Heterogeneity of aggression. Identifying the type of aggression a patient is displaying may help guide the selection of appropriate treatments that target the underlying dysfunctional circuits. However, violence and aggression arise from a complex combination of neurobiological, genetic, and environmental factors, and are often presented in the context of comorbid conditions.
Psychotic violence
Psychotic violence is attributed to positive symptoms of psychosis, most commonly paranoid delusions of threat or persecution, command hallucinations, and grandiosity [2–35,47]. Such psychotic symptoms may lead to violent behavior due to the assailant misunderstanding or misinterpreting environmental stimuli. In line with this, a recent study determined that 59% of individuals with schizophrenia who had committed acts of homicide were experiencing delusions, with a worsening of delusions in the months leading up to the homicidal act [19]. Psychotic violence is hypothetically linked to excessive neuronal activity in the mesolimbic dopamine pathway (Figure 9.2), where positive symptoms of psychosis are hypothetically mediated [47]. Psychotic violence linked to positive symptoms should hypothetically respond to suppression of this dopamine overactivity, and if standard doses of monotherapies or clozapine are ineffective, may be responsive to high-dosing or antipsychotic polypharmacy [2,3,37–46]. If standard doses fail to attain adequate plasma drug levels, i.e., a pharmacokinetic failure (Figure 9.3) [2,11–15,48–50], or if standard doses do attain adequate plasma drug levels but are nevertheless ineffective in reducing violence, i.e., a pharmacodynamic failure (Figure 9.3) [2,11–15,48–50], clozapine, high doses of monotherapies, or administration of two antipsychotics may be effective for positive symptoms driving psychotic violence (Figure 9.3) [2–35]. The rationale, consensus guidelines, evidence, and case-based examples are beginning to emerge from the literature for how to treat patients with psychotic violence unresponsive to standard, first-line, evidence-based treatments [2–35], but much further research in this area remains to be done, and the positive results from the reported studies need to be replicated.
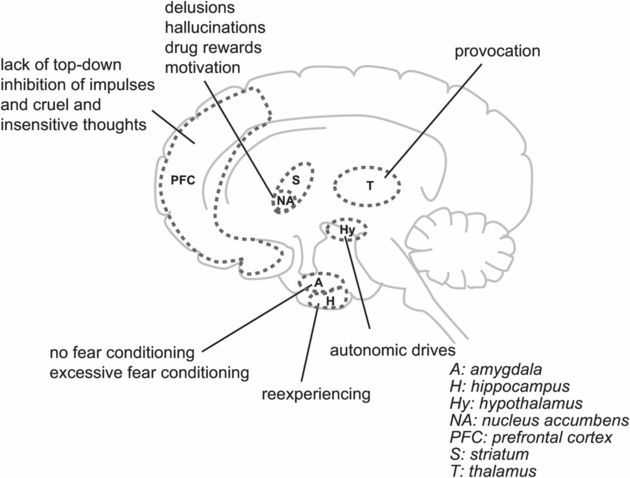
Brain areas related to violent and aggressive behavior. Impaired neurotransmission in various brain regions may contribute to the propensity for violent or aggressive behavior. The specific type of aggressive behavior is likely correlated with dysfunction in specific neural circuits. For example, both impulsive and psychotic aggression have been hypothesized to involve excessive reactivity to perceived threats (bottom-up out of control) and inadequate cortical regulation (top-down out of control). Although they are perhaps present in all patients with schizophrenia, structural and functional abnormalities in the frontal and temporal cortices as well as reduced connectivity between these brain areas may be more severe in aggressive patients than in those who are not aggressive. In the amygdala, fear conditioning seems to be excessive in both psychotic and impulsive aggression, whereas individuals with psychopathic aggression seem to lack fear conditioning.
