Visual loss as a primary complaint varies depending on whether one or both eyes are affected. Is the visual loss abrupt in onset, gradual or is it suddenly discovered long after onset? Is the visual loss complete or partial? Finally, does the visual loss include visual hallucinations or illusions? The causes of visual loss rapidly narrow down to a very small number of possibilities based on the temporal sequence of the patient’s symptoms, age, and sex, and the presumed anatomic location of the lesion (
Table 10.1).
Patient’s visual complaints usually first seek an ophthalmologist or optometrist. Referral to a neurologist from the ophthalmologist usually includes a CT or MRI already in hand. If the problem appears to be a tumor, the eye specialist will most often refer the patient directly to a neurosurgeon.
The visual pathway provides a number of diagnostic constellations of easily examined elements that can be carried out at the bedside. The pupils, the retina, and the optic disc can be objectively examined. Subjective visual tests include color, visual acuity, and visual fields, and these help to direct localization to the retina optic nerve, chiasm, optic tract, lateral geniculate, geniculocalcarine tract, and visual cortex. Damage to visual association cortices, especially parietal and inferior temporal, will produce higher function disturbances such as central achromatopsia, alexia without agraphia, prosopagnosia, and Anton’s syndromes.
I. BEDSIDE OR OFFICE CLINICAL EXAMINATION OF THE VISUAL SYSTEM
A. Visual acuity (high contrast/low contrast).
Although it is best to examine visual acuity using a distance Snellen chart, the neurologist almost always examines visual acuity using a handheld Rosenbaum card or a Jaeger print card. If a near card is used, be sure that the patient has their glasses on. In the office you can keep a pair of drugstore reading glasses of plus 2 or plus 3 for those patients over 40 years old who have forgotten their reading glasses. Push the patient to give you the very best acuity possible. Do not take no for an answer. Do not rush them. Start by telling them the first letter or number of the line and allow them to move the card back and forth. Do not begin with the largest letter; rather, ask the patient to read the 20/30 and the 20/25 lines and finally the 20/20 lines. If that fails, gradually work your way down the card. If the patient cannot read any letters on the Snellen card, the next approach is counting fingers. Three fingers held up are the equivalent of the big E on the distance acuity chart. A patient who can count fingers at 20 feet has 20/200 acuity. Thus counting fingers at 5 feet would be a rough equivalent of 20/800. If they cannot count fingers, try hand movements. If the patient cannot detect hand movement, try light perception by turning a hand light off and on. If the patient sees light reliably, see if the light can be localized in space, above, below, or side to side.
Visual acuity should be normal with a retrochiasmal pure HH or with a pure bitemporal hemianopia. Acquired visual acuity deficits not due to refractive error (nearsightedness or farsightedness) imply that macular or central vision is defective. Loss of Snellen acuity is commonly accompanied by other central visual loss such as defective color vision, Amsler’s grid defects, pallor of the optic disc, evidence of macular disease and, if unilateral, a RAPD. Bilateral, but grossly asymmetrical, retinal or optic nerve visual loss will also show an RAPD in the eye with the greatest field loss.
B. Confrontation visual fields
should be performed at a distance of 1 m from the patient. Have the patient cover one eye with the palm of the hand and direct him to look at
your nose. Divide the visual field in front of the patient into an imaginary plane of superior and inferior nasal and temporal quadrants. The center of the visual field is your nose. Present your fingers (one, two, or five) rapidly in the periphery of each quadrant. Redirect the patient’s attention to your nose and not to look at your fingers. Slowness or failure to respond accurately in one quadrant or hemifield may be the earliest sign of a homonymous field loss. After rapid finger counting, present your hands, palms forward, first in the two upper quadrants, then in the lower quadrants. Ask the patient to compare the palms for brightness and clarity. Finally, place the index finger of one hand on your nose and the index finger of the other hand on the peripheral nasal, temporal, superior, and inferior fields. Have the patient look at your nose and ask which finger is brightest and clearest. This tests for a central scotoma. You can also place one hand above and one below the horizontal meridian and have the patient look at your nose. This allows the patient to compare hands for brightness and clarity and helps identify altitudinal visual field defects as are seen with retinal and optic disc diseases, especially anterior ischemic optic neuropathy (AION) or BRAO.
C. Color vision testing.
A book of Ishihara or Hardy-Rand-Rittler pseudoisochromatic color plates can be used to test color vision one eye at a time. Although these tests were designed to identify red-green color blindness, they can be used as a rough indicator of color acuity, which is another test of central macular function. Test one eye at a time when looking for evidence of unilateral optic nerve damage. About 1 % or 2 % of men are red-green color blind or defective as are 0.5% of women. There are other, more complicated, color tests but neurologically they provide no more information.
D. Pupil tests.
The single most important and useful objective bedside test of anterior visual pathway function is the pupillary light response. When the ganglion cells or their axons in one eye, optic disc or optic nerve, are damaged, the pupil reaction to light will be less vigorous in the affected eye as compared with the unaffected or normal eye. If both optic nerves or retinas are damaged, a relative afferent pupil defect will be seen in the eye with the largest amount of visual field loss.
There is no such entity as a bilateral relative afferent pupil defect. The RAPD is a defect in the reaction to light of one eye
relative to the other. One should swing the light from eye to eye allowing it to rest for 1-second intervals and there will be a brisker reaction (a more complete response) to light in the normal unaffected eye and a less brisk reaction or dilation of the pupil in the affected eye. This is the
RAPD. If one eye is blind, the direct reaction to light will be absent—this is an
amaurotic pupil. For details of the RAPD, refer to
Chapter 12.
