlusion of the contralateral internal carotid artery, ischemic complications of carotid endarterectomy increase up to 9% with extraordinary morbidity and mortality rates [20, 29, 30]. Therefore, the most optimal tactics of neuroprotection is the timely and correct use of the TIS based on the identification of preoperative risk factors and intraoperative markers of cerebral ischemia [19, 24, 26, 31, 32].
2 Instrumental Markers of Cerebral Ischemia
In order to prevent adverse events after reconstructive surgery of the carotid arteries it is necessary to consider preoperative and intraoperative risk factors affecting the development of cerebral ischemia.
Preoperative risk factors for cerebral ischemia. Initially the tactics of surgical treatment are determined based on a preoperative study of collateral circulation, the state of the circle of Willis, the presence of stenosis or occlusion of the contralateral carotid artery and arteries of vertebrobasilar system. That plays a significant role when it is impossible to determine obvious markers of cerebral ischemia according to intraoperative neurophysiological monitoring, which is typical in the case of CBF subcompensation.
- 1.
Methods for assessing cerebral circulation. Transcranial Doppler ultrasonography (TDUS) is the most general method that allows to locate the linear velocity of blood flow in the middle cerebral artery (MCA) on the repair side and measure the retrograde pressure. Recently the use of the latter technique is significantly limited due to the risk of cerebral embolism because of invasive introduction of a pressure sensor, qualitative character of blood flow assessment, and the low specificity of the method.
- 2.
Methods for assessing brain metabolism by transcranial cerebral oximetry (CO).
- 3.
Methods for assessing brain function, which include EEG and somatosensory evoked potentials.
Markers of cerebral ischemia are developed for each instrumental modality, that performance characteristics (sensitivity and specificity) are dependent on the degree of CBF decompensation. Within this paradigm of the decision making for brain protection approach there are preoperative risk factors, intraoperative markers of ischemia, and neurodynamic monitoring based levels of diagnostics.
Neurodynamic intraoperative monitoring. During awake carotid endarterectomy under local anesthesia, the direct contact with the patient is maintained to assess the neurological status continuously. Signs of depression, confusion, the appearance of speech defects, or pyramidal symptoms are regarded as an ischemic incident and are considered as indications for temporary indwelling shunt use. This type of intraoperative monitoring of the patient’s clinical condition is referred to neurodynamic monitoring and is considered an alternative of neurophysiological intraoperative monitoring. The neurodynamic monitoring is allowing surgeon time to select measures (install TIS, increase O2 pressure, shorten the procedure, etc.) to protect the brain against an ischemic incident. As mentioned earlier, the rapid installation of a temporary shunt and adequate anesthesiologic support might suppress the emerging clinical symptoms, but increase the risk of the “ischemic incident” into “acute ischemic stroke” transformation.
In this regard, the diagnostic criteria for cerebral ischemia according to various modalities have a special importance especially in cases of direct clinical evaluation of the patient during carotid endarterectomy with local anesthesia when the patient remains conscious.
2.1 Preoperative Assessment of Collateral Flow
Presence of natural cerebral collateral pathways plays an important role in prognosis of carotid occlusion outcome [30]. According to radiological studies, confirmed by autopsies, different variants of incomplete circle of Willis occur in more than 40% of cases, in 8–15% cases with severe collateral flow insufficiency through its anterior and posterior parts [30, 33]. Such patients suffer from increased risks of cerebral ischemic complications, even after temporary carotid occlusion. According to Sundt’s classification, performing the carotid endarterectomy for such a group of patients is accompanied by higher complication rates up to 6% despite obligatory use of TIS [23]. Denial of cerebral protection in this group can lead to perioperative ischemic stroke in more than 80% of cases [34]. Another angiographic marker of carotid clamping intolerance is hemodynamic failure of contralateral carotid arteries. According to a meta-analysis of more than 26,000 CEA procedures in 21 series the diagnosis of occlusion or severe stenosis of contralateral internal carotid artery raises the morbidity and mortality rates of carotid surgery up to 1.5 times [29].
Nevertheless, preoperative assessment of individual features of collateral flow could not be considered as a unique method for predicting cerebral ischemia during carotid endarterectomy, with sensitivity of 74%, specificity of 57%, and positive predictive value of 79% [35]. Thus, angiographic markers of possible ischemic complications must be evaluated together with intraoperative instrumental neuromonitoring techniques.