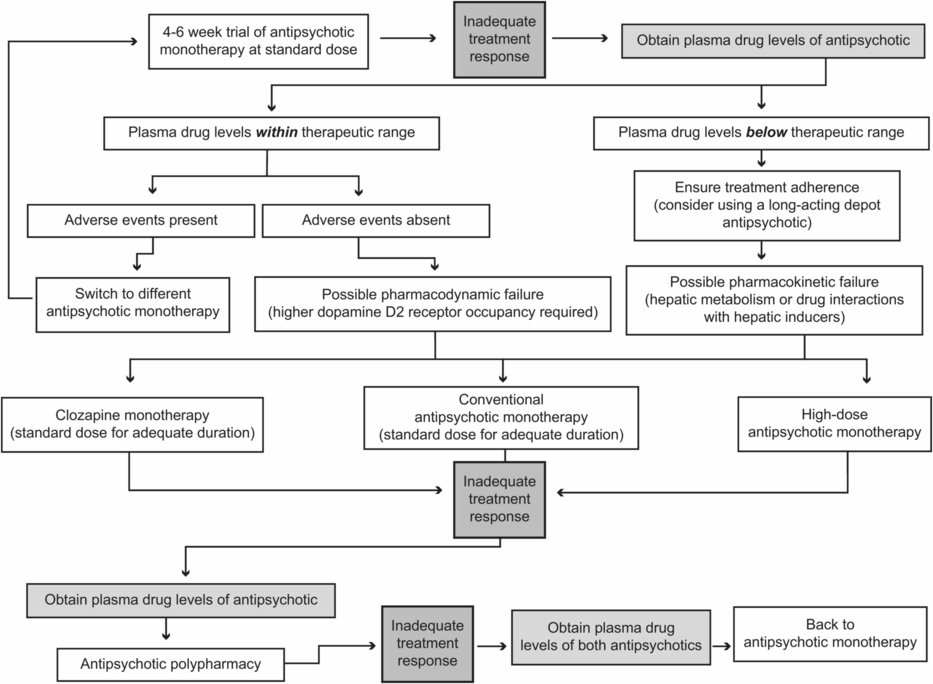
Antipsychotic treatment algorithm. Following several unsuccessful atypical antipsychotic monotherapy trials, a trial with a conventional antipsychotic or with clozapine is recommended. High-dose monotherapy may also be considered for such treatment-resistant patients. Antipsychotic polypharmacy is recommended only after antipsychotic monotherapy has failed. Note that throughout the treatment algorithm, monitoring of plasma drug levels of each antipsychotic is critical when determining the next course of action.
Impulsive violence
Sometimes called reactive violence, impulsive violence generally involves no planning, and is usually an immediate response to an environmental stimulus [38–43]. Impulsive violence may reflect emotional hypersensitivity and exaggerated threat perception [37–43], and may be linked with an imbalance between “top-down” cortical inhibitory controls and “bottom-up” impulsive drives (Figure 9.4) [7,43,51–58]. Both impulsive and psychotic aggression can occur in patients with schizophrenia, although impulsive violence is common in many other disorders, including mood disorders, personality disorders, substance abuse, and many more.
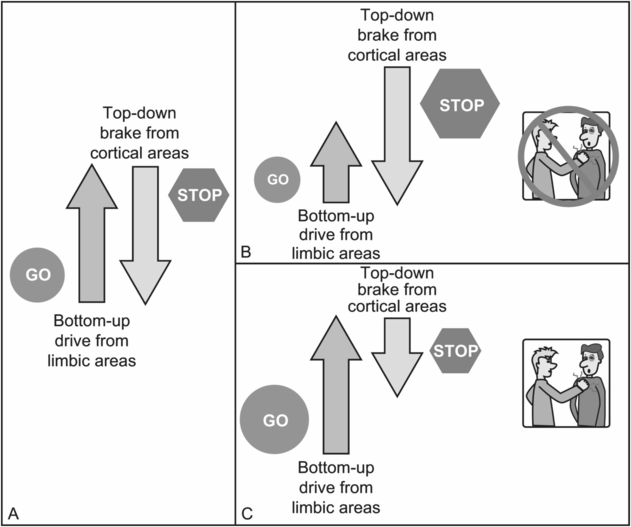
Bottom-up limbic drive and top-down cortical brake. (A) Impulsive drives in response to perceived threats stem from limbic regions, including the amygdala. Activity in limbic regions is modulated by input from cortical brain regions, including the prefrontal cortex (PFC). The balance between this limbic drive and the opposing cortical brakes determines whether one will act out an impulsive behavior such as aggression. (B) If the limbic drive is not overly strong, and/or if the cortical brake is sufficient to control impulsive drives coming from limbic areas, an individual will not act out with violent or aggressive behavior. (C) If the limbic drive is overly strong, and/or if the cortical brake system is not strong enough, an individual will be at an increased risk of violent behavior.