E. Ophthalmoscopy.
Ophthalmoscopy is an objective part of the neuro-ophthalmologic examination. Recognition of changes in the optic disc is a key to the diagnosis of diseases affecting the anterior visual pathways. Details of this part of the examination are not considered in this chapter. When in doubt, dilate the pupils (with two drops of 2.5% epinephrine or 1% mydracil eye drops) for a good look. Observe and record the size of the ratio of cup to disc, appearance of the vessels, whether there are hemorrhages, exudates, or pigment changes or swelling of the disc.
F. Visual evoked potentials (VEPs).
This test tends to be overused by neurologists in the evaluation of the visual pathways, but it can be especially valuable in the patient with a past history suggestive of optic neuritis (ON) where visual acuity is normal, there is no color vision loss and only slight optic disc pallor. Such patients frequently have a prolonged P100 latency. The VEP can be useful in patients suspected of functional visual loss. Patients, however, can confound the VEP by focusing in the distance, past the VEP screen, and thereby alter p100. A normal VEP latency in this setting is useful, but an abnormal latency may be a red herring.
G. Electroretinography
(ERG) is a test of retinal receptor function most commonly used to detect conditions like retinitis pigmentosa and paraneoplastic retinopathies. With certain bilateral retinopathies this may be a definitive test.
H. Formal visual field testing
consists of three types of tests.
Tangent screen exam is rarely used today. It is carried out on a flat black felt screen located 1 m away from the patient. It can be especially helpful in that it allows the examiner to back the patient up to 2 or 3 m and can be used to look for functional “tunnel” vision or to magnify small central visual defects.
Kinetic perimetry is carried out on a Goldmann perimeter using variable size and brightness lights and uses a supra-threshold kinetic technique. It is done in an ophthalmologist’s office. This form of perimetry is technician-dependent and has largely been supplanted by static perimetry.
Static perimetry done on Humphrey® or Octopus perimetry® tests threshold static targets presented at every 2° and tests the inner 24° to 30° of the visual field. This test is also done in an ophthalmologist’s office and is the usual formal perimetry done today.
II. ACUTE TRANSIENT MONOCULAR VISUAL LOSS
A. Clinical features.
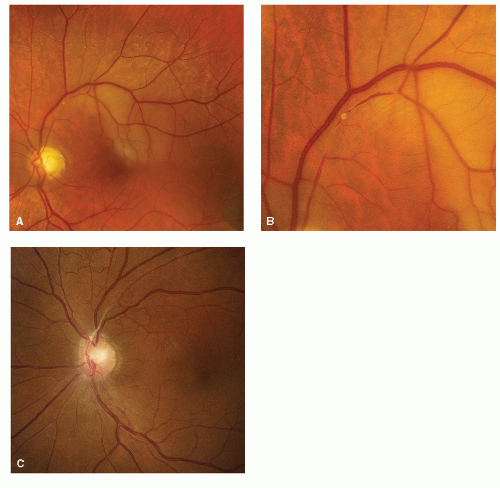
Acute transient monocular visual loss (TMVL), also called amaurosis fugax, is relatively common and has a host of different causes (
Table 10.1). Before concluding that the patient has had a monocular event, explore the possibility that the “monocular” event was really a transient binocular HH. This is particularly true if the visual loss is followed by a headache. Patients with homonymous visual defects rarely complain that half of objects are gone or that they cannot see well to one side. The patient may insist that they closed one eye or the other and the visual loss was only unilateral, but this is just seeing what they think they should be seeing. Monocular visual loss is occasionally reported as being sudden in onset when in reality it is
suddenly discovered when the unaffected normal eye was covered and the patient suddenly appreciates that they could not see well out of the affected eye. It is understandable why the patient would assume
visual loss was sudden. The mystery is why some people do not detect severe visual loss in one eye until it is brought to their attention. A most common cause of true transient monocular visual loss is artery-to-artery or cardiac-to-artery embolism, but there are many other causes of transient or permanent acute visual loss (
Fig. 10.1).
B. Approach to transient monocular visual loss.
1.Be sure that the spells occur in one eye and are not homonymous.
2. Look for ophthalmoscopic evidence of asymmetric optic disc cupping (glaucoma), optic disc anomaly, optic disc swelling, or residues of retinal embolism (hemorrhages, exudates, embolic plugs of cholesterol, platelets, fibrin, or calcium).
3. Look for a relative afferent pupil defect, visual field loss in both eyes, loss of visual acuity, and finally whether color vision is equal and normal in both eyes.
4. Look for proptosis (sign of intraorbital disease) causing intermittent amaurosis due to vascular compression particularly occurring with eye movement.
5. Auscultate the heart and carotid arteries for murmurs and bruits.
6. Laboratory studies.
CBC, including platelet count.
Sedimentation rate (ESR)—In all patients over age 50.
C-reactive protein (CRP)—In all patients over age 50. In patients of age 40 or less, strongly consider evaluation for hypercoagulable states:
Protein C.
Protein S.
Factor V Leiden.
Antithrombin.
Prothrombin gene mutation 20210A.
PT (INR) and aPTT.
Lupus anticoagulant (LA).
Anticardiolipin antibodies (IgG, IgM, and IgA).
Fibrinogen.


 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access