2.2 Transcranial Doppler Ultrasonography
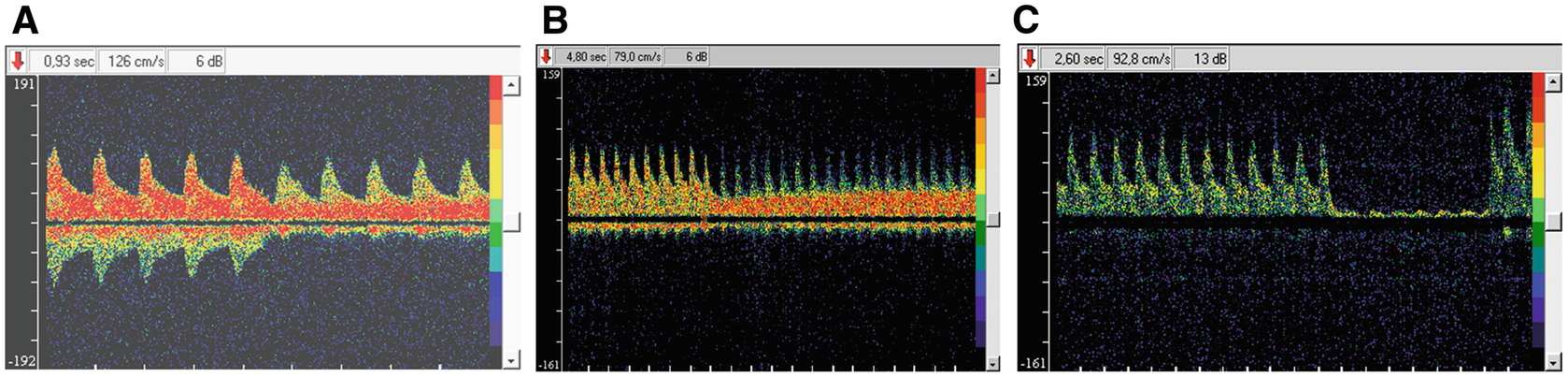
Compensation (a), subcompensation (b) and decompensation (c) stages of cerebral blood flow by TDUS ipsilateral to carotid endarterectomy
TDUS technique is based on monitoring the difference in the frequency between waves emitted and reflected by the Doppler probe from moving blood cells in vessels. The frequency is recorded as the linear velocity of blood flow (LVB) in the controlled cerebral vessels. TDUS application to the cerebral vessels allow measurement with specificity of 90–95% [32]. The LVB measurements to assess blood flow in the major arteries are ascertained by significant correlation of volumetric and linear velocities in the initial segments of the middle cerebral artery (MCA) and irrelevant alteration of cerebral vessels diameter due to changes of vascular wall tonus [38].
The main TDUS marker of cerebral ischemia during temporary carotid clamping is the value of linear blood flow decrease in ipsilateral MCA after carotid clamping. For patients with cerebral blood flow compensation after carotid occlusion the characteristic changes of LVB do not exceed 40% of initial value (Fig. 2a).
TDUS signs of CBF decompensation are presented by a decrease of the mean LVB of more than 85%—these changes are considered as carotid clamping intolerance and an objective indication for using TIS (Fig. 2c). At the same time the final values of the mean LVB at clamping should not exceed 15–20 cm/s—the value of the LVB accompanied with the changes on the electroencephalography (EEG). This threshold level of LVB is confirmed by somatosensory evoked potentials (SSEP) monitoring data and measurements of retrograde pressure [39–41].
Decrease of LVB in the ipsilateral MCA after carotid clamping between 40% and 85% is considered as CBF subcompensation form (Fig. 2b). The tolerance to temporary carotid clamping and the need of TIS at this stage is currently the most controversial issue. Some researchers suggest the use of TIS on the subcompensation stage routinely as on the decompensation stage. Using this threshold for cerebral ischemia TDUS method demonstrates high sensitivity for ischemic events with low specificity—only in 63% cases TDUS signs of cerebral ischemia could be confirmed by EEG changes [16]. In these cases other modalities of neuromonitoring are helpful for improving TDUS patency for ischemia detection. It is well known that compensation of cerebral circulation occurs by the circle of Willis and is primarily determined by TDUS and by means of cortical arterial collaterals. To assess the distal blood flow electrophysiological monitoring and cerebral oximetry are considered as more reliable monitoring techniques [31, 42, 43].
Another important feature of the TDUS is the ability to detect episodes of cerebral embolism [37]. Migration of emboli from the region of carotid artery reconstruction accounts for up to 50% of all specific complications after carotid endarterectomy [15, 44]. Depending on the structure, there are air and solid microemboli. Air microemboli are less dangerous and not manifested by the development of neurological symptoms or an asymptomatic radiological image of brain structures damage. The registration of solid microemboli that might be fragments of a decaying atherosclerotic plaque, as well as thrombotic deposits on its uneven surface, bear serious symptom of ischemic complications development [37, 44]. Intraoperative registration of more than 10 microemboli per minute might cause “silent” cerebral ischemia registered by magnetic resonance imaging (MRI) in the reconstructed internal carotid artery (ICA) territory and the development of neurological symptoms in 4% patients [37].