Both impulsive and psychotic violence have been hypothesized to involve excessive reactivity to perceived threat (bottom-up out of control) coupled with inadequate cortical regulation (top-down out of control) (Figure 9.4) [7,43,51–58]. Consistent with this, structural and functional abnormalities in frontal and temporal cortices and reduced connectivity between these brain areas have been reported [7,43,51–58]. Although perhaps present in all patients with schizophrenia, these findings may be more severe in aggressive patients compared with those who are nonviolent [7,43,51–58]. Impaired top-down control is most strongly associated with portions of the prefrontal cortex (PFC), including the dorsolateral PFC (DLPFC) and ventromedial PFC (VMPFC), as well as the orbitofrontal cortex (OFC) [7,43,51–58]. These regions are involved in decision making; dysfunction in these areas results in lack of recognition of consequences, inability to use previously learned information about reward and punishment, misinterpretation of emotionally neutral stimuli as being negative, and impaired recognition of social cues [7,43,51–58].
Within the temporal lobe, the amygdala is most highly implicated in violent and aggressive behaviors. The amygdala is involved in the rapid detection of threat, as well as the excitation of fight-or-flight responses. Modulation of amygdala activity comes from prefrontal brain areas; connections between these prefrontal and limbic regions may be impaired in individuals who are impulsively aggressive [7,43,51–58]. The amygdala is hyperactive in the case of impulsive aggression, but hypoactive in the case of psychopathic (predatory) aggression [43,48,51].
As in psychotic violence, impulsive violence may be responsive to antipsychotic polypharmacy or high dosing when standard doses of monotherapies or clozapine fail to control impulsive violence [4–35]. Additional studies of impulsive violence and high-dose antipsychotics or antipsychotic polypharmacy are greatly needed.
Predatory violence
Predatory, or psychopathic, violence involves aggressive acts that are characterized by planning of assaults, predatory gain, and lack of remorse [2,37–46]. A moderate proportion of all violent acts and a high proportion of the most severe violent acts are due to psychopathy (Figure 9.4) [2,32–46]. The neurobiological basis of psychopathy is currently under intense investigation, and findings suggest that predatory/psychopathic violence may be associated with deficient fear conditioning in the amygdala [43,48,51].
Even though predatory violence in patients with psychotic illnesses may be common in forensic settings, this can occur in psychotic patients who have positive symptoms under control, and whose predatory violence will not respond to antipsychotics, including high dosing and polypharmacy. In fact, it is not clear whether predatory violence responds to any kind of treatment, least of all, psychopharmacologic interventions [2,37–46]. Psychopathic violence is not responsive to antipsychotics when comorbid psychotic symptoms are under control. Such patients may require restricted housing or “therapeutic security” rather than antipsychotics [1,2].
Confounding factors
When selecting treatments for violent patients in forensic settings, it is important to consider the numerous confounding factors that may contribute to violent behavior, such as substance use disorders, personality disorders, cognitive dysfunction, mood disorders, noncompliance, etc. [2,37–46,59–61] Substance use issues are highly prevalent in patients with mental illness; approximately half of patients with schizophrenia have a comorbid substance use disorder [37–46]. Substances of abuse in particular may exacerbate symptoms of schizophrenia and lead to violence due to the effects of drugs of abuse on impulse control [16]. In fact, the risk of violent behavior in patients with schizophrenia is four times greater if there is comorbid substance abuse [20]. Addressing substance use disorders is an integral part of the treatment plan for patients with schizophrenia, and may help prevent violence in this population [2,37–46].