9–12% cases might have no an acoustic window to perform ultrasound location in the MCA.
Need to monitor the location of the sensor and the depth of artery location to avoid wrong detection of the posterior cerebral artery position (PCA). This situation can occur when the depth of the artery’s position is increased or patient’s head is smaller than usual resulting in PCA branches at a depth of 55–60 mm. There are often no changes in blood flow during temporary clamping of the carotid arteries in these cases. However, a paradoxical increase in blood flow is observed when the PCA is correctly located in certain cases that are regarded as signs of compensation when patient underwent clamping of the carotid arteries without complications.
False-positive results reducing the specificity of TDUS in the diagnosis of cerebral ischemia when the location of the internal carotid artery is proximal to the PCA. In such cases, a critical decrease in the linear velocity of blood flow may be recorded despite good compensation of the blood flow in the Willis circle.
Individual characteristics of the patient and technical problems due to vessel location might affect low values of blood flow in the MCA (less than 50 cm/s) decreasing the informativeness of TDUS.
2.3 Cerebral Oximetry
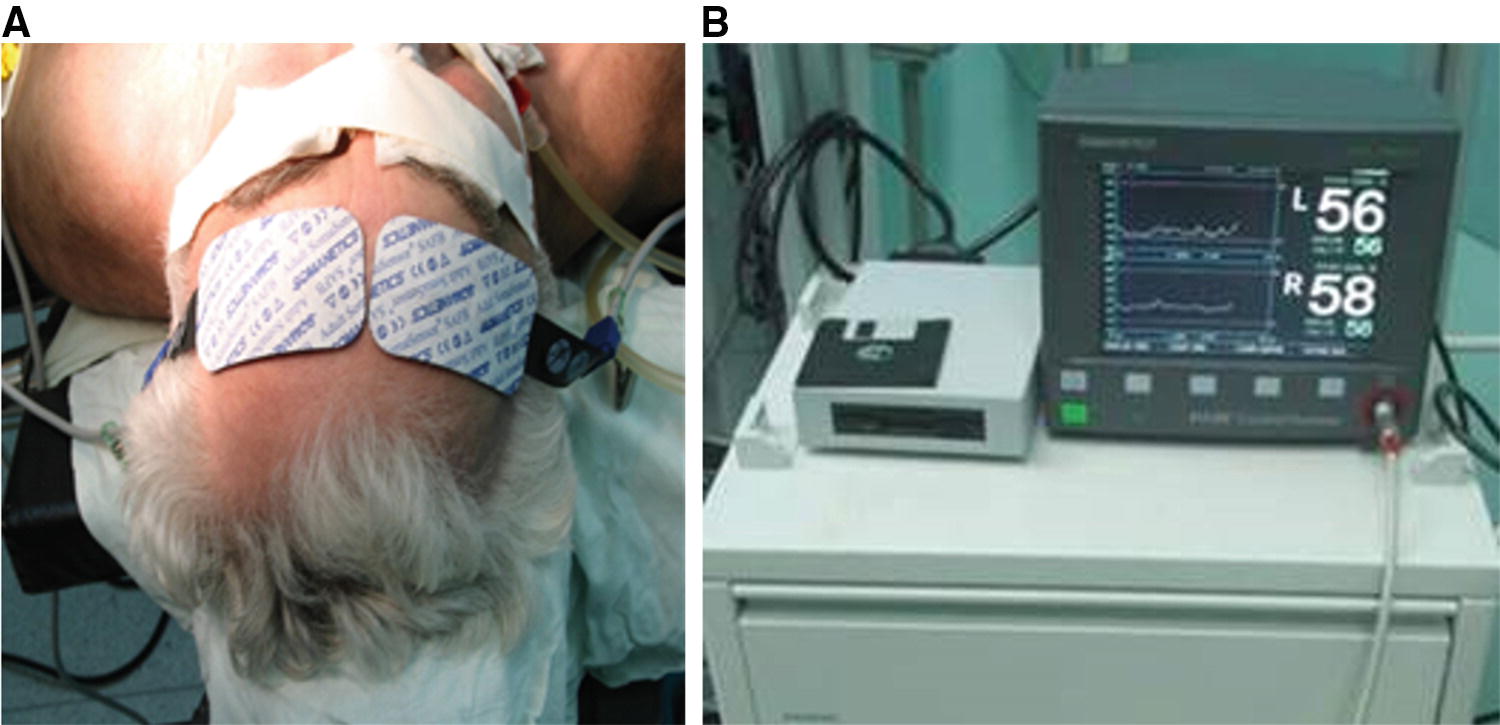
Cerebral oximetry monitoring of brain metabolism during CEA by NIRS: (a) typical placement of CO sensors; (b) monitoring terminal

(a) The pattern of blood flow compensation during carotid occlusion: 1—test clamp; 2—main carotid clamping, start of carotid endarterectomy. rSO2 level decrease by 6–8% below averaged initial values; 3—release of carotid clamps; 4—mild signs if hyperemia after CEA. Upper curve—ipsilateral, lower curve—contralateral hemisphere to CEA. (b) The pattern of blood flow decompensation during carotid occlusion: 1—test clamp; 2—main carotid clamping, start of carotid endarterectomy. rSO2 level decrease by 20–25% below averaged initial values; 3—insertion of TIS with rSO2 compensation; 4—release of carotid clamps; 4—mild signs of hyperemia after reperfusion with quick return to initial CO values. Upper curve—contralateral hemisphere to CEA, lower curve—ipsilateral
2.4 Brain Function Monitoring
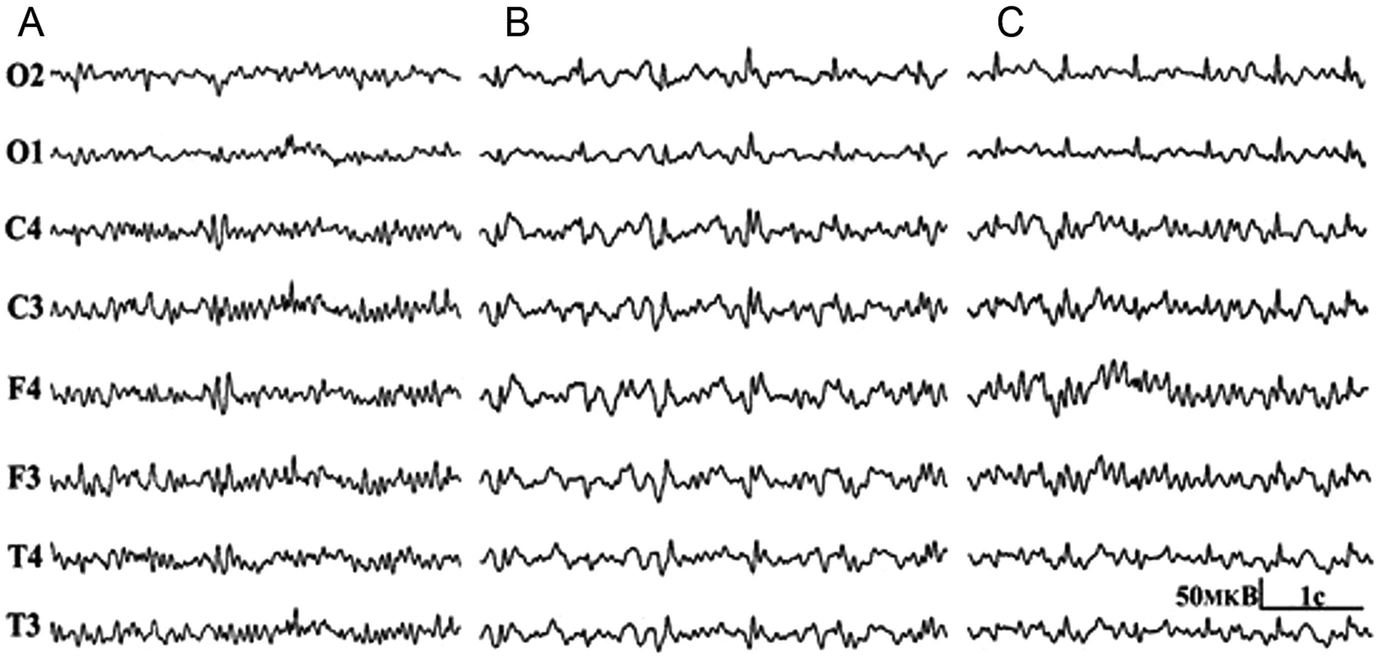
EEG represents a method of choice to determine the tolerance to clamping of the carotid arteries and the need to install a TIS [16, 24, 40]. The success of this reliable technique is facilitated by established correlation of EEG recordings with the development of ischemic complications during carotid endarterectomies performed without TIS [40]. The development of a pathological EEG pattern at the time of the carotid artery clamping varies from 8.5% to 31% and weakly relates to preoperative angiography data [16, 24].

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