Neurotransmitters and Violence
An oversimplification of the role of neurotransmitters in violence, particularly impulsive violence, involves hypothetical imbalances in the neurotransmitters dopamine (DA) and serotonin (5HT) [7,43,48]. In the prefrontal cortexes of aggressive patients, 5HT is decreased whereas DA is increased [7,43,48,62]. Dopamine is involved in the initiation and performance of aggressive behaviors, and elevated levels of striatal DA have been reported in individuals with impulsive disorders; this hyperdopaminergia may weaken inhibitory pathways that regulate impulsivity [7,43,48,62–65]. Notably, excessive DA in mesolimbic areas of the brain is also believed to underlie psychosis; not surprisingly, antagonism of dopamine D2 receptors forms the basis of antipsychotic treatments. This may be why high degrees of blocking dopamine D2 receptors may also have therapeutic actions in impulsive violence as well.
Serotonin modulates prefrontal activity; thus the serotonergic dysfunction observed in the OFC and in the anterior cingulate of aggressive patients suggests a lack of sufficient top-down control [7,43,48,62–65]. In fact, during aggressive confrontations, 5HT levels in the PFC may decrease by as much as 80% [7]. Aggressive behavior and suicide by violent means has been correlated with low cerebrospinal fluid (CSF) levels of 5HIAA, which is a measure of 5HT concentration [51,63]. Additionally, whereas 5HT depletion increases aggressive behavior, increasing 5HT levels brings about increased activity in the PFC as well as diminished aggression [64]. This reduction in serotonergic activity observed in impulsively aggressive individuals is not found in patients with predatory aggression, which further supports the heterogeneic nature of violent behavior [23,57]. The serotonin 5HT2A receptor in particular may be implicated in aggressive behavior: a recent study showed that availability of the 5HT2A receptor is greater in aggressive patients compared to non-aggressive patients or healthy controls [64]. Contrary to the antiaggressive consequences of antagonism at 5HT2A receptors, agonism at 5HT2C receptors has also been shown to reduce impulsivity [65]. Therefore, targeting of specific serotonergic subtypes, a feature of many atypical antipsychotics, has potential for antiaggressive therapies.
Targeting Psychotic and Impulsive Violence
Once violence has been assessed in the forensic setting and a psychiatric diagnosis has been made [1–36], the next step is to deconstruct the type of violence into its psychotic, impulsive, or predatory subtypes [36]. Once the type of violence in the context of the specific DSM psychiatric diagnosis is known, the succeeding step is to find a rational treatment. The RDoC approach is to consider which brain circuits may be malfunctioning and thus hypothetically result in the unique symptom profile of an individual patient, which neurotransmitters may hypothetically regulate the efficiency of information processing in those brain circuits, and then finally, which psychopharmacologic treatments to select to rationally reduce symptoms of violence by targeting the hypothetically malfunctioning brain circuits of a given patient [36]. Interventions not only include rational psychopharmacologic measures, usually to occupy high proportions of dopamine D2 receptors, or act at serotonergic systems or ion channels, but also utilize various psychotherapies such as dialectical behavioral therapy (DBT) that can have potentially powerful therapeutic actions in selected patients [2].
In terms of empiric psychopharmacologic interventions that seem to be effective in some psychotic and impulsive patients who fail first-line, evidence-based treatments from randomized controlled trials, one current strategy is to achieve high degrees of dopamine D2 receptor occupancy by utilizing higher than normal doses of an antipsychotic, or even two concomitant antipsychotics, one often a depot formulation, while monitoring plasma drug levels [2–35].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







